Chapter: Clinical Anesthesiology: Perioperative & Critical Care Medicine: Acid-Base Management
Diagnosis of Acid Base Disorders
DIAGNOSIS OF ACID BASE DISORDERS
Interpretation
of acid–base status from analysis of blood gases requires a systematic
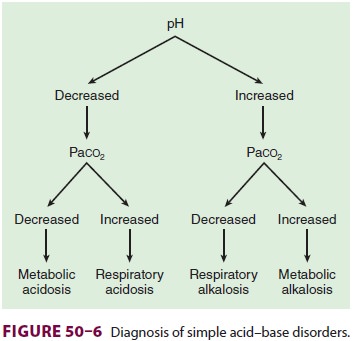
approach. A rec-ommended approach follows (Figure 50–6):
·
Examine arterial pH: Is acidemia or alkalemia present?
·
Examine Paco2: Is the change in Paco2 consistent
with a respiratory component?
·
If the change in Paco2 does not
explain the change in arterial pH, does the change in [HCO3−] indicate a metabolic
component?
·
Make a tentative diagnosis (see Table 50–1).
·
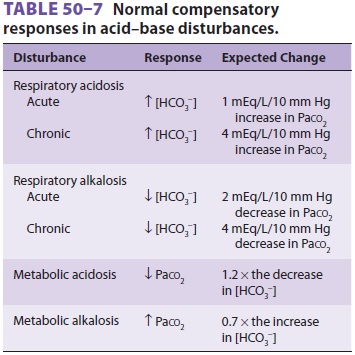
Compare the change in [HCO3−] with the change in
Paco 2. Does a compensatory response exist (Table 50–7)? Because arterial pH
is related to the ratio of Pa co2 to [HCO3−], both respiratory and
renal compensatory mechanisms are always such that Paco2and [HCO3−] change in the same
direction.
·
A change in opposite directions implies a mixed acid–base disorder.
·
If the compensatory response is more or less than expected, by
definition, a mixed acid–base disorder exists.
·
Calculate the plasma anion gap in the case of metabolic acidosis.
·
Measure urinary chloride concentration in the case of metabolic
alkalosis.
An
alternative approach that is rapid, but per-haps less precise, is to correlate
changes in pH with changes in CO2 or HCO3. For a
respiratory dis-turbance, every 10 mm Hg change in CO2 should change
arterial pH by approximately 0.08 U in the opposite direction. During metabolic
disturbances, every 6 mEq change in HCO 3 also changes arterial pH
by 0.1 in the same direction. If the change in pH exceeds or is less than
predicated, a mixed acid–base disorder is likely to be present.
Related Topics