Chapter: Mechanical : Engineering Thermodynamics : Basic Concepts And Definitions
Point and Path Functions
Point and Path
Functions
Thermodynamic functions
are classified into two categories namely point and path functions. Point
functions are those for which the change depends on only the end states and not
on the path followed. Hence point functions are inexact differentials
Path functions are
those for which the change depends not only on the end states but also on the
path followed. Hence path functions are exact differentials
In can be observed the
change in any property during a process depends only on end states. Therefore
all the properties are point functions.
.
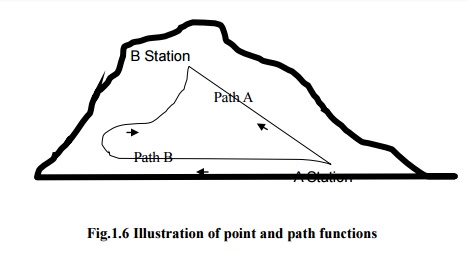
To
demonstrate path and point functions, let us consider two stations A and B on a
hill as shown in the Fig.1.6. While moving from station A to station B, let the
distance traveled and increase in height from the mean sea level are observed.
Distance traveled in path 1 is different from that in path 2. Hence it may be
regarded as path function. But the change in height is same in both path 1 and
path 2, therefore it is a point function.
State
Postulate and Property Diagrams
As
mentioned earlier, properties are meant for describing the state of a system.
To fix a state, all the properties need not be specified. If any two
independent intensive properties are specified, rest of the properties
automatically assumes certain values. This is known as state postulate.
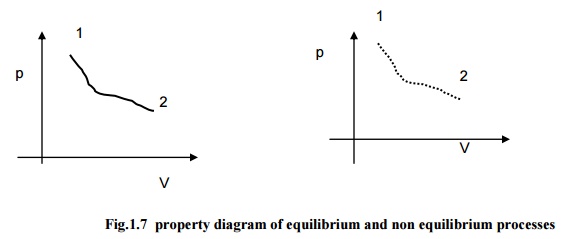
Fig.1.7
property diagram of equilibrium and non equilibrium processes
Consider
pressure and specific volume (Volume per unit mass) are the two
independent, intensive properties, describing the state of a compressible
system. On a p-V diagram the state will assume a point as represented in the
Fig.1.7(a). Let the system be taken to another state such that all the
intermediate states are equilibrium states. The curve connecting the initial
state and final state, passing through all the intermediate states is
indicating the path of the process. In non-quasi-equilibrium process as the
intermediate status can not be defined, the path is denoted by dashed line as
given in Fig.1.7(b)
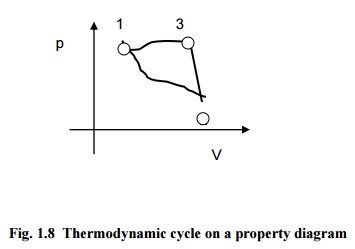
Fig.
1.8 Thermodynamic cycle on a property diagram
Fig.1.8 indicates a system undergoing a cycle
consisting of three quasi-equilibrium processes.
Related Topics