Chapter: Essential Microbiology: Microbial Metabolism
Glycolysis
Why glucose?
By concentrating on glucose catabolism in this way, you may think we are ignoring the fate of other nutrient molecules. If you take another look at Figure 6.15, however, you will notice that the breakdown products of lipids, proteins and nucleic acids also find their way into our pathway sooner or later, having undergone transformations of their own.
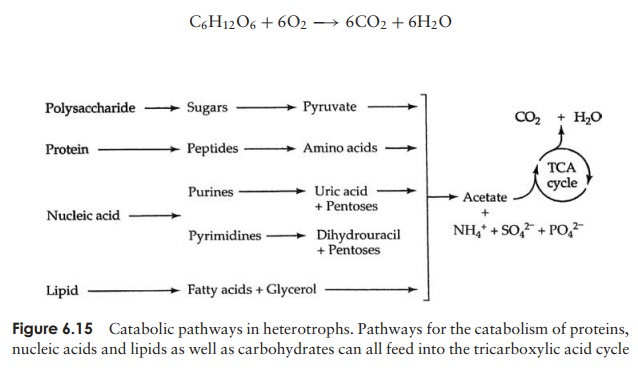
Glycolysis
The initial sequence of reactions, in which a molecule of glucose is converted to two molecules of pyruvate∗ , is called glycolysis(Figures 6.16 and 6.17). In the first phase of glycolysis, glucose is phosphorylated and its six-carbon ring structure rearranged, before being cleaved into two three-carbon molecules. In the second phase, each of these undergoes oxidation, resulting in pyruvate.
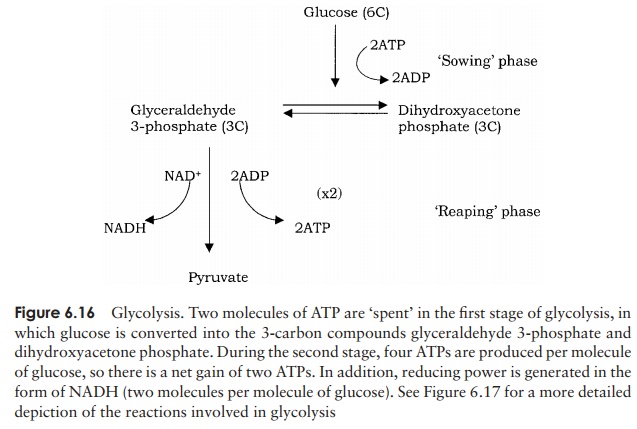
Also known as the Embden–Meyerhof pathway, glycolysis is used for the metabolism of simple sugars not just by microorganisms, but by most living cells. The pathway, which takes place in the cytoplasm, comprises a series of 10 linked reactions, in which each molecule of the six-carbon glucose is converted to two molecules of the three-carbon pyruvate, with a net gain of two molecules of ATP.
The full pathway of reactions is shown in Figure 6.17. Note how, in terms of energy, glycolysis can be divided conveniently into a ‘sowing’ phase, in which two molecules of ATP are expended per molecule of glucose, followed by a ‘reaping’ phase which yieldsfour molecules of ATP. The overall energy balance is therefore a gain of two moleculesof ATP for each molecule of glucose oxidised to pyruvate. In addition, the second phasefeatures the conversion of two molecules of NAD+ to NADH, which, as we will see, act as an important source of reducing power in subsequent pathways.
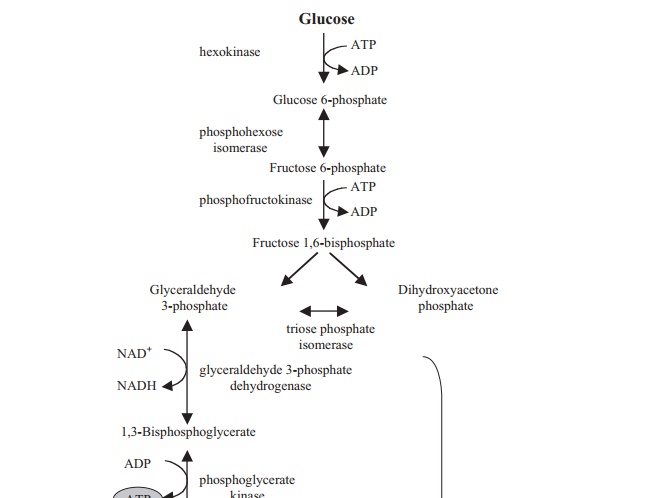
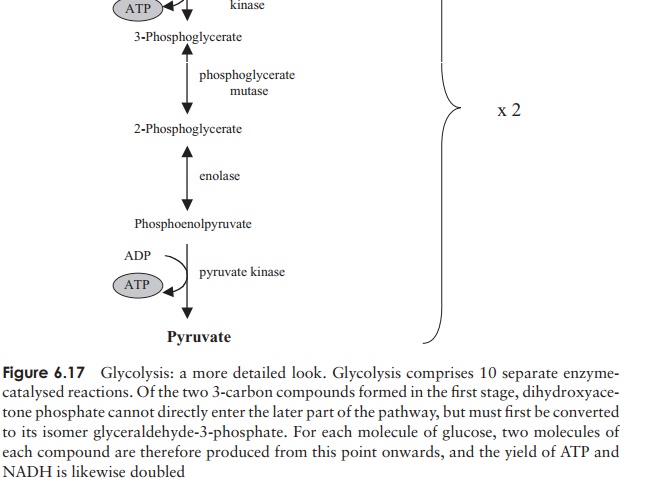
The reactions by which ATP is generated from ADP in the second phase of glycolysis are examples of substrate-level phosphorylation, so-called because the phosphate group is transferred directly from a donor molecule.
