Chapter: Modern Analytical Chemistry: Obtaining and Preparing Samples for Analysis
Classifying Separation Techniques: Separations Based on Size
Separations Based on Size
The simplest physical
property that can be exploited in a separation is size. The separation is accomplished using
a porous medium through which only the analyte or inter- ferent can pass.
Filtration, in which
gravity, suction, or pressure is used to pass a sample through
a porous filter
is the most commonly encountered separation technique based on size.
Particulate interferents can be separated from dis- solved analytes by filtration, using a filter whose pore size retains the interferent. This separation technique is important in the analysis of many natural waters, for which the presence of suspended solids may interfere in the analysis. Filtration also can be used to isolate analytes present as solid particulates from dissolved ions in the sample matrix. For example, this is a necessary step in gravimetry, in which the analyte is isolated as a precipi- tate.
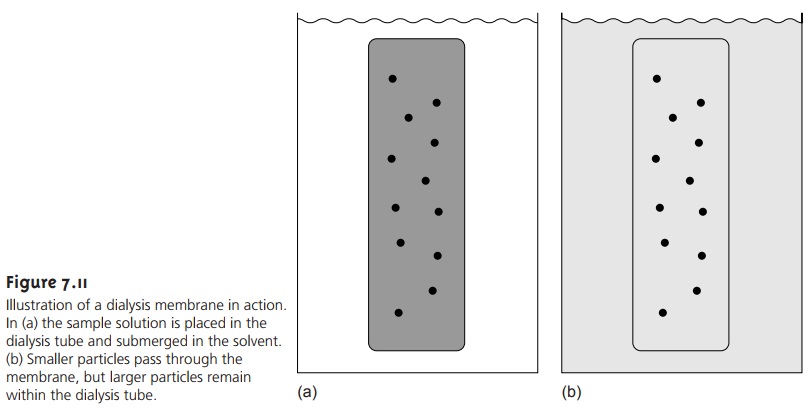
Another example of a separation technique based on size is dialysis,
in which a semipermeable membrane is used to separate
the analyte and interferent. Dialysis membranes are usually constructed from cellulose, with pore sizes
of 1–5 nm. The sample
is placed inside a bag or tube constructed from the membrane.
The dialysis membrane
and sample are then placed
in a container filled with a solution
whose composition differs
from that of the sample.
If the concentration of a particular
species is not the same on the two sides of the membrane, the resulting concentra- tion gradient provides
a driving force
for its diffusion across the membrane. Al- though small particles
may freely pass through the membrane, larger particles are unable to pass (Figure
7.11). Dialysis is frequently used
to purify proteins, hormones, and enzymes. During kidney dialysis, metabolic waste products, such as urea,
uric acid, and
creatinine, are removed
from blood by passing it over a dialysis membrane.
Size-exclusion
chromatography, which also is called gel
permeation or molecular- exclusion chromatography, is a third example
of a separation technique based on
size. In this technique a column is packed with
small, approximately 10-ÎĽm,
porous particles of cross-linked dextrin or polyacrylamide. The pore size of the particles is controlled by the degree
of cross-linking, with
greater cross-linking resulting in smaller pore sizes. The sample to be separated is placed into a stream
of solvent that is
pumped through the column at a fixed
flow rate. Particles too large to enter the pores are not retained and pass through
the column at the same
rate as the
solvent. Those particles capable of entering
into the pore structure
take longer to pass through the column. Smaller
particles, which penetrate more deeply into the pore structure, take the longest
time to pass through the column. Size-exclusion chro- matography is widely
used in the
analysis of polymers and in biochemistry, where it is used for the separation of proteins.
Related Topics