Chapter: Basic & Clinical Pharmacology : General Anesthetics
Barbiturates - Intravenous Anesthetics
BARBITURATES
This
section focuses on the use of thiopental and methohexital for induction of
general anesthesia; however, these barbiturate hypnotics have been largely
replaced as induction agents by propofol.
The
anesthetic effect of barbiturates presumably involves a combination of
enhancement of inhibitory and inhibition of excitatory neurotransmission.
Although the effects on inhibitory transmission probably result from activation
of the GABAA receptor complex, the effects on excitatory
transmission are less well understood.
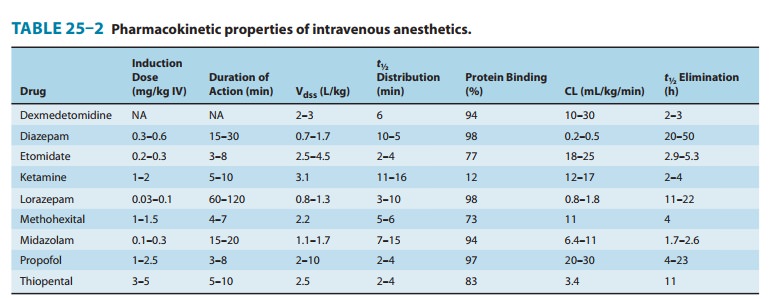
Pharmacokinetics
Thiopental
and methohexital undergo hepatic metabolism, mostly by oxidation but also by N-dealkylation, desulfuration, and
destruction of the barbituric acid ring structure. Barbiturates should not be
administered to patients with acute intermittent porphyria because they
increase the production of porphyrins through stimulation of aminolevulinic
acid synthetase. Methohexital has a shorter elimination half-time than
thiopental due to its larger plasma clearance (Table 25–2), leading to a faster
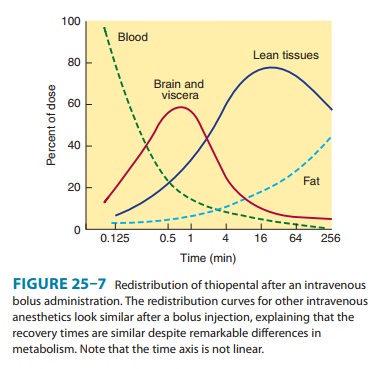
and more complete recovery after bolus injection. Although thio-pental is
metabolized more slowly and has a long elimination half-time, recovery after a
single bolus injection is comparable to that of methohexital and propofol
because it depends on redistri-bution to inactive tissue sites rather than on
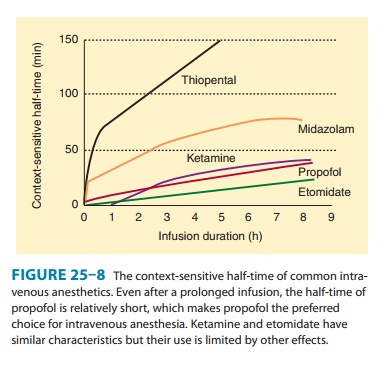
metabolism (Figure 25–7). However, if administered through repeated bolus
injec-tions or continuous infusion, recovery will be markedly prolonged because
elimination will depend on metabolism under these cir-cumstances (see also
context-sensitive half-time, Figure 25–8).
A. CNS Effects
Barbiturates
produce dose-dependent CNS depression ranging from sedation to general
anesthesia when administered as bolus injections. They do not produce
analgesia; instead, some evidence suggests they may reduce the pain threshold
causing hyperalgesia. Barbiturates are potent cerebral vasoconstrictors and
produce pre-dictable decreases in cerebral blood flow, cerebral blood volume,
and ICP. As a result, they decrease CMRO2 consumption in a
dose-dependent manner up to a dose at which they suppress all EEG activity. The
ability of barbiturates to decrease ICP and CMRO2 makes these drugs
useful in the management of patients with space-occupying intracranial lesions.
They may provide neu-roprotection from focal cerebral ischemia (stroke,
surgical retrac-tion, temporary clips during aneurysm surgery), but probably
not from global cerebral ischemia (eg, from cardiac arrest). Except for
methohexital, barbiturates decrease electrical activity on the EEG and can be
used as anticonvulsants. In contrast, methohexital activates epileptic foci and
may therefore be useful to facilitate electroconvulsive therapy or during the
identification of epileptic foci during surgery.
B. Cardiovascular Effects
The
decrease in systemic blood pressure associated with adminis-tration of
barbiturates for induction of anesthesia is primarily due to peripheral
vasodilation and is usually smaller than the blood pressure decrease associated
with propofol. There are also direct negative inotropic effects on the heart.
However, inhibition of the baroreceptor reflex is less pronounced than with
propofol; thus, compensatory increases in heart rate limit the decrease in
blood pressure and make it transient. The depressant effects on sys-temic blood
pressure are increased in patients with hypovolemia, cardiac tamponade,
cardiomyopathy, coronary artery disease, or cardiac valvular disease because
such patients are less able to compensate for the effects of peripheral
vasodilation. Hemo-dynamic effects are also more pronounced with larger doses
and rapid injection.
C. Respiratory Effects
Barbiturates
are respiratory depressants, and a usual induction dose of thiopental or
methohexital typically produces transient apnea, which will be more pronounced
if other respiratory depressants are also administered. Barbiturates lead to
decreased minute ventilation through reduced tidal volumes and respiratory rate
and also decrease the ventilatory responses to hypercapnia and hypoxia.
Resumption of spontaneous breathing after an anesthetic induction dose of a
barbiturate is characterized by a slow breathing rate and decreased tidal
volume. Suppression of laryngeal reflexes and cough reflexes is probably not as
profound as after an equianesthetic propofol administration, which makes
barbiturates an inferior choice for airway instrumentation in the absence of
neuromuscular blocking drugs. Furthermore, stimulation of the upper airway or
trachea (eg, by secretions, laryngeal mask airway, direct laryngos-copy,
tracheal intubation) during inadequate depression of airway reflexes may result
in laryngospasm or bronchospasm. This phe-nomenon is not unique to barbiturates
but is true whenever the drug dose is inadequate to suppress the airway
reflexes.
D. Other Effects
Accidental
intra-arterial injection of barbiturates results in excru-ciating pain and
intense vasoconstriction, often leading to severe tissue injury involving
gangrene. Approaches to treatment include blockade of the sympathetic nervous
system (eg, stellate ganglion block) in the involved extremity. If
extravasation occurs, some authorities recommend local injection of the area
with 0.5% lido-caine (5–10 mL) in an attempt to dilute the barbiturate
concen-tration. Life-threatening allergic reactions to barbiturates are rare,
with an estimated occurrence of 1 in 30,000 patients. However,
barbiturate-induced histamine release occasionally is seen.
Clinical Uses & Dosage
The
principal clinical uses of thiopental (3–5 mg/kg IV) or metho-hexital (1–1.5
mg/kg IV) is for induction of anesthesia (uncon-sciousness), which usually
occurs in less than 30 seconds. Patients may experience a garlic or onion taste
after administration. Barbiturates such as methohexital (20–30 mg/kg) may be
adminis-tered per rectum to facilitate induction of anesthesia in mentally
challenged and uncooperative pediatric patients. When a barbitu-rate is
administered with the goal of neuroprotection, an isoelectric EEG indicating
maximal reduction of CMRO2 has traditionally been used as the end
point. More recent data demonstrating equal protection after smaller doses have
challenged this practice. The use of smaller doses is less frequently
associated with hypotension, thus making it easier to maintain adequate
cerebral perfusion pressure, especially in the setting of increased ICP.
Related Topics