Chapter: Organic Chemistry: Alkenes and alkynes
Conjugated dienes
CONJUGATED DIENES
Key Notes
Structure
A
conjugated diene consists of two alkene units separated by a single bond.
Bonding
The
‘single’ bond connecting the two alkene units of a conjugated diene has partial
double-bond character. There are two possible explanations. Firstly, the use of
sp2 hybridized orbitals to
form the σ bond
results in a shorter bond. Secondly, the π systems of the two alkene units can overlap to
give partial double-bond character.
Electrophilic addition
Electrophilic
addition to a diene produces a mixture of two products arising from
1,2-addition and 1,4-addition. The reaction proceeds through an allylic
carbocation which is stabilized by delocalization and which accounts for the
two possible products obtained.
Diels–Alder cycloaddition
Heating
a conjugated diene with an electron-deficient alkene results in the formation
of a cyclohexene ring. The mechanism is concerted and involves no
intermediates.
Structure
A conjugated diene
consists of two alkene units separated by a single bond (Fig. 1a). Dienes which are separated by
more than one single bond are called nonconjugated dienes (Fig. 1b).
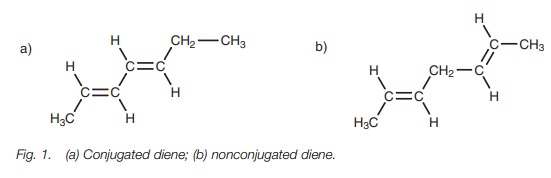
Bonding
A conjugated diene does not behave like two isolated alkenes. For example, the length of the ‘single’ bond connecting the two alkene units is slightly shorter than one would expect for a typical single bond (1.48 Å vs. 1.54 Å). This demonstrates that there is a certain amount of double-bond character present in this bond. Two explanations can be used to account for this. First of all, the bond in question links twosp2 hybridized carbons rather than two sp3 hybridized carbons.
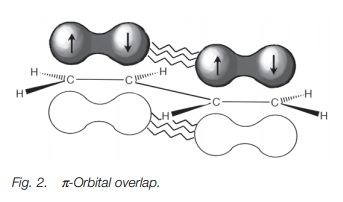
Therefore, ansp2hybridized
orbital from each carbon is used for the single bond. Since thishybridized
orbital has more s-character than an sp3 hybridized orbital, the
bond is expected to be shorter. An alternative explanation is that the π orbitals of the two alkene systems can overlap to produce the
partial double-bond character (Fig. 2).
Electrophilic addition
The reactions of a conjugated diene reflect the
fact that a conjugated diene should be viewed as a functional group in its own
right, rather than as two separate alkenes. Electrophilic addition to a
conjugated diene results in a mixture of two possible products arising from
1,2-addition and 1,4 addition (Fig. 3).
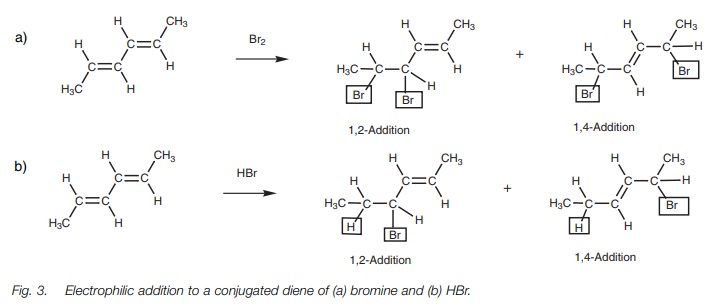
In 1,2-addition, new atoms have been added to
each end of one of the alkene units. This is the normal electrophilic addition
of an alkene with which we are familiar. In 1,4-addition, new atoms have been
added to each end of the entire diene system. Furthermore, the double bond
remaining has shifted position (isomerized) to the 2,3 position.
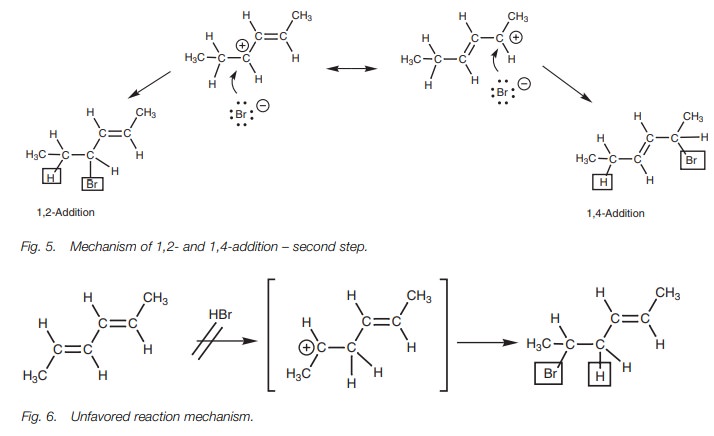
The mechanism of 1,4-addition starts off in the same way as a normal elec- trophilic addition. We shall consider the reaction of a conjugated diene with hydrogen bromide as an example (Fig. 4). One of the alkene units of the diene uses its π electrons to form a bond to the electrophilic hydrogen of HBr. The H–Br bond breaks at the same time to produce a bromide ion. The intermediate carbocation produced has a double bond next to the carbocation center and is called an allylic carbocation.
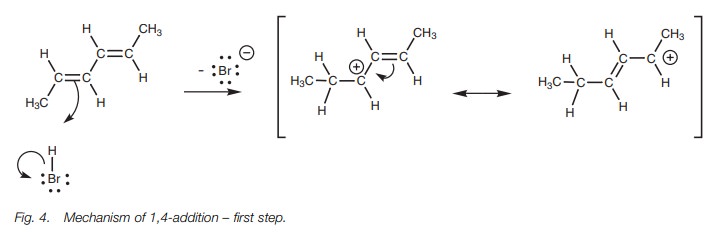
This system is now set up for resonance involving the remaining alkene and the carbocation center, resulting in delocalization of the positive charge between positions 2 and 4.
Due to this delocalization, the carbo-cation is stabilized and this in turn explains two features of this reaction. First of all, the formation of two different products is now possible since the second stage of the mechanism involves the bromide anion attacking either at position 2 or at position 4 (Fig. 5). Secondly, it explains why the alternative 1,2-addition product is not formed (Fig. 6). The intermediate carbocation required for this 1,2-addition cannot be stabilized by resonance. Therefore, the reaction proceeds through the allylic carbocation instead.
Diels–Alder cycloaddition
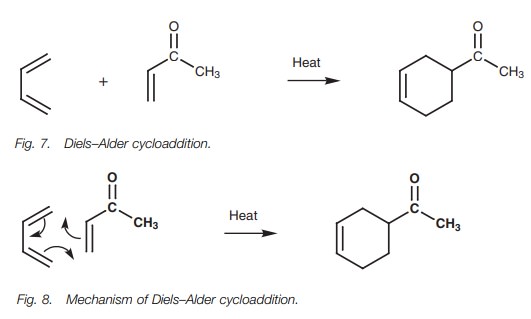
The Diels–Alder cycloaddition reaction is an
important method by which six- membered rings can be synthesized. The reaction
involves a conjugated diene and an alkene (Fig.
7). The alkene is referred to as a dienophile
(diene-lover) and usually has an electron-withdrawing group linked to it (e.g.
a carbonyl group or a nitrile). The mechanism is concerted with new bonds being
formed at the same time as old bonds are being broken (Fig. 8). No intermediates are involved.
Related Topics