Chapter: Human Nervous System and Sensory Organs : Telencephalon
Telencephalon
Telencephalon
Overview
Subdivision of the Hemisphere (A, B)
The embryonic hemispheric vesicle (A) clearly shows the subdivision of the telen-cephalon into four parts, some of which develop early (phylogenetically old por-tions), while others develop late (phylo-genetically new portions). The four parts are the paleopallium, the striatum, theneopal-lium, and the archipallium.
The hemispheric wall is called the pallium, or brain mantle, because it covers the dien-cephalon and brain stem and envelops them like a mantle.
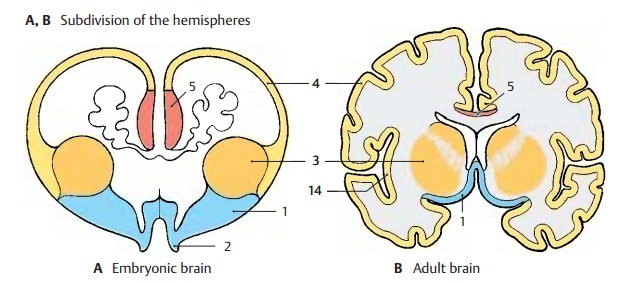
The paleopallium (blue) (AB1) is the oldest portion of the hemisphere. It forms the floor of the hemisphere and corresponds, with the olfactory bulb(A2) and adjacent paleo-cortex, to the olfactory brain, orrhinencephalon, in the narrower sense. Theneostriatum (deep yellow) (AB3) develops above the paleopallium; it, too, is part of the hemispheric wall, although it does not appear on the outer aspect of the hemisphere.
The largest area is made up by the neopal-lium (light yellow). Its outer aspect, the neo-cortex (AB4), develops very lateand encircles ventrally a transitional area to the paleocortex that lies over the striatum; this is the insula (B14).
The medial hemispheric wall is formed by the archipallium (red) (AB5), an old por-tion of the brain; its cortical band, the archi-cortex, later curls up to form thehippocampus (Ammon’s horn).
The relationships in the mature brain are determined by the massive expansion of the neocortex, which pushes the paleocortex and the transitional cortex of the insula into the deeper parts of the brain. The archicor-tex becomes displaced caudally and appears on the surface of the corpus callosum only as a thin layer (B5, F10).
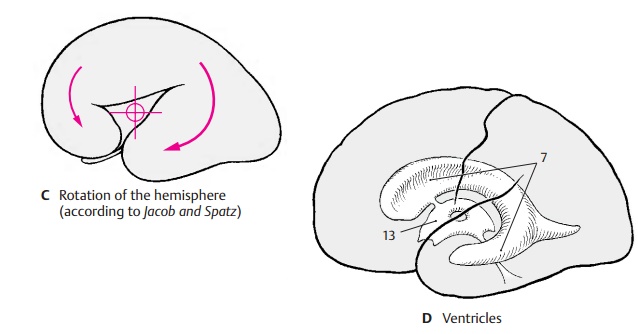
Rotation of the Hemisphere (C – F)
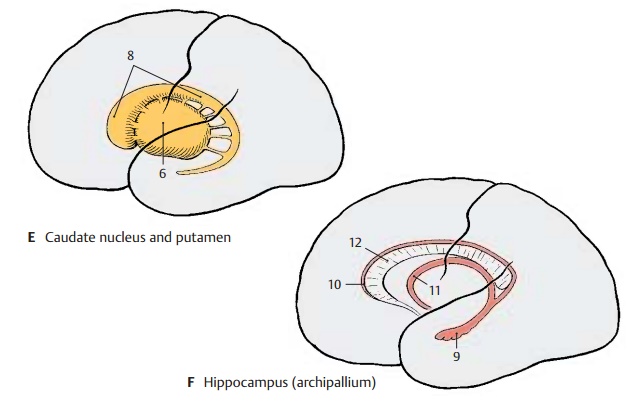
The hemispheric vesicle does not expand evenly in all directions during its develop-ment but widens primarily in caudal and basal directions. The temporal lobe is formed in this way, and it finally turns ros-trally in a circular movement (C); to a lesser degree, such a rotation can also be observed with the frontal lobe. The axis around which the hemispheric vesicle rotates is the insu-lar region; like the putamen (E6) lying beneath it, the insula does not participate in the movement. Other structures of the hemisphere, however, follow the rotation and end up having an arched shape in the mature brain. The lateral ventricle (D7) forms such an arch with its anterior and in-ferior horns. The lateral portion of the stri-atum, the caudate nucleus (E8), participates in the rotation as well and follows precisely the arched shape of the lateral ventricle. The main part of the archipallium, thehippo-campus (F9), moves from its original dorsalposition in basal direction and comes to lie in the temporal lobe. The remnants of the archipallium on the dorsal aspect of the cor-pus callosum, the indusium griseum (F10), and the fornix (F11) reflect the arched ex-pansion of the archipallium. Thecorpus cal-losum (F12) also expands in caudal directionbut follows the rotation only partially as it develops only late toward the end of this process.
