Chapter: Psychology: Learning
The Neural Basis for Learning
THE NEURAL
BASIS FOR LEARNING
We just suggested that different
species all need to learn the same kinds of lessons— including how events in
the world are related to each other, and what the consequences are of the
organism’s own actions. But do all organisms accomplish this learning through
the same neural mechanisms? If we zoom in for a closer look, will we find the
biology of learning is the same from one species to the next? As is often the
case, there are important commonalities between species; but there are also
significant differences: The biological mechanisms that allow learning in
mammals are somewhat different from the mechanisms crucial for reptiles,
amphibians, or invertebrates (Macphail, 1996; Woolf, 1998). Indeed, even within
a single species, the biological mechanisms needed for learning can vary and
seem to depend on the CS, the US, and the procedure (R. Clark, Manns, &
Squire, 2003; Fanselow & Poulos, 2005). Thus, the brain circuits underlying
fear conditioning (with electric shock as the US) are centered in the amyg-dala;
the brain circuits underlying eyeblink conditioning (with a puff of air to the
eye as the US) are centered in the cerebellum. Conditioning with a long delay
between the CS and US typically involves the hippocampus, while conditioning
with a shorter delay may not (Berman & Dudai, 2001; Lattal, Honarvar, &
Abel, 2004).
Even with these variations, some
biological principles do apply to all cases of learn-ing. In all cases,
learning depends on neural plasticity—the
capacity for neurons to change the way they function as a result of experience.
In all cases, the plasticity involves changes at the synapse—that is, changes
in the way neurons communicate with each other. These changes, in turn, can
involve any of three adjustments: Some neurons, after learning, end up sending
a stronger signal than they did before. Other neurons become more sensitive to
the signals they’ve been receiving all along. And, third, learning can lead to
the creation of entirely new connections among neurons— new synapses—allowing
for new lines of communication within the nervous system.
Evidence for these points comes from many sources, including studies of the marine mollusk Aplysia. Because the nervous systems of these creatures contain a mere 20,000 neurons, they’re good candidates for detailed analysis. Researchers have been able to document that, after conditioning, the Aplysia’s sensory neurons—the neurons that receive the CS—literally release more neurotransmitter into the synapse than they did before the conditioning trials. This is a crucial part of why, at the end of learning, these neurons are able to trigger a response—the CR—that they couldn’t trigger at the start. This process, which can be documented in many species in addition to Aplysia, produces an increase in the neural signal being sent, and is called presynaptic facilitation (e.g., Lisman, 2003; Pittenger & Kandel, 2003).
Other forms of neural plasticity,
in other organisms, involve postsynaptic changes— that is, they influence the
receiving side of the synapse. A particularly important mech-anism in this
category is long-term potentiation (LTP;
T. Bliss & Lomo, 1973; T. Bliss, Collingridge, & Laroche, 2006;
Martinez & Derrick, 1996)—potentiation
because the mechanism involves an increase in the responsiveness of a neuron
(an increase in the neuron’s potential for firing) and long term because this potentiation lasts for days, per-haps even
weeks.
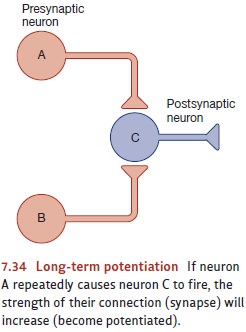
LTP is produced when one neuron
activates another neuron over and over. The repeated stimulation causes the
postsynaptic neuron to become more sensitive to this input than it was before,
so it’s more likely to respond to this input in the future. In addi-tion, the
increased responsiveness can spread to other nearby neurons. Here’s an
exam-ple: Let’s suppose that within some brief period of time neuron A in
Figure 7.34 repeatedly causes neuron C to fire. This will cause neuron C to become
more responsive to A than it was initially. But, of course, C also has other
synapses that receive input from other neurons, such as neuron B in the figure.
These receptors too will become more sen-sitive as a result of neuron A’s
repeated activity, provided that these other neurons fire at the same time as
neuron A. In other words, the spread of potentiation is activity depen-dent and so will spread to neuron B only if B was
active at the same time as the neuron thatcaused the potentiation in the first
place—in this case, neuron A (W. Levy & Steward, 1979; McNaughton, Douglas,
& Goddard, 1978). In this way, LTP provides a cellular mechanism through
which associations—in our example, the association between A’s and B’s
activity—can be detected and recorded in the brain (Fanselow & Poulos,
2005; E. Kandel & Hawkins, 1992; Koekoek et al., 2003; Martinez &
Derrick, 1996).
Presynaptic facilitation and LTP
both involve changes in how efficiently a synapse functions—the first involves
changes in how neurons send signals, and the second involves changes in how
neurons receive signals. A third form of neural plasticity involves the
creation of entirely new synapses. These changes seem to take place largely on
the dendrites of the postsynaptic neurons; the den-drites can grow new dendritic spines (Moser, 1999; Woolf,
1998). These spines are the “receiving stations” for most synapses; so growing
more spines means that, as learning proceeds, the neuron is gaining new lines
of communication with its cellular neighbors.
Related Topics