Chapter: Basic & Clinical Pharmacology : General Anesthetics
Propofol - Intravenous Anesthetics
PROPOFOL
In
most countries, propofol is the most frequently administered drug for induction
of anesthesia and has largely replaced barbitu-rates for this indication.
Because its pharmacokinetic profile allows
for continuous infusions, propofol is also used during maintenance of
anesthesia and is a common choice for sedation in the setting of monitored
anesthesia care. Increasingly, propofol is also used for sedation in the ICU as
well as conscious sedation and short-duration general anesthesia in locations
outside the operating room (eg, interventional radiology suites, emergency
department; see Topic: Sedation & Monitored Anesthesia Care, earlier).
Propofol
(2,6-diisopropylphenol) is an alkyl phenol with hyp-notic properties that is
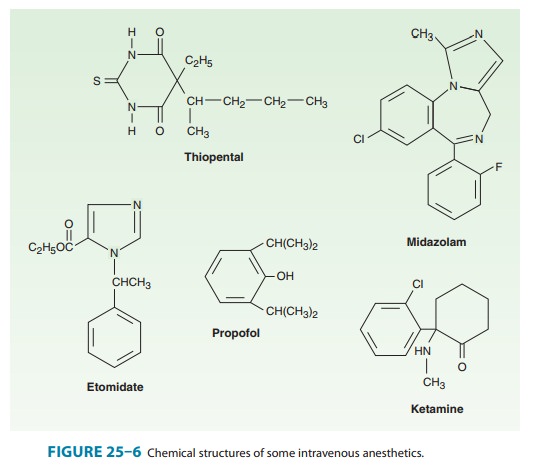
chemically distinct from other groups of intravenous anesthetics (Figure 25–6).
Because of its poor solubil-ity in water, it is formulated as an emulsion
containing 10% soy-bean oil, 2.25% glycerol, and 1.2% lecithin, the major
component of the egg yolk phosphatide fraction. Hence, susceptible patients may
experience allergic reactions. The solution appears milky white and slightly
viscous, has a pH of approximately 7, and a propofol concentration of 1% (10
mg/mL). In some countries, a 2% formu-lation is available. Although retardants
of bacterial growth are added to the formulations, solutions should be used as
soon as pos-sible (no more than 8 hours after opening the vial) and proper
sterile technique is essential. The addition of metabisulfite in one of the
formulations has raised concern regarding its use in patients with reactive
airway disease (eg, asthma) or sulfite allergies.
The
presumed mechanism of action of propofol is through potentiation of the
chloride current mediated through the GABAA receptor complex.
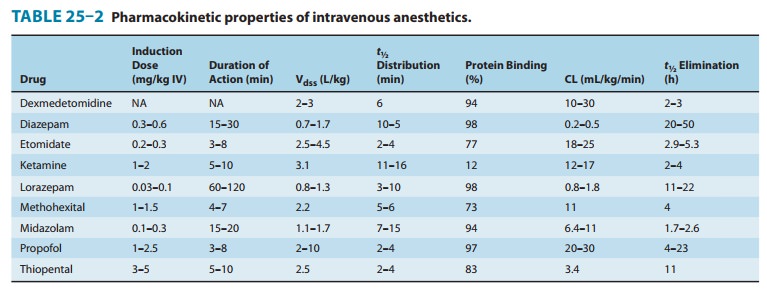
Pharmacokinetics
Propofol
is rapidly metabolized in the liver; the resulting water-soluble compounds are
presumed to be inactive and are excreted through the kidneys. Plasma clearance
is high and exceeds hepatic blood flow, indicating the importance of
extrahepatic metabolism, which is thought to occur to a significant extent in
the lungs and may account for the elimination of up to 30% of a bolus dose of
the drug (Table 25–2). The recovery from propofol is more complete,with less
“hangover” than that observed with thiopental, likely due to the high plasma
clearance. However, as with other intravenous drugs, transfer of propofol from
the plasma (central) compart-ment and the associated termination of drug effect
after a single bolus dose are mainly the result of redistribution from highly
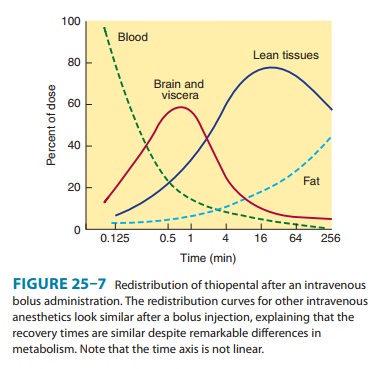
perfused (brain) to less-well-perfused (skeletal muscle) compart-ments (Figure
25–7). As with other intravenous agents, awakening after an induction dose of
propofol usually occurs within 8–10 minutes. The kinetics of propofol (and
other intravenous anes-thetics) after a single bolus dose or continuous
infusion are best described by means of a three-compartment model. Such models
have been used as the basis for developing systems of target-controlled
infusions.
