Chapter: Essentials of Anatomy and Physiology: Respiratory System
Gas Transport in the Blood
GAS TRANSPORT IN THE BLOOD
Oxygen Transport
After O2 diffuses through the respiratory membrane into the blood, about 98.5% of the O2 transported in the blood combines reversibly with the iron-containing heme groups of hemoglobin . About 1.5% of the O2remains dissolved in the plasma. Hemoglobin with O2 bound to its heme groups is called oxyhemoglobin (ok′ sē-hē-mō-glō′ bin).
The ability of hemoglobin to bind to O2 depends on the Po2. At high Po2, hemoglobin binds to O2, and at low Po2, hemoglobin releases O2. In the lungs, Po2 normally is sufficiently high so that hemoglobin holds as much O2 as it can. In the tissues, Po2 is lower because the tissues are using O2. Consequently, hemoglobin releases O2 in the tissues. Oxygen then diffuses into the cells, which use it in cellular respiration. At rest, approximately 23% of the O2 picked up by hemoglobin in the lungs is released to the tissues.
The amount of O2 released from oxyhemoglobin is influ-enced by four factors. More O2 is released from hemoglobin if(1) the Po2 is low, (2) the Pco2 is high, (3) the pH is low, and(4) the temperature is high. Increased muscular activity results in a decreased Po2, an increased Pco2, a reduced pH, and an increased temperature. Consequently, during physical exercise, as much as 73% of the O2 picked up by hemoglobin in the lungs is released into skeletal muscles.
Carbon Dioxide Transport and Blood pH
Carbon dioxide diffuses from cells, where it is produced, into the tissue capillaries. After CO2 enters the blood, it is transported in three ways: (1) About 7% is transported as CO2 dissolved in the plasma; (2) 23% is transported in combination with blood proteins, primarily hemoglobin; and (3) 70% is transported in the form of bicarbonate ions.
Carbon dioxide (CO2) reacts with water to form carbonic acid (H2CO3), which then dissociates to form H+ and bicarbonate ions (HCO3−):
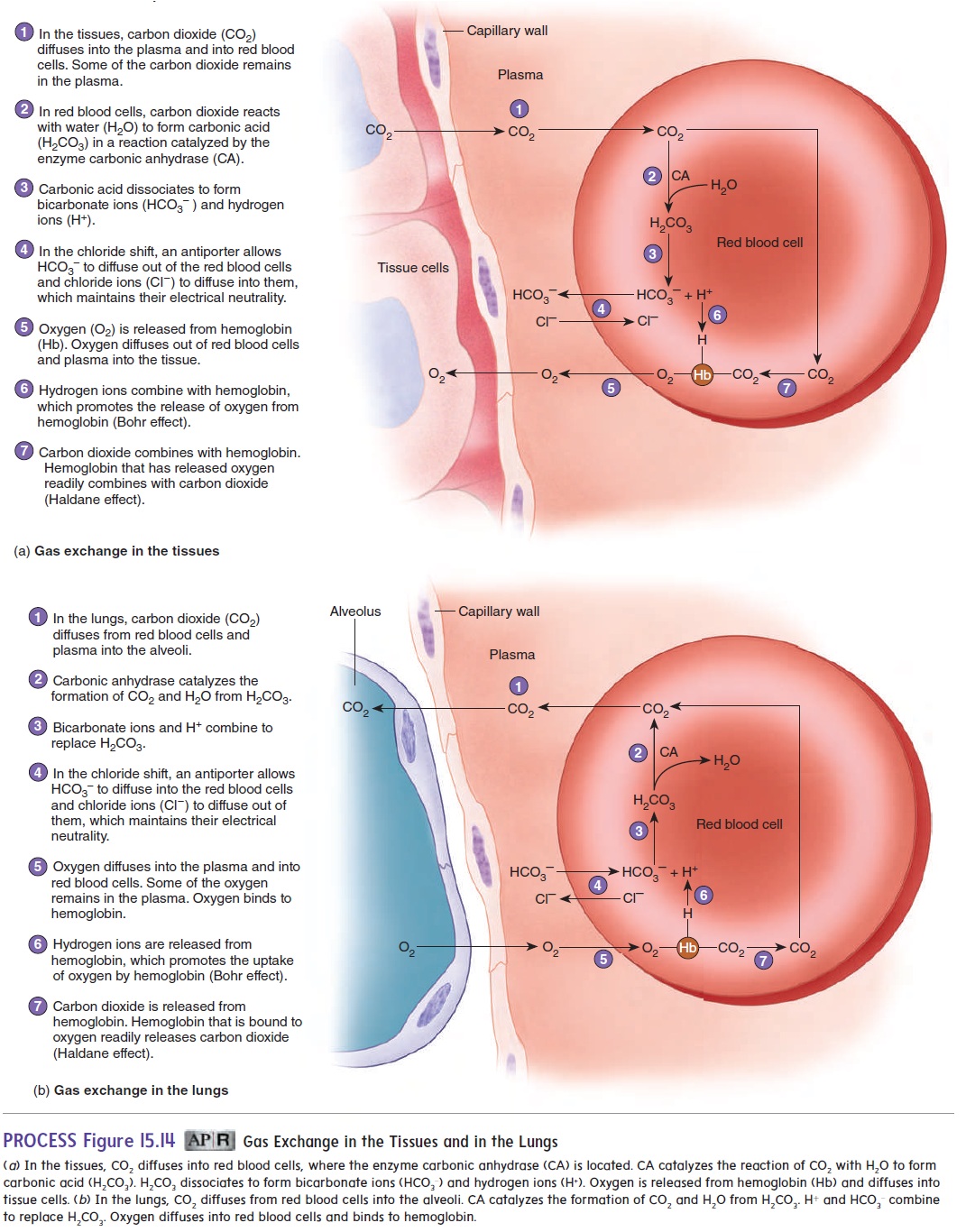
An enzyme called carbonic anhydrase (kar-bon′ ik an-hı̄′ drās) is located inside red blood cells and on the surface of capillary epi-thelial cells. Carbonic anhydrase increases the rate at which CO2 reacts with water to form H+ and HCO3− in the tissue capillaries (figure 15.14a). Thus, carbonic anhydrase promotes the uptake of CO2 by red blood cells.
In the capillaries of the lungs, the process is reversed, so that the HCO3− and H+ combine to produce H2CO3, which then forms CO2 and H2O (figure 15.14b). The CO2 diffuses into the alveoli and is expired.
Carbon dioxide has an important effect on the pH of blood. As CO2 levels increase, the blood pH decreases (becomes more acidic) because CO2 reacts with H2O to form H2CO3. The H+ that results from the dissociation of H2CO3 is responsible for the decrease in pH. Conversely, as blood levels of CO2 decline, the blood pH increases (becomes less acidic, or more basic).
Related Topics