Chapter: Psychology: Treatment of Mental Disorders
Pharmacological Treatments of Mental Disorders
Pharmacological Treatments
The
past half century or so has seen the development of a number of psychotropicdrugs, medications that
control, or at least moderate, the manifestations of mentaldisorders. These
medications have had an enormous impact on mental-health care and have allowed
patients with many different disorders to be treated successfully, often
without hospitalization. The development of psychotropic drugs has gone hand in
hand with advances in our understanding of mental illness. As we have learned
more about the causes of each disorder, better understanding has often pointed
the way toward new drug treatments, and—conversely—new drug treatments have
often allowed us to test claims about what the causes might be. At the same time,
many of these treatments have important limitations, which we will also
consider.
ANTIPSYCHOTIC MEDICATIONS
As we discussed, schizophrenia involves many elements, including a spe-cific chemical abnormality involving the neurotransmitter dopamine. A major source of evidence for this neurochemical abnormality was the effectiveness of certain drugs called antipsychotics. The classical antipsychotics, most of which were developed in the 1950s, reduce the major positive symptoms of schizophrenia, such as thought disorder and hallucination , apparently by blocking dopamine receptors in key brain pathways; the most common versions of these antipsychotics include Thorazine, Haldol, and Stelazine.
Unfortunately,
these drugs are less effective in treating negative symptoms, such as flat
affect and the inability to feel pleasure. This shortcoming has been addressed
by a newer set of medications, the so-called atypical antipsychotics, such as
Risperdal, Clozaril, and Seroquel (Glick, Murray, Vasudevan, Marder, & Hu,
2001). Like the clas-sical antipsychotics, the atypical antipsychotics block
the neurotransmission of dopamine, but their enhanced benefits, especially with
negative symptoms, are proba-bly caused by other mechanisms. These may include
alterations in serotonin neuro-transmission or a more selective effect on
particular subsets of dopamine neurons (Bantick, Deakin, & Grasby, 2001;
Kapur & Remington, 2001; Tandon & Kane, 1993; Wirshing, Marder, Van Putten,
& Ames, 1995).
These
medications have provided enormous advances in our treatment of schizo-phrenia;
however, the benefits have not come without problems. In the United States, the
development of the first antipsychotic medications in the 1950s spurred a
move-ment called deinstitutionalization,
intended to allow for better and less expensive care for patients in their own
communities, at local community mental-health centers rather than at large,
centralized hospitals. To some extent, this movement worked. In the 1950s,
mental hospitals housed about 600,000 patients; the current figure is a mere
10% of that figure (Lamberg, 2004). Medications have also made it possible to
discharge patients with schizophrenia more quickly than ever. Prior to the
introduction of the antipsychotic medications, two out of three patients with
schizophrenia spent most of their lives in mental hospitals. In the 1980s, the
average stay was about two months (J. M. Davis, 1985a; Lamb, 1984; Lamb &
Bachrach, 2001).
But
deinstitutionalization was at best a partial success. Many individuals with
serious mental illness have not found appropriate care in community settings,
nor have they been integrated into the community, in terms of employment, or
social relations, or support services. As a result, some 200,000 mentally ill
individuals are homeless in the United States, and an equal number are in jail,
often for minor crimes (Fleishman, 2004; Lamberg, 2004; Figure 17.15).
Thousands more exist in marginal living situations, and although it is true
that fewer patients with schizo-phrenia remain in psychiatric hospitals, the
number of times they are readmitted for short stays has increased by 80% since
the 1960s (Rosenstein, Milazzo-Sayre, & Manderscheid, 1989). The current
inadequacy—and, in many cases, the complete lack—of appropriate community
services for mental illness may be linked to the point, raised earlier, that
the outlook for a patient with schizophrenia is appre-ciably worse in the
United States than it is in countries where such support is more available.

ANTIDEPRESSANT AND MOOD - STABILIZING MEDICATIONS
Even
with all their complications, the antipsychotic medications have provided
enor-mous advances in the treatment of schizophrenia. Medications have been
just as important in the treatment of other mental disorders. Shortly after the
introduction of antipsychotics as a treatment for schizophrenia, drugs were
found that seemed to act specifically on depression. These early antidepressants fell into two major
classes, the monoamine oxidase (MAO) inhibitors, such as Nardil, and the
tricyclic antidepressants (so called because of their chemical structure), such
as Tofranil. Of these, the tricyclics became the more widely used, mostly
because patients tak-ing MAO inhibitors must conform to difficult dietary
restrictions (Burke & Preskhorn, 1995).
