Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: Fish genetics
Freshwater fishes - Fish genetics: Phylogeography
Freshwater fishes
These fishes cannot get out of the water and travel over dry land to the next drainage, with rare exceptions such as “walking catfish” in the genus Clarias. For most freshwater fishes, opportunities for dispersal are few, the genetic differences between drainages are high, and vicariance models generally work well to explain evolutionary patterns. Geographic and oceanographic barriers can explain the majority of sister species (species that are each other’s closest relative) relationships, although differences in freshwater characteristics (such as the Andes-derived white water, and the lowland-derived black water of the Amazon) may be a factor as well. Looking beyond these geographic and oceanographic barriers, four primary factors shape the phylogeography of freshwater fishes:
1 Changes in drainage routes. Stream captures are the most widely studied phenomenon, where erosion, earthquakes, or other geographic changes divert a stream from one drainage to another drainage. The high diversity of freshwater fishes in central North America may be due in part to streams in the Appalachian Mountain Range that switched from flowing toward the Atlantic coast to flowing toward the Mississippi River (Hocutt & Wiley 1986). Flooding, which in essence causes temporary stream capture, may also transfer fishes between drainages.
2 Glaciation. During these “ice ages” the temperate fishes (distributed between 25° and 65° latitude) were massively displaced by the cooling and advancing glaciers. At the end of each glacial epoch, enormous proglacial lakes formed at the retreating edge of ice sheets, some larger than the contemporary Great Lakes of North America. These large and shifting water masses provided extensive opportunities for dispersal (Hocutt & Wiley 1986; Behnke 1992).
3 Coastal opportunities for dispersal. Some freshwater fishes are tolerant of high salinity conditions and can survive for extended periods (days or weeks) in coastal waters. For example, the freshwater cichlids are members of the suborder Labroidei that includes surfperches, damselfishes, wrasses, and parrotfishes, all marine groups. Hence it is no surprise that some cichlid species can tolerate salt water. The other coastal opportunity for dispersal occurs during periods of heavy rainfall. Chesapeake Bay is a 300 km long estuary that usually contains ocean water at one end and fresh water at the other. However, in the aftermath of hurricane events, fresh water extends out of the mouth of the bay, as do the freshwater fishes that are usually confined to individual rivers.
4 Plate tectonics, wherein the movements of continents can separate or join populations of freshwater fishes.
Here we briefly examine three of these phenomena, whereas the fourth (coastal dispersal) is covered in the next section (anadromous fishes).
Reconstructing stream captures
The South Island of New Zealand has two primary drainage systems, the Clutha in the north and Southland in the south. These two historically isolated drainages retain distinct faunas: a phylogeographic survey of galaxiid mudfishes reveals a number of cryptic species among taxa that were previously believed to span both drainages (Waters et al. 2001). Furthermore, these mtDNA studies have de - monstrated two stream captures in this glacially influenced region. The older stream capture involved the Nevis River, which changed course from a southern to a northern drainage system, introducing a lineage of the mudfish Galaxias gollumoides that is characteristic of the Southland drainage (Waters et al. 2001). Molecular clock estimates indicate an ancient colonization, on the order of 300,000–500,000 years ago. In contrast, geological studies indicate that the Von River changed course (also from south to north) during the most recent glacial interval, about 12,000 years ago. The corresponding mtDNA survey indicates the presence of another mudfish derived from the Southland drainage (Burridge et al. 2007). These colonizations of the northern drainage are significant, as they comprise two of the nine native freshwater fishes.
Glacial eradication and recovery
The most recent glaciation affected North America severely, with a greater ice sheet than the Asian and European glaciations combined. This ice sheet reached as far south as the 44° latitude (where Toronto, Ontario and Bangor, Maine are today) about 23,000 years ago, followed by deglaciation 15,000 to 8000 years ago. The current distributions of several salmonids, in particular Lake Trout (Salvelinus
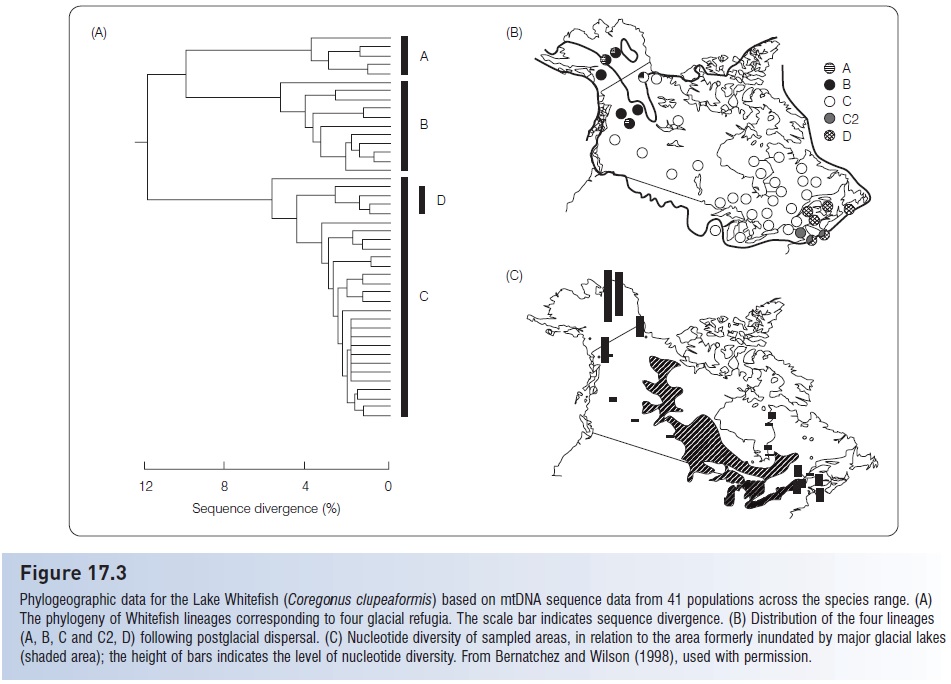
Figure 17.3
Phylogeographic data for the Lake Whitefish (Coregonus clupeaformis) based on mtDNA sequence data from 41 populations across the species range. (A) The phylogeny of Whitefish lineages corresponding to four glacial refugia. The scale bar indicates sequence divergence. (B) Distribution of the four lineages (A, B, C and C2, D) following postglacial dispersal. (C) Nucleotide diversity of sampled areas, in relation to the area formerly inundated by major glacial lakes (shaded area); the height of bars indicates the level of nucleotide diversity. From Bernatchez and Wilson (1998), used with permission.