What happens next to the pyruvate produced by glycolysis depends on the organism concerned, and on whether the environment is aerobic or anaerobic; we shall look at these possibilities in due course.
Glycolysis is not the only way to metabolise glucose
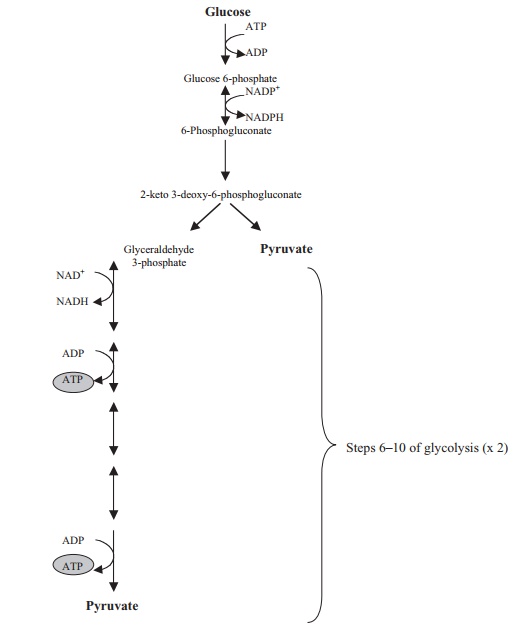
Although glycolysis is widespread in both the microbial and nonmicrobial worlds, sev-eral bacterial types use alternative pathways to oxidise glucose. For certain Gram-negative groups, notably the pseudomonads, the main route used is the Entner–Doudoroff pathway, producing a mixture of pyruvate and glyceraldehyde-3-phosphate (Figure 6.18). The former, like that produced in glycolysis, can enter a number of pathways, while the latter can feed into the later stages of glycolysis. The net result of catabolism by the Entner–Doudoroff pathway is the production of one molecule each of ATP, NADH and NADPH per molecule of glucose degraded.

A secondary pathway, which may operate in tandem with glycolysis or the Entner– Doudoroff pathway, is the pentose phosphate pathway, sometimes known as the hexosemonophosphate shunt (Figure 6.19). Like glycolysis, the pathway can operate in thepresence or absence of oxygen. Although glyceraldehyde-3-phosphate can once again enter the glycolytic pathway and lead to ATP generation, for most organisms the path-way has a mainly anabolic (biosynthetic) function, acting as a source of precursor molecules for other metabolic pathways. The pentose phosphate pathway is a useful
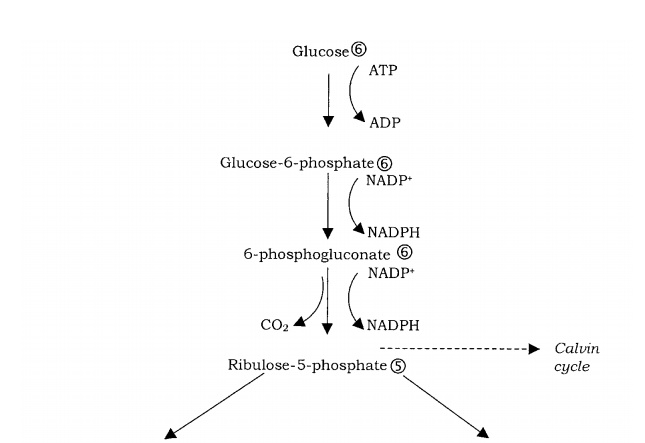
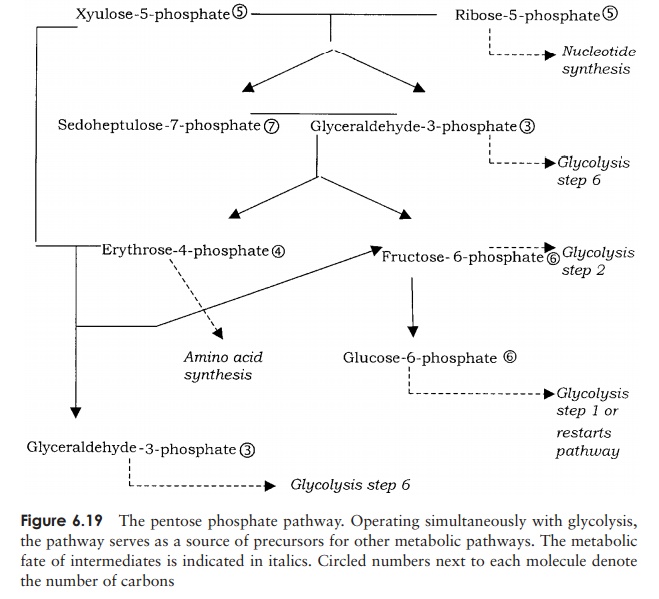
source of reducing power in the form of NADPH. In addition it acts as an important source of precursors in the synthesis of essential molecules; ribose-5-phosphate is an important precursor in the synthesis of nucleotides, while the four-carbon erythrose 4-phosphate is required for the synthesis of certain amino acids and ribulose-5-phosphate is an intermediate in the Calvin cycle of carbon fixation.
Related Topics