Chapter: Psychology: Learning
Instrumental Conditioning: The Major Phenomena of Instrumental Conditioning
The Major
Phenomena of Instrumental Conditioning
As Skinner noted, classical and
instrumental conditioning are different in impor-tant ways: Classical
conditioning builds on a response (UR) that’s automatically triggered by a
stimulus (US); instrumental conditioning involves behaviors that appear to be
voluntary. Classical conditioning involves learning about the relation between
two stimuli (US and CS); instrumental conditioning involves learning about the
relation between a response and a stimulus (the operant and a reward). Even
with these differences, modern theorists have argued that the two forms of
conditioning have a lot in common. This makes sense because both involve
learning about relationships among
simple events (stimuli or responses).
It’s perhaps inevitable, then, that many of the central phenomena of instrumental learning parallel those of classical conditioning. For example, in classical conditioning, learning trials typically involve the presentation of a CS followed by a US. In instrumental conditioning, learning trials typically involve a response by the organ-ism followed by a reward or reinforcer. The reinforcement often involves the presenta-tion of something good, such as grain to a hungry pigeon. Alternatively, reinforcement may involve the termination or prevention of something bad, such as the cessation of a loud noise.
In both forms of conditioning,
the more such pairings there are, the stronger the learning. And if we discontinue
these pairings so that the CS is no longer followed by the US or the response
by a reinforcer, the result is extinction.
GENERALIZATION AND DISCRIMINATION
An instrumental response is not
directly triggered by an external stimulus, the way a CR or UR is. But that
doesn’t mean external stimuli have no role here. In instrumental con-ditioning,
external events serve as discriminative
stimuli, signaling for an animal what sorts of behaviors will be rewarded
in a given situation. For example, suppose a pigeon is trained to hop onto a
platform to get some grain. When a green light is on, hopping on the platform
pays off. But when a red light is on, hopping gains no reward. Under these
circumstances, the green light becomes a positive discriminative stimulus and
the red light a negative one (usually labeled S+ and S–,
respectively). The pigeon swiftly learns this pattern and so will hop in the
presence of the first and not in the presence of the second.
Other examples are easy to find.
A child learns that pinching her sister leads to pun-ishment when her parents
are on the scene but may have no consequences otherwise. In this situation, the
child may learn to behave well in the presence of the S+ (i.e., when
her parents are there) but not in other circumstances. A hypochondriac may
learn that loud groans will garner sympathy and support from others but may
bring no benefits when others are not around. As a result, he may learn to
groan in social settings but not when alone.
Let’s be clear, though, about the
comparison between these stimuli and the stimuli central to classical
conditioning. A CS+ tells the animal about events in the world: “No
matter what you do, the US is coming.” The S+, on the other hand,
tells the animal about the impact of its own behavior: “If you respond now,
you’ll get rewarded.” The CS– indicates that no matter what the
animal does, no US is coming. The S–, in con-trast, tells the animal
something about its behavior—namely, that there’s no point in responding right
now.
Despite these differences,
generalization in instrumental conditioning functions much the way it does in
classical conditioning, and likewise for discrimination. One illustration of
these parallels lies in the generalization gradient. We saw earlier that if an
organism is trained with one CS (perhaps a high tone) but then tested with a
differ-ent one (a low tone), the CR will be diminished. The greater the change
in the CS, the greater the drop in the CR’s strength. The same pattern emerges
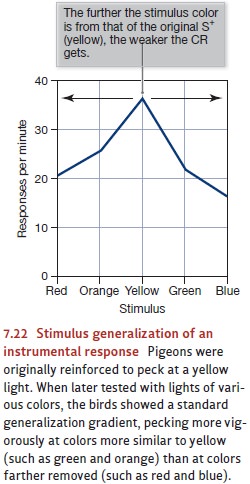
in instrumental condi-tioning. In one experiment, pigeons were trained to peck
at a key illuminated with yellow light. Later, they were tested with lights of
varying wavelengths, and the results showed an orderly generalization gradient
(Figure 7.22). As the test light became less similar to the original S+,
the pigeons were less inclined to peck at it (Guttmann & Kalish, 1956).