D13 Third ventricle.
Evolution (A – D)
During primate evolution, the telen-cephalon has undergone changes similar to those taking place during human embry-onic development; it developed late and then overgrew the other parts of the brain. Thus, the cerebellum (A1) is still completely exposed in the brain of primitive mammals (hedgehog), while it becomes more and more covered by the hemispheres of the tel-encephalon during primate evolution.
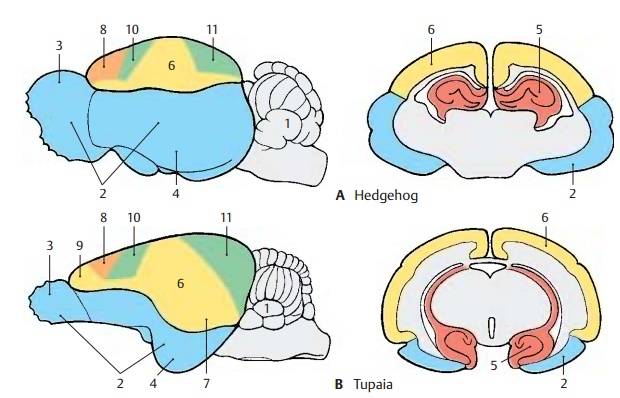
The paleopallium (rhinencephalon) (blue) (A – C2) with olfactory bulb (A – C3) and pir-iform lobe (A–C4) forms the largest part ofthe hemisphere in the primitive mam-malian brain (A), and the archipallium (red) (A – D5) still has its original dorsal position above the diencephalon. These two old components of the hemisphere then be-come overgrown by the neopallium (yellow) (A – D6) during the course of evolution. The paleopallium of prosimians (C) is still of considerable size. In humans (D), however, it becomes displaced deep into the base of the brain and no longer appears in the lateral view of the brain. The archipallium (hippocampus), which lies above the dien-cephalon in the hedgehog (A5), appears as a part of the temporal lobe at the base of the brain in humans (D5). Only a narrow rem-nant remains above the corpus callosum (indusium griseum).
