Chapter: 11th Chemistry : UNIT 2 : Quantum Mechanical Model of Atom
Electronic configuration of atoms
Electronic configuration of atoms
The distribution of electrons into various orbitals of an atom is called its electronic configuration. It can be written by applying the aufbau principle, Pauli exclusion principle and Hund's rule. The electronic configuration is written as nlx , where n represents the principle of quantum number, 'l' represents the letter designation of the orbital [s(l=0), p (l=1), d(l=2) and f(l=3)] and 'x' represents the number of electron present in that orbital.
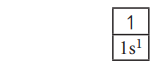
Let us consider the hydrogen atom which has only one electron and it occupies the lowest energy orbital i.e. 1s according to aufbau principle. In this case n=1; l=s; x=1.
Hence the electronic configuration is 1s1. (spoken as one-ess-one).
The orbital diagram for this configuration is,
The electronic configuration and orbital diagram for the elements upto atomic number 10 are given below :

The actual electronic configuration of some elements such as chromium and copper slightly differ from the expected electronic configuration in accordance with the Aufbau principle.
For chromium - 24
Expected configuration :
1s2 2s2 2p6 3s2 3p6 3d4 4s2
Actual configuration :
1s2 2s2 2p6 3s2 3p6 3d5 4s1
For copper - 29
Expected configuration :
1s2 2s2 2p6 3s2 3p6 3d9 4s2
Actual configuration :
1s2 2s2 2p6 3s2 3p6 3d10 4s1
The reason for above observed configuration is that fully filled orbitals and half filled orbitals have been found to have extra stability. In other words, p3, p6, d5, d10, f7 and f 14 configurations are more stable than p2, p5, d4, d 9, f6 and f13. Due to this stability, one of the 4s electrons occupies the 3d orbital in chromium and copper to attain the half filled and the completely filled configurations respectively.
Related Topics