Chapter: Clinical Anesthesiology: Anesthetic Management: Cardiovascular Physiology & Anesthesia
Cardiac Action Potentials
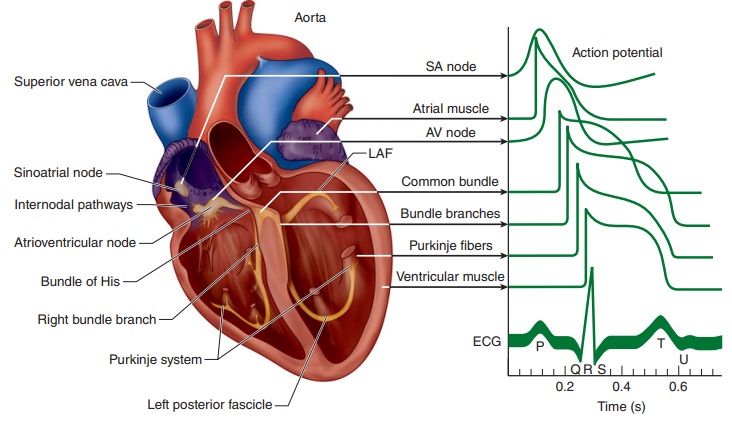
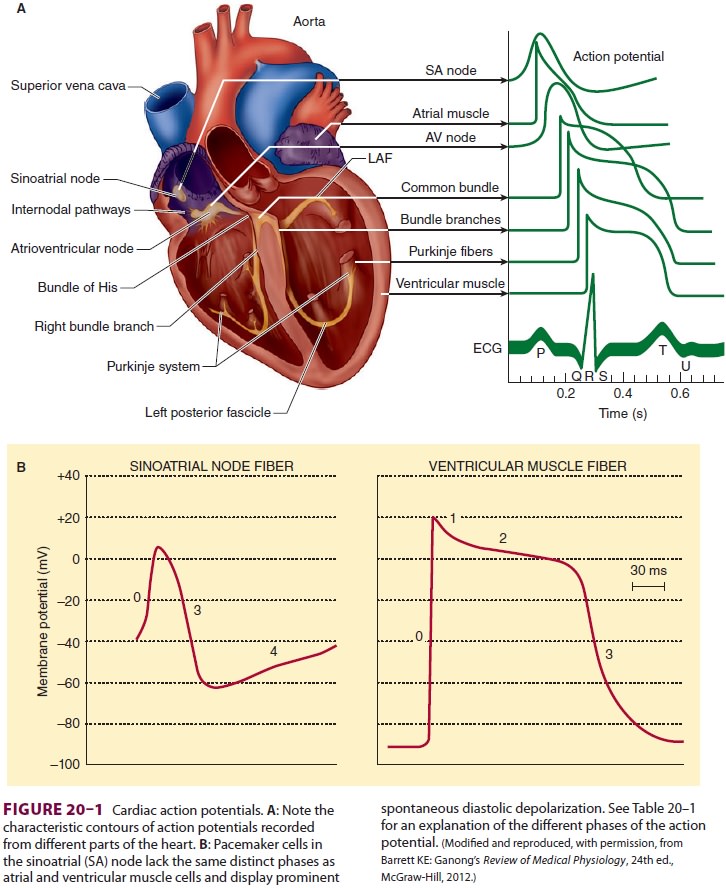
CARDIAC ACTION POTENTIALS
At
rest, the myocardial cell membrane is nominally permeable to K+,
but is relatively impermeable to Na+. A membrane-bound Na+–K+-adenosine
tri-phosphatase (ATPase) concentrates K + intracellu-larly in exchange for
extrusion of Na+
out of the cell. Intracellular Na+
concentration is kept low, whereas intracellular K +
concentration is kept high relative to the extracellular space. The relative
impermeability of the membrane to calcium also maintains a high extracellular
to cytoplasmic calcium gradient. Movement of K+ out of the cell and down its
con-centration gradient results in a net loss of positive charges from inside
the cell. An electrical poten-tial is established across the cell membrane,
with the inside of the cell negative with respect to the extracellular
environment, because anions do not accompany K+. Thus, the resting membrane
poten-tial represents the balance between two opposing forces: the movement of
K+
down its concentration gradient and the electrical attraction of the
nega-tively charged intracellular space for the positively charged potassium
ions.
The
normal ventricular cell resting membrane potential is –80 to –90 mV. As with
other excitable tissues (nerve and skeletal muscle), when the cell membrane
potential becomes less negative and reaches a threshold value, a characteristic
action potential (depolarization) develops ( Figure 20–1 and Table 20–1).
The action potential transiently raises the membrane potential of the
myocardial cell to +20
mV. In contrast to action potentials in axons, the spike in cardiac action
potentials isfollowed by a plateau phase that lasts 0.2–0.3 sec. Whereas the
action potential for skeletal muscle and nerves is due to the abrupt opening of
voltage-gated sodium channels in the cell membrane, in cardiac muscle, it is
initiated by voltage-gated sodium chan-nels (the spike) and maintained by
voltage-gated cal-cium channels (the plateau). Depolarization is also
accompanied by a transient decrease in potassium permeability. Subsequent
restoration of normal potassium permeability and termination of sodium and
calcium channel permeability eventually restores the membrane potential to its
resting value.
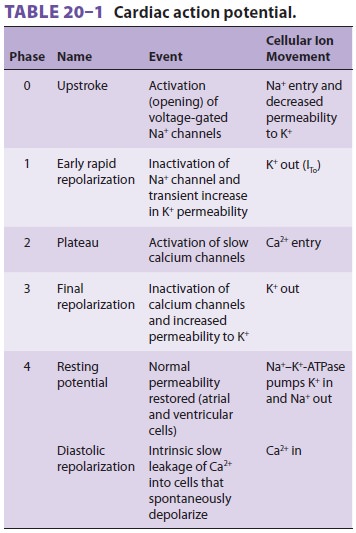
Following
depolarization, the cells are typically refractory to subsequent normal
depolarizing stim-uli until “phase 4.” The effective refractory period is the
minimum interval between two depolarizing impulses that will propagate. In
fast-conducting myocardial cells, this period is generally closely cor-related
with the duration of the action potential. In contrast, the effective
refractory period in more slowly conducting myocardial cells can outlast the
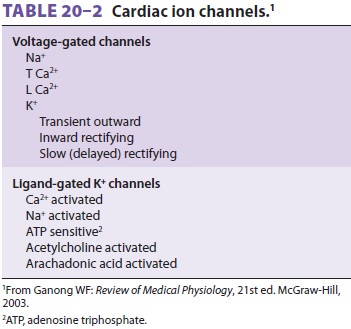
duration of the action potential. Table 20–2 lists some of the multiple types
ofion channels in cardiac muscle membrane. Some are activated by a change in
cell membrane voltage, whereas others open only when bound by ligands. T-type
(transient) voltage-gated calcium channels play a role in phase 0 of
depolarization. During the plateau phase (phase 2), Ca2+
inflow occurs through slow L-type (long-lasting), voltage-gated calcium
channels. Three major types of potassium channels are responsible for repolarization.
The first results in a transient outward K + current (ITo), the second is responsible for a short rectifying
current (IKr), and the
third produces a slowly acting rectifying current (IKs) that helps to restore the cell membrane poten-tial
to its resting value.
Related Topics