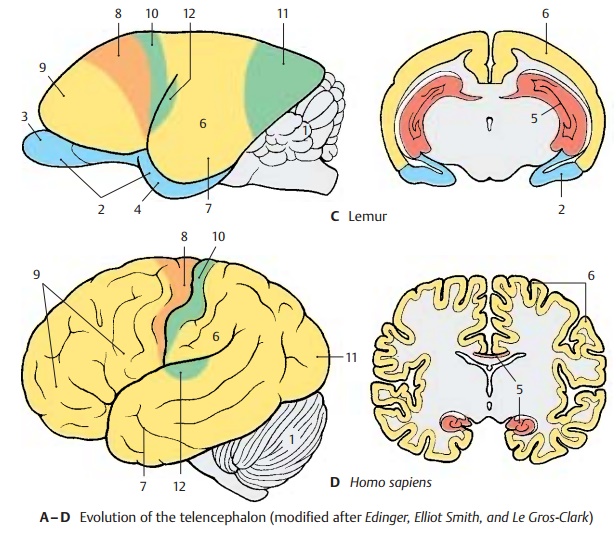
The positional changes largely correspond to the rotation of the hemisphere during embryonic development; they also lead to the formation of the temporal lobe (B – D7). While still absent from the brain of the hedgehog (A), the temporal lobe is already recognized as a ventrally directed projec-tion in the brain of the tree shrew (Tupaia), the most primitive of primates (B). In the prosimian brain (C), a caudally directed temporal lobe has developed that finally turns rostrally in the human brain (D). In ad-dition, sulci and gyri develop in the region of the neopallium. Whereas the neopallium of primitive mammals is smooth (lissen-cephalic brains), a relief of convolutionsdevelops only in higher mammals (gyren-cephalic brains). The development of sulciand gyri considerably enlarges the surface of the cerebral cortex. In humans, only one-third of the cortical surface lies at the sur-face of the hemispheres, two-thirds lie deep in the sulci.
Two types of cortical areas can be distin-guished on the neocortex: the primary areasof origin (light red) and termination areas (green) of long pathways, and between them the secondary association areas (yellow).
The area of origin of motor pathways, the motor cortex (A–D8), constitutes the entirefrontal lobe in the hedgehog. An association area (B – D9) appears for the first time in primitive primates (Tupaia) and achieves extraordinary expansion in the human brain. The termination area of sensory path-ways, the sensory cortex (A – D10), borders caudally on the motor cortex. Owing to the enlargement of the adjacent association area, most of the termination area of the visual pathway, the visual cortex (A – D11), becomes displaced to the medial hemispheric surface in humans. The termi-nation area of the acoustic pathway, the auditory cortex (CD12), becomes displaceddeep into the lateral sulcus (fissure of Syl-vius) by the expansion of the temporal asso-ciation areas. Thus, the association areas ex-pand much more during evolution than the primary areas; they represent the largest part of the neocortex in humans.
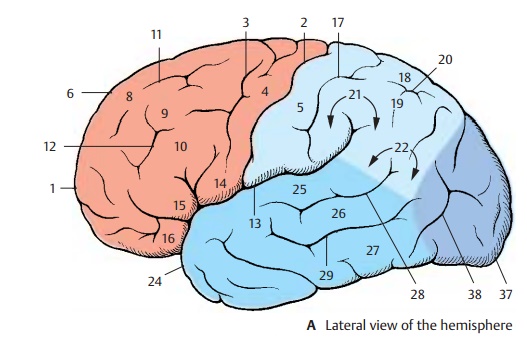
Cerebral Lobes (A – C)
The hemisphere is divided into four cere-bral lobes:
The frontal lobe (red)
! The parietal lobe (light blue)
! The temporal lobe (dark blue)
! The occipital lobe (purple)
The hemispheric surface consists of grooves, or sulci, and convolutions, or gyri. We distin-guish primary, secondary, and tertiary sulci. The primary sulci appear first and are equally well developed in all human brains (central sulcus, calcarine sulcus). The sec-ondary sulci are variable. The tertiary sulci appear last, being irregular and different in each brain. Thus, each brain has its own sur-face relief as an expression of individuality, like the features of the face.
The frontal lobe extends from the frontalpole (AC1) to thecentral sulcus(AB2), whichtogether with the precentral sulcus (A3) de-fines the precentral gyrus (A4). The latter is grouped with the postcentral gyrus (A5) to form the central region, which spreads be-yond the edge of the hemisphere (AB6) to the paracentral gyrus (B7). Furthermore, thefrontal lobe exhibits three major convolu-tions: the superior frontal gyrus (A8), the middle frontal gyrus (A9), and theinferior frontal gyrus (A10); they are separated bythe superior frontal sulcus (A11) and the in-ferior frontal sulcus (A12). Three parts aredistinguished at the inferior frontal gyrus that define the lateral sulcus (sulcus of Syl-vius) (AC13): the opercular part (A14), the triangular part (A15), and the orbital part (A16).
