Chapter: Biochemistry: Viruses, Cancer, and Immunology
Viruses are Used for Gene Therapy
Viruses Are
Used for Gene Therapy
Although viruses have usually been seen as problems for humans,
there is one field now in which they are being used for good. Viruses can be
used to make alterations in somatic cells, whereby a genetic disease is treated
by the introduction of a gene for a missing protein. This is called gene therapy. The most successful form
of gene therapy to date involves the gene for adenosinedeaminase (ADA), an enzyme involved in purine catabolism.
If this enzyme is missing, dATP builds up in tissues, inhibiting the action of
the enzyme ribonucleotide reductase. This results in a deficiency of the other
three deoxyribonucleoside triphosphates (dNTPs). The dATP (in excess) and the
other three dNTPs (deficient) are precursors for DNA synthesis. This imbalance
particularly affects DNA synthesis in lymphocytes, on which much of the immune
response depends. Individuals who are homozygous for adenosine deaminase
deficiency develop severe combined
immune deficiency (SCID), the “bubble-boy”syndrome. These individuals are
highly prone to infection because of their highly compromised immune systems.
The ultimate goal of the planned gene therapy is to take bone marrow cells from
affected individuals; introduce the gene for adenosine deaminase into the cells
using a virus as a vector; and then reintroduce the bone marrow cells in the
body, where they will produce the desired enzyme. The first clinical trials for
ADA-SCID were simple enzyme-replacement therapies begun in 1982. The patients
were given injections of ADA. Later clinical trials focused on correction of
the gene in mature T cells. In 1990, transformed T cells were given to
recipients via transfusions.
In trials at the National Institutes of Health (NIH), two girls,
ages 4 and 9 at the start of treatment, showed improvement to the extent that
they could attend regular public schools and have no more than the average
number of infections. Administration of bone marrow stem cells in addition to T
cells was the next step; clinical trials of this procedure were undertaken with
two infants, ages 4 months and 8 months, in 2000. After 10 months, the children
were healthy and had restored immune systems.
There are two types of delivery methods in human gene ther-apy. The
first, called ex vivo, is the type
used to combat SCID. Ex vivo delivery means that somatic cells are removed from
the patient, altered with the gene therapy, and then given back to the patient.
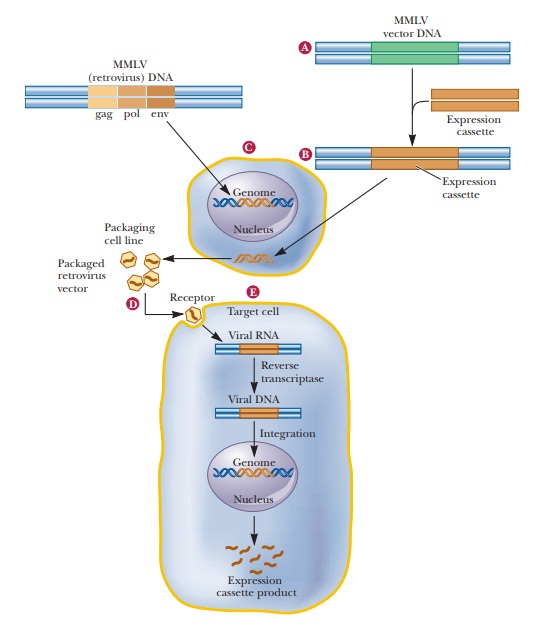
The most common vector for this is the retrovirus Maloney murine leukemia virus (MMLV). The figure shows howthe virus
is used for gene therapy. Some of the MMLV is altered to remove the gag, pol, and env genes, rendering the virus unable to replicate. These genes are
replaced with an expression cas-sette, which
contains the gene being administered, such as theADA gene, along with a
suitable promoter. This mutated virus is used to infect a packaging cell line.
Normal MMLV is also used to infect the packaging cell line, which is not
susceptible to the MMLV. The normal MMLV does not replicate in the packaging
cell line, but its gag, pol, and env genes restore the mutated virus’s
ability to replicate, but only in this cell line. These controls are necessary
to keep mutant viruses from escap-ing to other tissues. The mutated virus particles
are collected from the packaging cell line and used to infect the target cells,
such as bone marrow cells in the case of SCID. MMLV is a ret-rovirus, so it
infects the target cell and produces DNA from its RNA genome, and this DNA can
then incorporate into the host genome, along with the promoter and ADA gene. In
this way, the target cells that were collected have been transformed and
produce ADA. These cells are then put back into the patient.
The second delivery method, called in vivo, means that the virus is used to directly infect the
patient’s tissues. The most common vector for this delivery is the adenovirus (which is a DNA virus). A
particular vector can be chosen based on spe-cific receptors on the target
tissue. Adenovirus has receptors in lung and liver cells, and it has been used
in clinical trials for gene therapy of cystic fibrosis and ornithine
transcarbamoylase deficiency.
Clinical trials using gene therapy to combat cystic fibrosis and
certain tumors in humans are under way. In mice, gene therapy has been
successful in fighting diabetes. The field of gene therapy is exciting and full
of promise, but there are many obstacles to success in humans. There are also
many risks, such as the risk of a dangerous immunological response to the vector
carrying the gene or the danger of a gene becoming incorpo-rated into the host
chromosome at a location that activates a cancer-causing gene.
The second delivery method, called in vivo, means that the virus is used to directly infect the
patient’s tissues. The most common vector for this delivery is the adenovirus (which is a DNA virus). A
particular vector can be chosen based on spe-cific receptors on the target
tissue. Adenovirus has receptors in lung and liver cells, and it has been used
in clinical trials for gene therapy of cystic fibrosis and ornithine
transcarbamoylase deficiency.
Clinical trials using gene therapy to combat cystic fibrosis and
certain tumors in humans are under way. In mice, gene therapy has been
successful in fighting diabetes. The field of gene therapy is exciting and full
of promise, but there are many obstacles to success in humans. There are also
many risks, such as the risk of a dangerous immunological response to the
vector carrying the gene or the danger of a gene becoming incorporated into the
host chromosome at a location that activates a cancer-causing gene.
Gene therapy via retroviruses. The Maloney murine leukemia virus(MMLV) is used
for ex vivo gene therapy. (a) Essential genes (gag, pol,env) are removed from the virus and (b) replaced with an
expression cas-sette containing the gene being replaced with gene therapy.
Removal of the essential viral genes renders the viruses unable to replicate.
(c) The altered virus is then grown in a packaging cell line that allows
replication.
(d)
Viruses are collected and then used to infect cultured target cells from the
patient needing the gene therapy. (e) The altered virus produces RNA, which
then produces DNA via reverse transcriptase. The DNA then integrates in the
patient’s cells’ genome, and then his or her cells produce the desired protein.
The cultured cells are then given back to the patient. (Adapted from Figure 1 in Crystal, R. G., 1995. Transfer of genes to
humans: Early lessons and obstacles to success. Science270, 404.)
Related Topics