The ability to distinguish an S–
from an S+ obviously allows an organism to tune its behavior to its
circumstances. Thus, the dolphins at the aquarium leap out of the water to get
a treat when their feeders are around; they don’t leap up in the presence of
other people. Your pet dog sits and begs when it sees you eating, in hopes that
you’ll share the snack; but the dog doesn’t beg when it sees you drinking. In
these and other cases, behaviors are emitted only when the available stimuli
indicate that the behavior will now be rewarded.
In fact, animals are quite
skilled at making discriminations and can use impressively complex stimuli as a
basis for controlling their behavior. In one study, pigeons were trained to
peck a key whenever a picture of water was in view, but not to peck otherwise.
Some of the water pictures showed
flowing streams; some showed calm lakes. Some pictures showed large bodies of
water photographed from far away; some showed small puddles photographed close
up. Despite these variations, pigeons mastered this discrim-ination task and
were even able to respond appropriately to new pictures that hadn’t been
included in the training trials (Herrnstein, Loveland, & Cable, 1976).
Apparently, pigeons are capable of discriminating relatively abstract
categories—categories not defined in terms of a few simple perceptual features.
Similar procedures have shown that pigeons can discriminate between pictures
showing trees and pictures not showing trees; thus, for example, they’ll learn
to peck in response to a picture of a leaf-covered tree or a tree bare of
leaves, but not to peck in response to a picture of a telephone pole or a
picture of a celery stalk. Likewise, pigeons can learn to peck whenever they’re
shown a picture of a particular human—whether she’s photographed from one angle
and close up or from a very different angle, far away, and wearing different
clothes (Herrnstein, 1979; Lea & Ryan, 1990; for other examples of complex
discriminations, see Cook, K. Cavoto, & B. Cavoto, 1995; Giurfa, Zhang,
Jenett, Menzel, & Srinivasan, 2001; Lazareva, Freiburger, & Wasserman,
2004; D. Premack, 1976, 1978; A. Premack & D. Premack, 1983; Reiss &
Marino, 2001; Wasserman, Hugart, & Kirkpatrick-Steger, 1995; Zentall,
2000).
SHAPING
Once a response has been made,
reinforcement will strengthen it. Once the dolphin has leapt out of the water,
the trainers can reward it, encouraging further leaps. Once the pigeon pecks
the key, food can be delivered, making the next peck more likely. But what
causes the animal to perform the desired response in the first place? What
leads to the first leap or the first peck? This is no problem for many
responses. Dolphins occa-sionally leap with no encouragement from a trainer,
and pecking is something pigeons do all the time. If the trainer is patient,
therefore, an opportunity to reinforce (and thus encourage) these responses
will eventually arrive.
But what about less obvious
responses? For example, rats quite commonly manipu-late objects in their
environment, and so they’re likely to press on a lever if we put one within
reach. But what if we place a lever so high that the rat has to stretch up on its
hind legs to reach it? Now the rat might never press the lever on its own.
Still, it can learn this response if its behavior is suitably shaped. This shaping is accomplished by a little
“coaching,” using the method of successive
approximations.
How could we train a rat to press
the elevated lever? At first, we reinforce the ani-mal merely for walking into
the general area where the lever is located. As soon as the rat is there, we
deliver food. After a few such trials, the rat will have learned to remain in
this vicinity most of the time, so we can now increase our demand. When the rat
is in this neighborhood, sometimes it’s facing one way, sometimes another; but
from this point on, we reinforce the rat only if it’s in the area and facing
the lever. The rat soon masters this response too; now it’s facing in the right
direction most of the time. Again, therefore, we increase our demand: Sometimes
the rat is facing the lever with its nose to the ground; sometimes it’s facing
the lever with its head elevated. We now reinforce the animal only when its
head is elevated—and soon this, too, is a well-established response. We
continue in this way, counting on the fact that at each step, the rat naturally
varies its behavior somewhat, allowing us to reinforce just those variations we
prefer. Thus, we can gradually move toward reinforcing the rat only when it
stretches up to the lever, then when it actually touches the lever, and so on.
Step by step, we guide the rat toward the desired response.