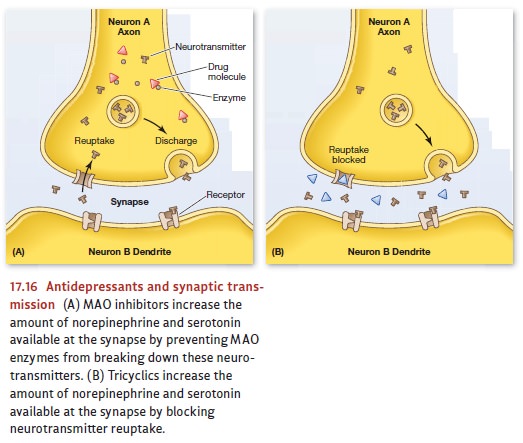
Both
the MAO inhibitors and the tricyclics appear to work in part by increasing the
amount of norepinephrine and serotonin available for synaptic transmission
(Blier & Abbott, 2001). These two classes of drugs accomplish this in different
ways (for details, see Figure 17.16), but both are effective in alleviating
depression, producing marked improvement in up to 65% of the patients who take
them (Hollister & Csernansky, 1990). The drugs are not interchangeable,
however, because some patients do far better with one than with another
(Hollon, Thase, & Markowitz, 2002), presumably because different patients
suffer from somewhat different neuro-chemical deficits.
These
early drug treatments for depression were quite successful, but the use of
med-ication for treating depression changed dramatically in 1988 with the
introduction of the first “designer drug” for depression, Prozac (Kramer,
1993). Prozac was engineered to act minimally on norepinephrine and dopamine
and maximally on serotonin, thus marking a new class of antidepressants, known
as selective serotonin reuptake
inhibitors(SSRIs). For most
patients, Prozac and the other SSRIs (such as Zoloft, Paxil, Celexa,and
Lexapro) ameliorate depression as effectively and as completely as their
predeces-sors (Mulrow et al., 2000), but they have fewer side effects. SSRIs
have become so popular—both for depression and for other disorders, such as
eating disorders and anxiety disorders—that they are now prescribed not only by
psychiatrists but also by primary-care physicians (Olfson & Klerman, 1993).
For
patients who are bothered by side effects from SSRIs and cannot take the
tricyclics or MAO inhibitors, a separate array of antidepressants is available.
These atypical antidepressants, which
work in various ways on serotonin, norepinephrine,and dopamine systems, include
Wellbutrin, Effexor, and Serzone. Of these, Wellbutrin is of special interest.
It seems to have little effect on serotonin and prob-ably operates on the
dopamine and norepinephrine systems instead. However it works, Wellbutrin has
none of the negative sexual side effects associated with some of the other
antidepressants; indeed, many patients actually report heightened sex-ual
interest and response. It is also generally stimulating, and in addition to
curbing nicotine cravings (for which it is sold as Zyban), it is widely used to
treat adults with attention-deficit/hyperactivity disorder.
While
some have touted these new antidepressants as panaceas, they—like all
medications—have limitations. First, the beneficial effects of the drug fully
emerge only after the medication is taken for a month or so. In addition,
considerable uncertainty persists about why each of the antidepressants helps
some people but not others. As aresult, therapists often resort to a process of
trial and error, prescribing a variety of drugs, one at a time, until they find
one that helps the particular patient. This trial and error can take quite some
time since, as we have said, the drugs often take effect only after a month or
so.
In
addition, all of these drugs do have side effects, which vary by drug and by
person but which, overall, include weight gain, nausea, diarrhea, and—in upward
of 30% of patients—insomnia, as well as a loss of sexual desire or response
(Clayton, 2001; Ferguson, 2001; Gursky & Krahn, 2000; E. Hollander &
McCarley, 1992; Jacobsen, 1992; Montgomery, 1995). There has also been some
concern that these drugs may be addictive, although many regard the evidence
supporting this claim as unpersuasive (Haddad, 1999).
Medications
to treat bipolar disorder are called mood
stabilizers (so called because they treat or prevent manic, mixed, or
depressive states). The first medication used specifically for its mood
stabilizing action was lithium carbonate. Patients who begin taking lithium can
generally expect that their manic episodes will subside within 5 to 10 days.
Because of this delay, antipsychotic medications are sometimes also prescribed
to reduce agitation until the lithium takes effect. In addition,
antidepressants are often prescribed along with lithium because lithium is more
effective in reducing manic than in reducing depressive symptoms (Sachs &
Thase, 2000).
Unfortunately,
lithium works in only 60 to 70% of patients with bipolar disorder (Miklowitz
& Johnson, 2007). Taking it also causes unpleasant side effects for many patients,
such as weight gain, sedation, dry mouth, and tremors. Moreover, the fact that
lithium is toxic at higher doses means that patients must have their blood
tested fre-quently and makes it a risky treatment for patients who are
potentially suicidal and might overdose. Nor can lithium be taken during
pregnancy or when a patient has kid-ney disease. In addition, many patients
with bipolar disorder choose not to take their medication because they do not
want to give up either the pleasures or the productivity often associated with
the manic state; they may decide that these elements outweigh the (sometimes
enormous) disruption and distress that the disorder also produces.