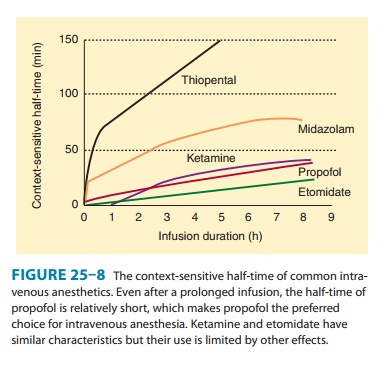
The
context-sensitive half-time of a
drug describes the elimination half-time after a continuous infusion as a
function of the duration of the infusion and is an important factor in the
suitability of a drug for use as maintenance anesthetic. The context-sensitive
half-time of propofol is brief, even after a prolonged infusion, and recovery
remains relatively prompt (Figure 25–8).
Organ System Effects
A. CNS Effects
Propofol
acts as hypnotic but does not have analgesic properties. Although the drug
leads to a general suppression of CNS activity, excitatory effects such as
twitching or spontaneous movement are occasionally observed during induction of
anesthesia. These effects may resemble seizure activity; however, most studies
sup-port an anticonvulsant effect of propofol, and the drug may be safely
administered to patients with seizure disorders. Propofol decreases cerebral
blood flow and the cerebral metabolic rate for oxygen (CMRO2), which
decreases intracranial pressure (ICP) and intraocular pressure; the magnitude
of these changes is com-parable to that of thiopental. Although propofol can
produce a desired decrease in ICP, the combination of reduced cerebral blood
flow and reduced mean arterial pressure due to peripheral vasodilation can
critically decrease cerebral perfusion pressure.
When
administered in large doses, propofol produces burst suppression in the EEG, an
end point that has been used when administering intravenous anesthetics for
neuroprotection during neurosurgical procedures. Evidence from animal studies
suggests that propofol’s neuroprotective effects during focal ischemia are
similar to those of thiopental and isoflurane.
B. Cardiovascular Effects
Compared
with other induction drugs, propofol produces the most pronounced decrease in
systemic blood pressure; this is a result of profound vasodilation in both
arterial and venous circulationsleading to reductions in preload and afterload.
This effect on sys-temic blood pressure is more pronounced with increased age,
in patients with reduced intravascular fluid volume, and with rapid injection.
Because the hypotensive effects are further augmented by the inhibition of the
normal baroreflex response, the vasodilation only leads to a small increase in
heart rate. Profound bradycardia and asystole after the administration of
propofol have been described in healthy adults despite prophylactic anticholinergic
drugs.
C. Respiratory Effects
Propofol
is a potent respiratory depressant and generally produces apnea after an
induction dose. A maintenance infusion reduces minute ventilation through
reductions in tidal volume and respi-ratory rate, with the effect on tidal
volume being more pro-nounced. In addition, the ventilatory response to hypoxia
and hypercapnia is reduced. Propofol causes a greater reduction in upper airway
reflexes than thiopental does, which makes it well suited for instrumentation of
the airway, such as placement of a laryngeal mask airway.
D. Other Effects
Although
propofol, unlike volatile anesthetics, does not augment neuromuscular block,
studies have found good intubating condi-tions after propofol induction without
the use of neuromuscular blocking agents. Unexpected tachycardia occurring
during propo-fol anesthesia should prompt laboratory evaluation for possible
metabolic acidosis (propofol infusion syndrome). An interesting and desirable
side effect of propofol is its antiemetic activity. Pain on injection is a
common complaint and can be reduced by pre-medication with an opioid or
coadministration with lidocaine. Dilution of propofol and the use of larger
veins for injection can also reduce the incidence and severity of injection
pain.
Clinical Uses & Dosage
The
most common use of propofol is to facilitate induction of general anesthesia by
bolus injection of 1–2.5 mg/kg IV. Increasing age, reduced cardiovascular
reserve, or premedication with benzo-diazepines or opioids reduces the required
induction dose; children require higher doses (2.5–3.5 mg/kg IV). Generally,
titration of the induction dose helps to prevent severe hemodynamic changes.
Propofol is often used for maintenance of anesthesia either as part of a
balanced anesthesia regimen in combination with volatile anesthetics, nitrous
oxide, sedative-hypnotics, and opioids or as part of a total intravenous
anesthetic technique, usually in combi-nation with opioids. Therapeutic plasma
concentrations for main-tenance of anesthesia normally range between 3 and 8
mcg/mL (typically requiring a continuous infusion rate between 100 and 200
mcg/kg/min) when combined with nitrous oxide or opioids.When used for sedation
of mechanically ventilated patients in the ICU or for sedation during
procedures, the required plasma concentration is 1–2 mcg/mL, which can be
achieved with a con-tinuous infusion at 25–75 mcg/kg/min. Because of its
pronounced respiratory depressant effect and narrow therapeutic range,
propo-fol should be administered only by individuals trained in airway
management. Subanesthetic doses of propofol can be used to treat postopera-tive
nausea and vomiting (10–20 mg IV as bolus or 10 mcg/kg/min as an infusion).
Related Topics