Using this technique, people have
trained animals to perform all kinds of complex behavior; many human behaviors
probably come about in the same way (Figure 7.23). For example, how do parents
in Western countries teach their children to eat with a spoon and fork? At
first, they reward the child (probably with smiles and praise) just for holding
the spoon. This step soon establishes a grasp-the-spoon operant; and, at that
point, the parents can require a bit more. Now they praise the child just when
she touches food with the spoon—and, thanks to this reinforcement, the new
operant is quickly established. If the parents continue in this way, gradually
increasing their expectations, the child will soon be eating in the “proper”
way.
Similar techniques are used in
therapeutic settings to shape the behavior of the hos-pitalized mentally ill.
Initially, the hospitalized patients might be rewarded just for get-ting out of
bed. Once that behavior is established, the requirement is increased so that,
perhaps, the patients have to move around a bit in their room. Then, later, the
patients are rewarded for leaving the room and going to breakfast or getting
their medicine. In this way, the behavior therapist can gradually lead the
patients into a more acceptable level of functioning .
WHAT IS A REINFORCER ?
We’ve now said a great deal about
what reinforcement does; it encourages some responses, discourages others, and
even—through the process of shaping—creates entirely new responses. But what is
it that makes a stimulus serve as a reinforcer?
Some stimuli serve as reinforcers
because of their biological significance. These primary reinforcers include food, water, escape from the scent of a
predator, and so on—all stimuli with obvious importance for survival. Other
reinforcers are social—think of the smiles and praise from parents that we
mentioned in our example of teaching a child to use a spoon and fork.
Other stimuli are initially
neutral in their value but come to act as reinforcers because, in the animal’s
experience, they’ve been repeatedly paired with some other, already established
reinforcer. This kind of stimulus is called a conditioned reinforcer, and it works just like any other
reinforcer. A plausible example is money—a
reward that takes its value from its association with other more basic
reinforcers.
Other reinforcers, however, fall
into none of these categories, so we have to broaden our notion of what a
reinforcer is. Pigeons, for example, will peck in order to gain infor-mation about the availability of
food (e.g., G. Bower, McLean, & Meachem, 1966;Hendry, 1969). Monkeys will
work merely to open a small window through which they can see a moving toy
train (Butler, 1954). Rats will press a lever to gain access to an exer-cise
wheel (D. Premack, 1965; but also Timberlake & Allison, 1974; Timberlake,
1995). And these are just a few examples of reinforcers.
But examples like these make it
difficult to say just what a “reinforcement” is, and in practice the stimuli we
call reinforcements are generally
identified only after the fact. Is a glimpse of a toy train reinforcing? We can
find out only by seeing whether an animal will work to obtain this glimpse.
Remarkably, no other, more informative definition of a reinforcer is available.
BEHAVIORALCONTRASTADINTRINSICMOTIVATION
Once we’ve identified a stimulus
as a reinforcer, what determines how effective the reinforcer will be? We know
that some reinforcers are more powerful than others—and so an animal will
respond more strongly for a large reward than for a small one. However, what
counts as large or small depends on the context. If a rat is used to get-ting
60 food pellets for a response, then 16 pellets will seem measly and the animal
will respond only weakly for this puny reward. But if a rat is used to getting
only 4 pellets for a response, then 16 pellets will seem like a feast and the
rat’s response will be fast and strong (for the classic demonstration of this
point, see Crespi, 1942). Thus, the effectiveness of a reinforcer depends largely
on what other rewards are available (or have recently been available); this
effect is known as behavioral contrast.
Contrast effects are important
for their own sake, but they may also help explain another (somewhat
controversial) group of findings. In one study, for example, nursery-school
children were given an opportunity to draw pictures. The children seemed to
enjoy this activity and produced a steady stream of drawings. The experimenters
then changed the situation: They introduced an additional reward so that the
children now earned an attractive “Good Player” certificate for producing their
pictures. Then, later on, the chil-dren were again given the opportunity to
draw pictures—but this time with no provision for “Good Player” rewards.
