Chapter: Clinical Anesthesiology: Perioperative & Critical Care Medicine: Critical Care
Positive Airway Pressure Therapy
POSITIVE AIRWAY PRESSURE THERAPY
Positive airway pressure therapy can be used
in patients who are breathing spontaneously as well as those who are
mechanically ventilated. The princi-pal indication for positive airway pressure
therapy is a decrease in FRC resulting in absolute or relative hypoxemia. By
increasing transpulmonary distend-ing pressure, positive airway pressure
therapy can increase FRC, improve (increase) lung compliance, and reverse
ventilation/perfusion mismatching.Improvement in the latter parameter will show
as a decrease in venous admixture and an improvement in arterial O 2 tension.
Positive End-Expiratory Pressure
Application of positive pressure during expiration as an adjunct to a
mechanically delivered breath is referred to as PEEP. The ventilator’s PEEP
valve provides a pressure threshold that allows expiratory flow to occur only
when airway pressure exceeds the selected PEEP level.
Continuous Positive Airway Pressure
Application of a positive-pressure threshold dur-ing both inspiration
and expiration with sponta-neous breathing is referred to as CPAP. Constant
levels of pressure can be attained only if a high-flow (inspiratory) gas source
is provided. When the patient does not have an artificial airway, tightly
fitting full-face masks, nasal masks, nasal “pillows” (ADAM circuit), or nasal
prongs (neonatal) can be used. Because of the risks of gastric distention and
regurgitation, CPAP masks should be used only on patients with intact airway
reflexes and with CPAP levels less than 15 cm H2O (less than lower esophageal
sphincter pressure in normal persons). Expiratory pressures above 15 cm H2O require
an artificial airway.
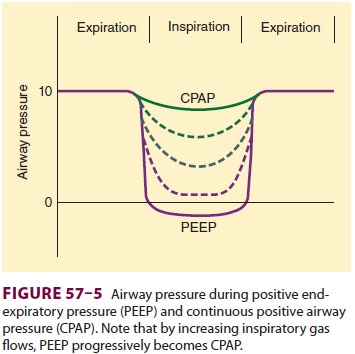
CPAP versus PEEP
The distinction between PEEP and CPAP is
often blurred in the clinical setting because patients may breathe with a
combination of mechanical and spon-taneous breaths. Therefore, the two terms
are often used interchangeably. In the strictest sense, “pure” PEEP is provided
as a ventilator-cycled breath. In contrast, a “pure” CPAP system provides only
sufficient continuous or “on-demand” gas flows (60–90 L/min) to prevent
inspiratory airway pres-sure from falling perceptibly below the expiratory
level during spontaneous breaths (Figure 57–5).
Some ventilators with demand valve–based CPAP systems may not be adequately
responsive and result in increased inspiratory work of breathing.
This situation can be corrected by adding low levels of (inspiratory)
PSV if in a volume-targeted mode or changing to a pressure-targeted mode. In
clini-cal practice, controlled ventilation, PSV, and CPAP/ PEEP support can be
delivered by most modern ICU ventilators. Manufacturers have also developed
specific devices to deliver bilevel inspiratory posi-tive airway pressure
(IPAP) with expiratory positive airway pressure (EPAP) in either a spontaneous
or time-cycled fashion. The term bilevel
positive air-way pressure (BiPAP) has become a commonly usedphrase, adding
to the confusion of airway pressure terminology.
Pulmonary Effects of PEEP & CPAP
The major effect of PEEP and CPAP on the
lungs is to increase FRC. In patients withdecreased lung volume, appropriate
levels of either PEEP or CPAP will increase FRC and tidal ven-tilation above
closing capacity, will improve lung compliance, and will correct
ventilation/perfusion abnormalities. The resulting decrease in intrapul-monary
shunting improves arterial oxygenation. The principal mechanism of action for
both PEEP and CPAP appears to be expansion of partially col-lapsed alveoli.
Recruitment (reexpansion) of col-lapsed alveoli occurs at PEEP or CPAP levels
above the inflection point, defined as the pressure level on
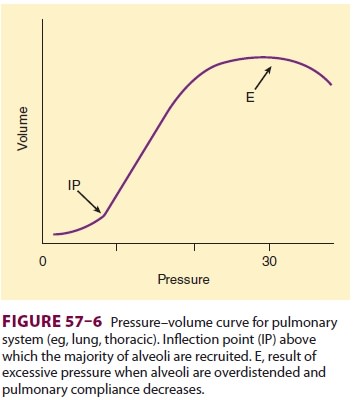
a pressure–volume curve at which collapsed
alveoli are recruited (open); with small changes in pressure there are large
changes in volume (Figure 57–6).
