Chapter: Pharmaceutical Biotechnology: Fundamentals and Applications : Formulation of Biotech Products, Including Biopharmaceutical Considerations
Solubility Enhancers - Excipients Used in Parenteral Formulations of biotech Products
Solubility Enhancers
Proteins, in particular those that are non-glycosylated, may have a tendency to aggregate and precipitate. Approaches that can be used to enhance solubility include selection of the proper pH and ionic strength conditions. Addition of amino acids such as lysine or arginine (used to solubilize tissue plasminogen activator, t-PA), or surfactants such as sodium dodecylsulfate to solubilize non-glucosylated IL-2 can also help to increase the solubility. Themechanism of action of these solubility enhancers depends on the type of enhancer and the protein involved and is not always fully understood.
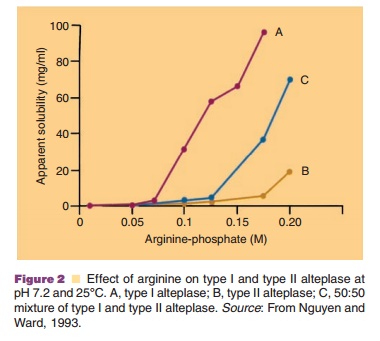
Figure 2 shows the effect of arginine concentra-tion on the solubility of t-PA (alteplase) at pH 7.2 and 25 C. This figure clearly indicates the dramatic effect of this basic amino acid on the apparent solubility of t-PA.
In the above examples, aggregation is physical in nature, i.e. based on hydrophobic and/or electrostatic interactions between molecules. However, aggrega-tion based on the formation of covalent bridges between molecules through disulfide bonds, and ester or amide linkages has been described as well (see Table 4). In those cases, proper conditions should be found to avoid these chemical reactions.
Related Topics