Chapter: Mechanical : Engineering Thermodynamics : Basic Concepts And Definitions
Pure Substances and Concept of continuum
Pure Substances
Substances
of fixed chemical composition throughout are known as pure substances.
That is, pure substances have homogenous and
invariable chemical composition irrespective of the phase or phases in which
they exist.
Example
a.Atmosphere
air
b.
Water
c. Nitrogen
d.
Water-steam mixture
e. Product
of combustion.
Though, mixture of water and steam is
considered a pure substance, air and liquid air cannot be, since, the chemical
composition of liquid air differs from that of gaseous air.
The
Ideal Gas
Based on the experimental work carried
out by Boyle, Charles and Gay-Lussac, pressure, temperature and specific volume
of many gases at low pressure and moderate temperature are related by the
following equation.
pv
= RT where R=
This
equation is known as equation of state of an ideal gas. The term R is known as
characteristic gas constant and Ru universal gas constant. In SI
unit Ru= 8.314 kJ/kgmol.K.
Concept of continuum
In microscopic approach the substance is assumed to be continuously distributed, ignoring the space between the molecules. This is known as continuum hypothesis.
Since the matter is treated as continuous, the density at a point can be defined as
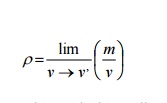
Where v’is the smallest volume for which a definite value of the ratio exists. Below the limiting value of v’, the fluctuation in average density will be high and a definite value for the ratio becomes impossible, with the mean free path* of the molecules approaching the order of magnitude of the dimension of the vessel
BASIC CONCEPTS AND DEFINITIONS
Thermodynamics is the
science of energy transfer which deals with the relations among heat, work and
properties of systems.
The name ‘thermodynamics’ is derived from the
Greek words therme, meaning ‘heat’ and dynamis meaning
power. Thus, thermodynamics is basically the study of heat and power.
Application Area of Thermodynamics
Energy transfer is
present in almost all the engineering activities. Hence, the principles of
thermodynamics are playing vital role in designing all the engineering
equipments such as internal combustion engines, rockets, jet engines, thermal
and nuclear power plants, refrigerators etc.
Related Topics