Although neither PEEP nor CPAP decreases total extravascular lung water,
studies suggest that they do redistribute extravascular lung water from the
interstitial space between alveoli and endothelial cells toward peribronchial
and perihilar areas. Both effects can potentially improve arterial oxygenation.
Excessive PEEP or CPAP, however, can
overdis-tend alveoli (and bronchi), increasing dead space ventilation and reducing
lung compliance; both effects can significantly increase the work of
breath-ing. By compressing alveolar capillaries, overdisten-tion of normal
alveoli can also increase pulmonary vascular resistance and right ventricular afterload.
A higher incidence of pulmonary barotrauma is
observed with excessive PEEP or CPAP, particularly
at levels greater than 20 cm H 2O. Disrup-tion of alveoli allows air to track interstitially along
bronchi into the mediastinum (pneumomediasti-num). From the mediastinum, air can
then rupture into the pleural space (pneumothorax) or the peri-cardium
(pneumopericardium) or can dissect along tissue planes subcutaneously
(subcutaneous emphy-sema) or into the abdomen (pneumoperitoneum or
pneumoretroperitoneum). A bronchopleural fistula is the result of failure of an
air leak to seal (close). Although barotrauma must be considered in any
discussion of CPAP and PEEP, in fact, it may be more clearly associated with
higher peak inspira-tory pressures that result with increasing level of PEEP or
CPAP. Other factors that may increase the risk of barotrauma include underlying
lung disease, stacking of breaths (from too frequent breaths or too short
expiratory times) so that intrinsic PEEP (dynamic hyperinflation or autoPEEP)
develops, large VT (>10–15 mL/kg), and younger age.
Adverse Nonpulmonary Effects of
PEEP & CPAP
Nonpulmonary adverse effects are primarily cir-culatory and are related
to transmission of the ele-vated airway pressure to the contents of the chest.
Fortunately, transmission is directly related to lung compliance; thus,
patients with decreased lung com-pliance (most patients requiring PEEP) are
least affected.
Progressive reductions in cardiac output are often
seen as mean airway pressure and, second-arily, mean intrathoracic pressure
rise. The principal mechanism appears to be intrathoracic pressure– related
inhibition of return of venous blood to the heart. Other mechanisms may include
leftward displacement of the interventricular septum (inter-fering with left
ventricular filling) because of the increase in pulmonary vascular resistance
(increased right ventricular afterload) from overdistention of alveoli, leading
to an increase in right ventricular volume. Left ventricular compliance may
therefore be reduced; when this occurs, to achieve the same cardiac output may
require a higher filling pressure. An increase in intravascular volume will
usually at least partially offset the effects of CPAP and PEEP on cardiac
output. Circulatory depression is most often associated with end-expiratory
pressures greater than 15 cm H2O.
PEEP-induced
elevations in central venous pressure and reductions in cardiac output decrease
both renal and hepatic blood flow. Circulating levels of antidiuretic hormone
and angiotensin are usually elevated. Urinary output, glomerular filtration,
and free water clearance decrease. Increased end-expiratory pressures, because
they impede blood drainage from the brain and blood return to the
heart, may increase intracranial pressure in patients whose ventricular
compliance is decreased. Therefore, in patients on mechanical ventilation for
acute lung injury and who have evi-dence of increased intracranial pressure,
the level of PEEP must be carefully chosen to balance oxygen-ation requirements
against potential adverse effects on intracranial pressure.
Optimum Use of PEEP & CPAP
The goal of positive-pressure therapy is to
increase oxygen delivery to tissues, while avoiding the adverse sequelae of
excessively increased (>0.5) FIO2. The latter is best
accomplished with an adequate car-diac output and hemoglobin concentration.
Ideally, mixed venous oxygen tensions or the arteriovenous oxygen content
difference should be followed. The salutary effect of PEEP (or CPAP) on arterial
oxygen tension must be balanced against any detrimental effect on cardiac
output. Volume infusion or inotro-pic support may be necessary and should be
guided by hemodynamic measurements.
At optimal
PEEP the beneficial effects of PEEP exceed any detrimental risks. Practically,
PEEP is usually added in increments of 3–5 cm H2O until the desired
therapeutic end point is reached. The most commonly suggested end point is an
arte-rial oxygen saturation of hemoglobin of greater than 88–90% on a nontoxic
inspired oxygen con-centration (≤50%). Many clinicians favor reducing
the inspired oxygen concentration to 50% or less because of the potentially
adverse effect of greater oxygen concentrations on the lung. Alternatively,
PEEP may be titrated to the mixed venous artery oxygen saturation (SVO2> 50–60%). Monitoring lung compliance and dead space has also been
suggested.
Related Topics