Remarkably, these children showed considerably less interest in drawing than
they had at the start and chose instead to spend their time on other
activ-ities (see, for example, Lepper, Greene, & Nisbett, 1973; also Kohn,
1993).
Some theorists say these data
illustrate the power of behavioral contrast. At the start of the study, the
activity of drawing was presumably maintained by certain reinforce-ments in the
situation—perhaps encouragement from the teachers or comments by other
children. Whatever the reinforcements were, they were strong enough to maintain
the behavior; we know this because the children were producing drawings at a
steady pace. Later on, though, an additional reinforcement (the “Good Player”
certifi-cate) was added and then removed. At that point the children were back
to the same rewards they’d been getting at the start, but now these rewards
seemed puny in com-parison to the greater prize they’d been earning during the
time when the “Good Player” award was available. As a consequence, the initial
set of rewards was no longer enough to motivate continued drawing.
Other theorists interpret these
findings differently. In their view, results like this one suggest that there
are actually two different types of reward. One type is merely tacked onto a
behavior and is under the experimenter’s control; it’s the sort of reward
that’s in play when we give a pigeon a bit of food for pecking a key, or hand a
factory worker a paycheck for completing a day’s work. The other type of reward
is intrinsic to the behav-ior and independent of the experimenter’s intentions;
these rewards are in play when someone is engaging in an activity just for the
pleasure of the activity itself.
In addition, these two forms of
reward can interfere with each other. Thus, in the study with the “Good Player”
certificates, the children were initially drawing pictures for an intrinsic
reward. Drawing, in other words, was a form of play engaged in for its own sake. However, once the external
rewards (the certificates) entered the situation, the same activity became a
form of work—something you do for a
payoff. And once the
activity was redefined in this
way, then the absence of a payoff meant there was no longer any point in
drawing.
Debate continues about which of
these interpretations is preferable—the one based on behavioral contrast or the
one based on intrinsic motivation. (It also seems plausi-ble that both interpretations may capture aspects
of what’s going on here.) Clearly, there’s more to be learned about
reinforcement and the nature of motivation. (For further exploration, see
Bowles, 2008; Deci, Koestner, & Ryan, 1999a, 1999b; Eisenberger, Pierce,
& Cameron, 1999; Henderlong & Lepper, 2002.)
SCHEDULES OF REIN FOR CEMENT
Let’s now return to the issue of
how extrinsic reinforcements work, since—by any-one’s account—these
reinforcements play a huge role in governing human (and other species’)
behavior. We do, after all, work for money, buy lottery tickets in hopes of
winning, and act in a way that we believe will bring us praise. But notice that
in all of these examples, the reinforcement comes only occasionally: We aren’t
paid after every task we do at work; we almost never win the lottery; and we
don’t always get the praise we seek. Yet we show a surprising resistance t0
extinction of those behaviors. About some things, we have learned that if you
don’t succeed, it pays to try again— an important strategy for achieving much
of what we earn in life.
This pattern, in which we’re
reinforced for only some of our behaviors, is known as partial reinforcement. In the laboratory, partial reinforcement can
be provided according to different schedules
of reinforcement—rules about how often and under what conditions a response
will be reinforced. Some behaviors are reinforced via a ratio schedule, in which you’re rewarded for producing a certain
number of responses (Figure 7.24). The ratio can be “fixed” or “variable.” In a
“fixed-ratio 2” (FR 2) schedule, for example, two responses are required for
each reinforcement; for an “FR 5” schedule, five responses are required. In a
variable-ratio schedule, the number of responses required changes from trial to
trial. Thus, in a “VR 10” sched-ule, 10 responses are required on average to get a reward—so it might
be that the first 5 responses are enough to earn one reward, but 15 more are
needed to earn the next.
Other behaviors are reinforced on
an interval schedule, in which
you’re rewarded for producing a response after a certain amount of time has
passed (Figure 7.25). Thus, on an “FI 3-minute” schedule, responses made during
the 3-minute interval aren’t rein-forced; but the first response after the 3
minutes have passed will earn a reward. Interval schedules can also be
variable: For a “VI 8-minute” schedule, reinforcement is available on average
after 8 minutes; but the exact interval required varies from trial to trial.


Related Topics