Chapter: Genetics and Molecular Biology: Lambda Phage Integration and Excision
Mapping Integrated Lambda
Mapping Integrated Lambda
How do we
know lambda phage associates with the chromosome when it lysogenizes a cell?
Lambda lysogens could possess copies of the phage DNA freely floating in the
cytoplasm. A problem generated by this mode of existence is that the molecules
would be randomly inherited by daughter cells on division. As a result, the
lower the average number of lambda molecules per cell, the higher the fraction
of daughters that might fail to inherit any and therefore become nonlysogenic.
To explain lambda’s low rate of spontaneously becoming nonlysogenic, which is
about 10- 6 per doubling, cells would have to possess unacceptably
large numbers of the phage genomes. A second method to ensure proper
inheritance of lysogeny would be for a nonintegrated lambda to possess a
special segregation mechanism. Several genetic elements use this mechanism for
stable inheritance. These include the phage P1 and the F factor, both of which
have been discussed earlier in other contexts. This mechanism does not require
large numbers of the lambda genome to be present. The third and simplest
segregation mechanism would be for lambda to attach to or integrate into the
host chromosome. Then it would be replicated and segregated into daughter cells
with the host chromosomes.
Elegant genetic experiments showed not only that lambda associates with the chromosome but that it associates with a specific site on the chromosome. Now, of course, a simple Southern transfer experiment could settle the issue. The original data suggesting that the lambda DNA was associated with a particular region of the chromosome came from mating experiments between lysogenic male bacteria and nonlysogenic female bacteria.
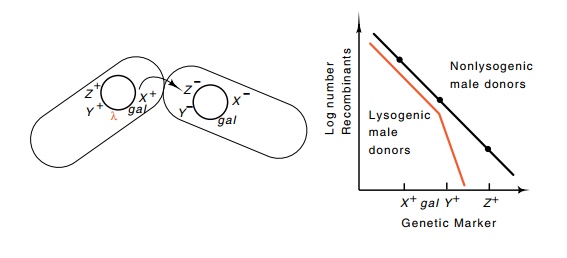
Figure 18.1 The transfer frequency of markers from lambda lysogens or non-lysogens. Marker X+ is located near the origin of transfer, and markers Y+ and Z+are transferred after the point of lambda insertion.
Transfer of genetic markers lying between the origin of transfer and the gal region showed no differences between
lysogenic and nonlysogenic males. The frequency of female recombinants
incor-porating genetic markers transferred after the gal genes, however, revealed a discrepancy (Fig. 18.1). Their
frequency was much lower if the DNA was transferred from lysogenic males than
if the DNA came from nonlysogenic males. This can be understood as follows: if
lambda is attached to the host genome in the vicinity of gal, then when it enters a nonlysogenic female, it finds itself in
an environment lacking lambda repressor. As a result, the phage induces and
proceeds through a lytic cycle analogous to infecting a cell. This process of a
phage inducing upon its transfer into a nonlysogenic female is called zygotic
induction. Its parallel for the lac
operon leads to temporary induction of the lacZYA
genes upon their introduction into cells deleted of the operon. Later as lac repressor accumulates, transcription
of the genes shuts off.
A minor paradox is raised by the lambda results
discussed above. The DNA is transferred to females in a linear fashion
beginning at a particu-lar point. Therefore it would seem that if a genetic
marker lying ahead of the integrated lambda were transferred to females, the
lambda itself eventually would also be transferred. Why are such cells not
killed by zygotic induction?
The resolution to the paradox is that the majority
of recipient cells that receive a gal-proximal
marker also do not also receive gal.
The DNA strand being transferred frequently breaks and conjugation stops. Even
though genetic markers lying near the origin are transferred at high
efficiency, markers lying farther away, and therefore transferred later in the
mating process, are transferred with substantially lower efficien-cies.
Consequently, most cells receiving a marker proximal to lambda or gal never receive lambda or gal; so zygotic induction occurs in only
a tiny fraction of the cells that receive markers transferred before gal.
Related Topics