ANTIANXIETY MEDICATIONS
Patients
suffering from disabling anxiety are often treated with medications that are
popularly called tranquilizers and technically known as anxiolytics. Most anxiolytics apparently work by increasing
neurotransmission at synapses containing the neurotransmitter gamma-aminobutric
acid (GABA; Brailowsky & Garcia, 1999; Shader & Greenblatt, 1995).
Probably the most commonly used anxiolytic, though, is self-administered and
not prescribed by a therapist: alcohol. Many people, patients or otherwise,
medicate themselves for their anxiety as part of their lifestyle, inviting the
risk of alcohol abuse.
Physicians
also sometimes treat the anxiety disorders by prescribing a beta-blocker, a medication intended to
control autonomic arousal. In addition, for the uncomfortable feelings
associated with anxiety, they often prescribe one of the benzodiazepines. Some of these medications, such as Valium Xanax,
or Klonopin are prescribed so often that their names have become household
words. They are useful as short-term treatments (usually taking effect in 30 or
40 minutes) for generalized anxiety disorder, panic dis-order, post-traumatic
stress disorder, alcohol withdrawal, insomnia, muscle spasms, tension
headaches, and various other stress-related disorders.
Benzodiazepines
are rarely used for long-term treatment, however, because they are highly
addictive, interact dangerously with alcohol, and like alcohol, can cause
profound fetal damage if a patient is pregnant. There are also concerns that
the benzo-diazepines can produce a “rebound” effect, so that a person who stops
taking the med-ication may end up more anxious than she was prior to taking the
drug (Chouinard, 2004). These are, of course, major concerns, and so
researchers continue to seek new anxiolytics. Happily, some of the newer drugs,
such as BuSpar, are not addictive and are becoming popular substitutes for the
older medications, especially for patients who are prone to alcohol or drug
abuse or have to take the medication over a long period of time (Lydiard,
Brawman-Mintzer, & Ballenger, 1996; Schweitzer, Rickels, & Uhlenhuth,
1995). Anxiety is also commonly treated using tricyclic antidepressants and
SSRIs.
COSTS AND BENEFITS OF MEDICATION
Overall,
drug therapies effectively reduce symptoms for people with a wide range of
mental disorders. Indeed, these drugs have lifted many people out of misery and
distress and help them resume a reasonably normal life. These drugs have also
allowed many patients to remain in a family or community setting when they
would otherwise have been relegated to the hospital. These are, by any measure,
enormous gains.
At
the same time, drug therapy for mental disorders is not an unqualified success.
Drugs do not cure the disorder; instead, they help to control it, and in many
cases, if the person stops taking his pills, he quickly relapses into his full
set of symptoms. It is also often tricky to get the doses correct for many of these drugs, because individuals differ
both in their drug reactions and in how rapidly they metabolize the medication.
There are also some group differences—for example, many African Americans
metabolize antidepressant medications more slowly than whites (U.S. Department
of Health and Human Services, 2001). This aggravates a concern that we have
already raised— namely, that a clinician prescribing one of these medications
often needs some amount of trial and error to find the right drug at the right
dose.
We
have also highlighted the fact that many of these drugs have unpleasant (or
even dangerous) side effects. These include increases as well as decreases in
sleepiness and agitation, decreased sexual performance, and facial tics. These
side effects are bad enough on their own, but, in addition, they often lead
people to stop taking their medication, in which case (obviously) the drugs can
bring no benefit. Thus, a col-lege student might refuse to take her
antidepressant because she is concerned about the associated weight gain, even
though, without the drug, her bulimia nervosa may cause long-term health
prob-lems. Likewise, someone might refuse to take medication because of a fear
that it will disrupt sexual functioning, even though, without it, depression is
disrupting his life to the point where a relationship is not possible.
A
more general concern about these psychoactive drugs is that they may be overprescribed
(Figure 17.17). This worry is magnified by the fact that these drugs are
increasingly used to treat a wide variety of conditions in children and
adolescents, even though we know less about their side effects in children than
in adults. Two flashpoints for this particular controversy are the use of SSRIs
for depres-sion and the use of stimulants for ADHD.
Some
researchers have raised questions about whether the SSRIs may increase the risk
of suicide in some young people. But others claim the opposite is the case, and
that by alleviating depression, SSRIs decrease
suicide risk (Bachtold, 2003; Couzin, 2004). In view of the potential for
risks, however, the U.S. Food and Drug Administration (FDA) now requires “black
box” warnings on SSRIs (literally, a black box around the label) regarding
possible increased risk of suicide (Figure 17.18).


As
we have seen, controversy also swirls around the number of prescriptions now
being written for ADHD, which has increased fourfold since the early 1990s.
Critics claim that active children are being medicated so that they will behave
themselves in crowded and understimulating classrooms where they are expected
to perform well on standardized tests. Others reply that medications are an
important treatment tool as long as children are thoroughly assessed by
appropriate professionals.
Related Topics