Chapter: Mechanical : Engineering Thermodynamics : Basic Concepts And Definitions
Temperature Scales
Temperature Scales
![]()
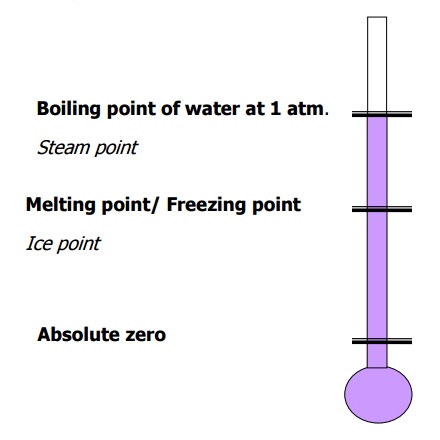
Freezing point of water known as ice
point and boiling point of water known as steam point are
taken as the reference states for all types of temperature scales.
The
various types as temperature scales in use are :
![]()
a)
Celsius scale
b)
Fahrenheit scale
c)
Kelvin scale
d)
Rankine scale
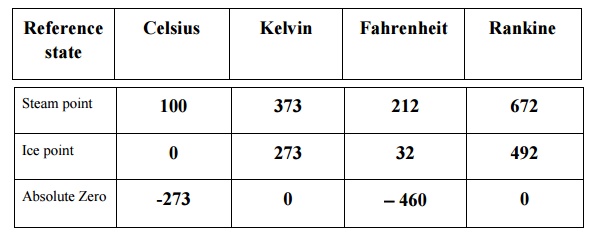
Reference
state Celsius Kelvin Fahrenheit Rankine
Steam point 100 373 212 672
Ice point 0 273 32 492
Absolute Zero -273 0 -460 0
Homogeneous
and Heterogeneous Systems
Matter can exist in any one of the three
phases namely solid, liquid and gas. A system consisting of a single phase is
known as homogeneous systems. If the matter exists in more than one phase, the
system is known as heterogeneous system.
BASIC CONCEPTS AND DEFINITIONS
Thermodynamics is the
science of energy transfer which deals with the relations among heat, work and
properties of systems.
The name ‘thermodynamics’ is derived from the
Greek words therme, meaning ‘heat’ and dynamis meaning
power. Thus, thermodynamics is basically the study of heat and power.
Application Area of Thermodynamics
Energy transfer is
present in almost all the engineering activities. Hence, the principles of
thermodynamics are playing vital role in designing all the engineering
equipments such as internal combustion engines, rockets, jet engines, thermal
and nuclear power plants, refrigerators etc.
Related Topics