These species, now broadly distributed from Alaska to the Atlantic drainages, must have persisted in refugia (perhaps at the fringe of their current range) for thousands of years. Hence patterns of genetic diversity can help to identify glacial refugial and recolonization pathways.
Bernatchez and Dodson (1991) used mtDNA to resolve four major lineages among populations of Lake Whitefish. These correspond to refugia in northern Eurasia, Beringia (Siberia-Alaska), the Mississippi valley, and perhaps two Atlantic locations (Fig. 17.3). The Mississippi haplotypes occupy the majority of the current range of Lake Whitefish, observed from New York to the Yukon. This is consistent with other fish distributions in indicating that the Mississippi fauna had the greatest opportunities for dispersal through proglacial lakes.
In a review of phylogeographic studies for 42 North American freshwater fishes, Bernatchez and Wilson (1998) observed a significant decline in mtDNA nucleotide diversity (see above, Population genetics) with increasing latitude, a clear indication of southern refugia during the last glacial period. Regression analysis indicates a steep (fivefold) drop in diversity from 25° to 46°N latitude, then consistently low diversity from 46° to 65°N (Fig. 17.4). It is remarkable that the analysis indicated the 46°N boundary for reduced genetic diversity, closely paralleling the southern limit of North American glaciers at 44°N. Almost universally, North American freshwater fishes are genetically depauperate in the deglaciated areas above the 44° to 46°N boundary. A similar pattern is apparent in Europe. Of the five mtDNA lineages observed in European Brown Trout (Salmo trutta), only one has colonized previously glaciated areas (Bernatchez 1995).
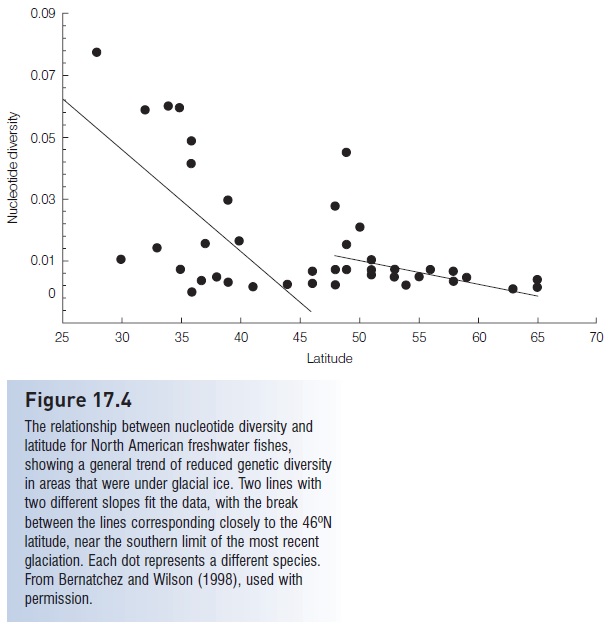
Figure 17.4
The relationship between nucleotide diversity and latitude for North American freshwater fishes, showing a general trend of reduced genetic diversity in areas that were under glacial ice. Two lines with two different slopes fit the data, with the break between the lines corresponding closely to the 46ºN latitude, near the southern limit of the most recent glaciation. Each dot represents a different species.
Plate tectonics: the mystery of the Asian Arowana (Scleropages formosus)
The distribution of arowanas (Osteoglossomorpha) has been a longstanding biogeographic mystery, as these primary freshwater fishes occur on four continents that are isolated by formidable marine barriers (see Fig. 16.13). Their distribution in South America, Africa, and Australia can be explained by the breakup of the southern supercontinent Gondwanaland (including South America, Africa, Antartica, Madagascar, and India) about 150 mybp. However, the distribution in Southeast Asia is hard to explain without a marine dispersal event from Australia. Despite the strict
freshwater requirements of arowanas, this was the favored explanation until recently. The Australia to Asia dispersal hypothesis was given further support by taxonomic studies based on morphology, which united in one genus the Asian Scleropages formosa with the Australian S. jardinii and S. leichardtii.
Based on a molecular clock for two mtDNA genes calibrated with several bony fishes, Kumazawa and Nishida (2000) estimate that the Asian and Australian arowanas actually diverged about 140 mybp. This timeframe coincides with the separation of India from the southern supercontinent, and subsequent transport into the northern hemisphere. India connected with Asia by about 40 mybp, and may have allowed the colonization of Asia at that time. This possibility is supported by the presence of fossil Scleropages in Sumatra, dating to the Eocene (35–57 mybp). Hence the biogeographic mystery of the arowanas unraveled when molecular clock data showed that theScleropages species in Asia and Australia diverged in the Early Cretaceous, much farther back than typical congeners.
Related Topics