Chapter: 11th Chemistry : UNIT 14 : Haloalkanes and Haloarenes
Chemical properties of Haloarenes
Chemical properties
A. Reactions invoving halogen atom
1. Aromatic nucleophilic substitution reaction
Halo arenes do not undergo nucleophilic substitution reaction readily. This is due to C–X bond in aryl halide is short and strong and also the aromatic ring is a centre of high electron density.
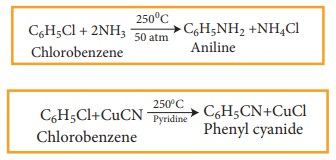
The halogen of haloarenes can be substituted by OH– , NH2–, or CN– with appropriate nucleophilic reagents at high temperature and pressure.
For Example
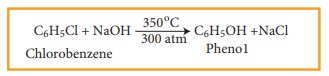
This reaction is known as Dow’s Process
2. Reaction with metals
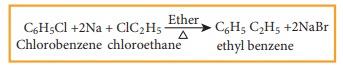
a) Wurtz Fittig reaction
Halo arenes reacts with halo alkanes when heated with sodium in ether solution to form alkyl benzene. This reaction is called wurtz fittig reaction.
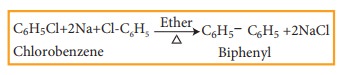
b) Fittig reaction
Haloarenes react with sodium metal in dry ether, two aryl groups combine to give biaryl products. This reaction is called fittig reaction
B) Reaction involving aromatic ring
3. Electrophilic substitution reaction
Haloarenes undergo aromatic electrophilic substitution reactions. The rate of eleclophilic substitution of halobenzene is lower than that of benzene. halogen is deactivating due to - I effect of halogen. The lone pair of electrons on the chlorine involves in resonance with the ring. It increases the electron density at ortho and para position (refer figure no 14.1). The halogen attached to the benzine ring with draw electron and thereby and hence the halogen which is attached to the benzene directs the incoming, electrophile either to ortho or to para position in electrophilie substitution reaction
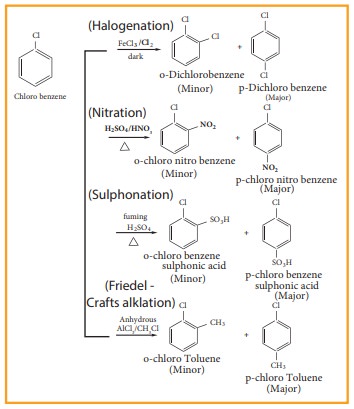
Toluenes.
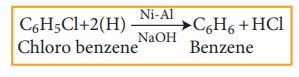
4) Reduction
Haloarenes on reduction with Ni-Al alloy in the presence of NaOH gives corresponding arenes.
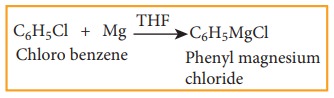
5) Formation of Grignard reagent
Haloarenes reacts with magnesium to form Grignard reagent in tetra hydrofuran (THF).
Related Topics