The parietal lobe adjoins the frontal lobe with the postcentral gyrus (A5) which is de-fined caudally by the postcentral sulcus (A17). This is followed by the superiorparietal lobule (A18) and the inferior parietal lobule (A19), which are separated by the in-traparietal sulcus (A20). The end of thelateral sulcus is surrounded by the supra-marginal gyrus (A21); theangular gyrus (A22) lies ventrally to it. The medial surface of the parietal lobe is formed by the pre-cuneus (B23).
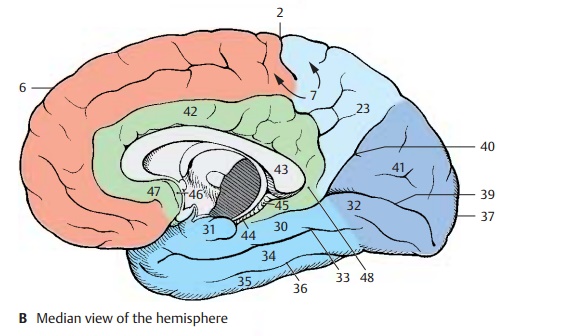
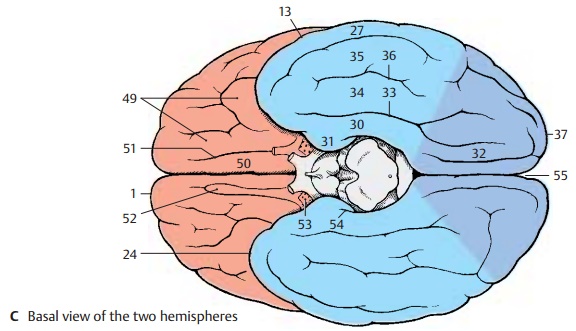
The temporal lobe includes the temporalpole (AC24) and three major convolutions:the superior temporal gyrus (A25), the middle temporal gyrus(A26), and the inferior temporal gyrus (AC27), which are separatedby the superior temporal sulcus (A28) and the inferior temporal sulcus (A29). Thetrans-verse temporal gyri (Heschl ’s convolutions)of the dorsal aspect of the temporal lobe lie in the depth of the lateral sulcus. On the medial surface is the parahippocam-pal gyrus (BC30) which merges rostrally intothe uncus (BC31) and caudally into the lin-gual gyrus (BC32). It is separated by thecol-lateral sulcus (BC33) from the middle occipi-totemporal gyrus (BC34). Ventrally lies the lateral occipitotemporal gyrus (BC35),delimited by theoccipitotemporal sulcus (BC36).
The occipital lobe includes the occipitalpole (A–C37) and is crossed by the trans-verse occipital sulcus (A38) and the deepcal-carine sulcus (B39). Together with theparieto-occipital sulcus (B40), the latter de-fines the cuneus (B41).
The cingulate gyrus (limbic gyrus) (green) (B42) extends around the corpus callosum (B43). Caudally, it is separated by the hippo-campal sulcus(B44) from the dentate gyrus (dentate band) (B45) and tapers rostrally into the paraterminal gyrus (B46) and into the subcallosal area (parolfactory area) (B47). Isthmus of cingulate gyrus (B48).
Base of the brain.The basal aspect of thefrontal lobe is covered by the orbital gyri (C49). Along the edge of the hemisphere runs the gyrus rectus(C50), laterally defined by the olfactory sulcus (C51) into which the olfactory bulb (C52) and the olfactory tract are embedded. The olfactory tract splits into the two olfactory striae which embrace the anterior perforated substance (olfactoryarea) (C53).
C54 Hippocampal sulcus.
C55 Longitudinal cerebral fissure.
Related Topics