Chapter: Pharmaceutical Biotechnology: Fundamentals and Applications : Pharmacokinetics and Pharmacodynamics of Peptide and Protein Drugs
Pharmacokinetics and Pharmacodynamics of Peptide and Protein Drugs
Pharmacokinetics and Pharmacodynamics of Peptide and Protein Drugs
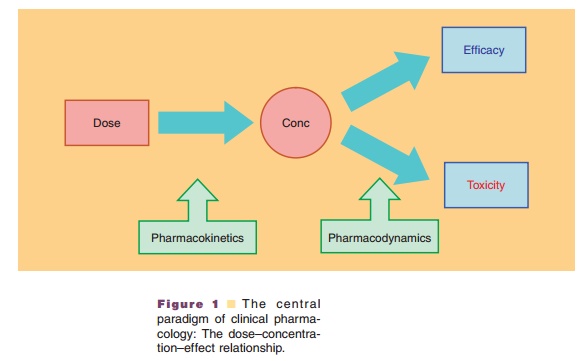
The rational use of drugs and the design of effective dosage regimens
are facilitated by the appreciation of the central paradigm of clinical
pharmacology that there is a defined relationship between the administered dose
of a drug, the resulting drug concentrations in various body fluids and
tissues, and the intensity of pharmacologic effects caused by these
concentrations (Meibohm and Derendorf, 1997). This dose–exposure–response
relation-ship and thus the dose of a drug required to achieve a certain effect
are determined by the drug’s pharmacoki-netic and pharmacodynamic properties
(Fig. 1).
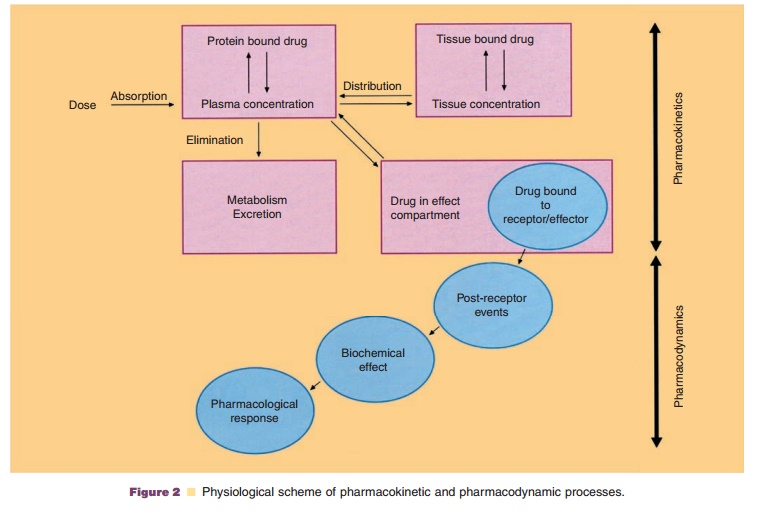
Pharmacokinetics describes the time course of the concentration of a
drug in a body fluid, preferably plasma or blood, that results from the
administration of a certain dosage regimen. It comprises all processes
affecting drug absorption, distribution, metabolism, and excretion. Simplified,
pharmacokinetics charac-terizes what the body does to the drug. In contrast, pharmacodynamics characterizes the intensity of a drug
effect or toxicity resulting from certain drug concentrations in a body fluid,
usually at the assumed site of drug action. It can be simplified to what the drugdoes to the
body (Fig. 2) (Holford and Sheiner, 1982;Derendorf and
Meibohm, 1999).
The understanding of the dose–concentration– effect relationship is
crucial to any drug — including peptides and proteins — as it lays the
foundation for dosing regimen design and rational clinical application. General
pharmacokinetic and pharmacodynamics prin-ciples are to a large extent equally
applicable to protein and peptide drugs as they are to traditional small
molecule-based therapeutics. Deviations from some of these principles and
additional challenges with regard to the characterization of the
pharmacokinetics and phar-macodynamics of peptide and protein therapeutics,
however, arise from some of their specific properties:
a.
Their structural similarity to
endogenous struc-tural or functional proteins and nutrients.
b.
Their intimate involvement in
physiologic pro-cesses on the molecular level, often including regulatory
feedback mechanisms.
c.
The analytical challenges to
identify and quan-tify them in the presence of a myriad of similar molecules.
d.
Their large molecular weight and
macromolecule character (for proteins).
This chapter will highlight some of the major pharmacokinetic properties
and processes relevant for the majority of peptide and protein therapeutics and
will provide examples of well-characterized pharmacody-namic relationships for
peptide and protein drugs. The clinical pharmacology of monoclonal antibodies,
includ-ing special aspects in their pharmacokinetic and phar-macodynamics, will
be discussed in further detail. For a more general discussion on pharma-cokinetic
and pharmacodynamic principles, the reader is referred to several textbooks and
articles that review the topic in extensive detail .
Related Topics