Chapter: Clinical Anesthesiology: Anesthetic Management: Cardiovascular Physiology & Anesthesia
Anatomy & Physiology of the Coronary Circulation
ANATOMY & PHYSIOLOGY OF THE CORONARY CIRCULATION
1. Anatomy
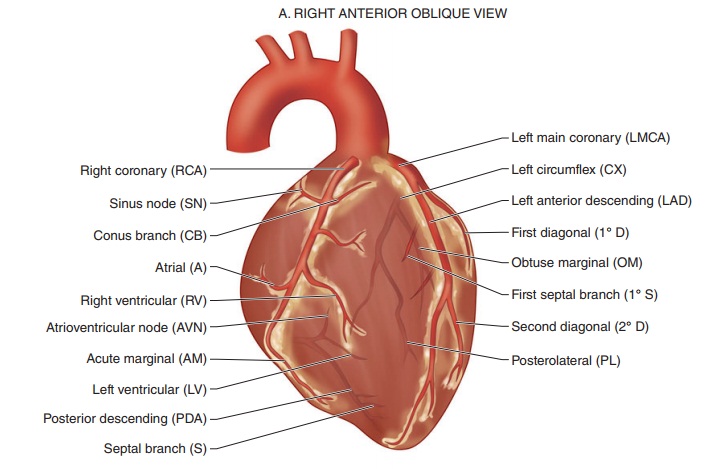
Myocardial blood supply is derived entirely from the right and left coronary arteries (Figure 20–10). Blood flows from epicardial to endocardial vessels. After perfusing the myocardium, blood returns to the right atrium via the coronary sinus and the ante-rior cardiac veins. A small amount of blood returns directly into the chambers of the heart by way of the thebesian veins.
The right coronary artery (RCA) normally sup-plies the right atrium, most of the right ventricle, and a variable portion of the left ventricle (inferior wall). In 85% of persons, the RCA gives rise to the posterior descending artery (PDA), which supplies the superior–posterior interventricular septum and inferior wall—a right dominant circulation; in the remaining 15% of persons, the PDA is a branch of the left coronary artery—a left dominant circulation.
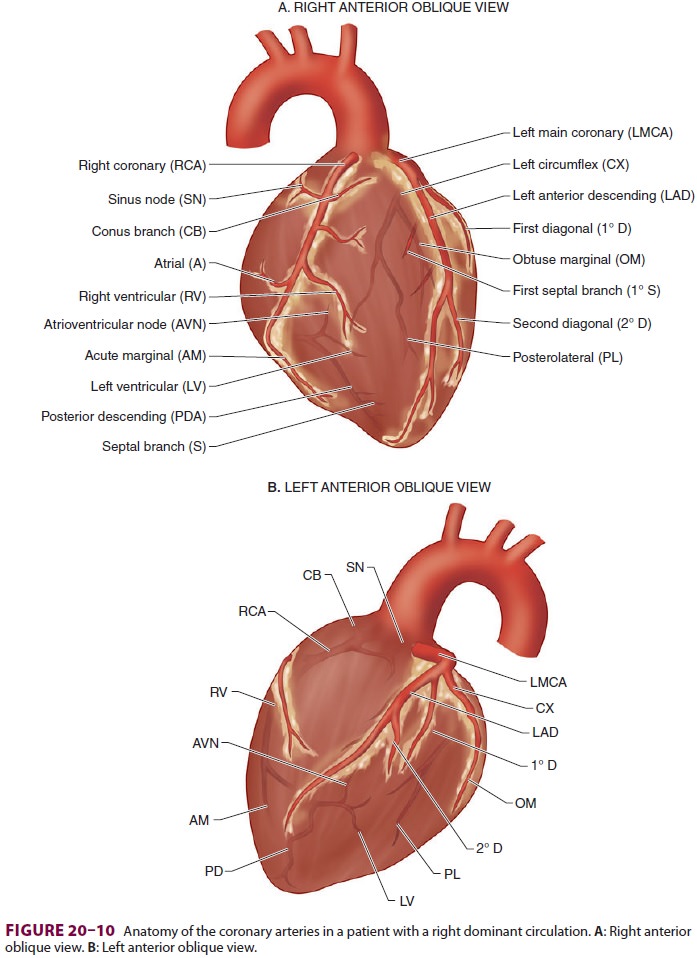
The left coronary artery normally supplies the left atrium and most of the interventricular septum and left ventricle (septal, anterior, and lateral walls). After a short course, the left main coronary artery bifurcates into the left anterior descending artery (LAD) and the circumflex artery (CX); the LAD supplies the septum and anterior wall and the CX supplies the lateral wall. In a left dominant circula-tion, the CX wraps around the AV groove and con-tinues down as the PDA to also supply most of the posterior septum and inferior wall.
The arterial supply to the SA node may be derived from either the RCA (60% of individuals)or the LAD (the remaining 40%). The AV node is usually supplied by the RCA (85% to 90%) or, less frequently, by the CX (10% to 15%); the bundle of His has a dual blood supply derived from the PDA and LAD. The anterior papillary muscle of the mitral valve also has a dual blood supply that is fed by diag-onal branches of the LAD and marginal branches of the CX. In contrast, the posterior papillary of the mitral valve is usually supplied only by the PDA and is therefore much more vulnerable to ischemic dysfunction.
2. Determinants of Coronary Perfusion
Coronary perfusion is unique in that it is inter-mittent rather than continuous, as it is in other organs. During contraction, intramyocardial pres-sures in the left ventricle approach systemic arterial pressure. The force of left ventricular contraction almost completely occludes the intramyocardial part of the coronary arteries; in fact, blood flow may transiently reverse in epicardial vessels. Even during the latter part of diastole, left ventricular pressure eventually exceeds venous (right atrial) pressure. Thus, coronary perfusion pressure isusually determined by the difference between aortic pressure and ventricular pressure, and theleft ventricle is perfused almost entirely during diastole. In contrast, the right ventricle is perfused during both systole and diastole ( Figure 20–11). Moreover, as a determinant of myocardial blood flow, arterial diastolic pressure is more important than MAP:

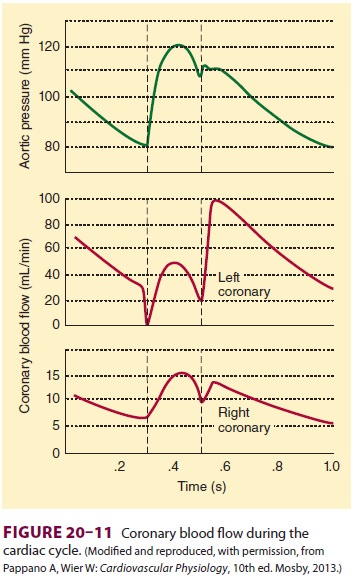
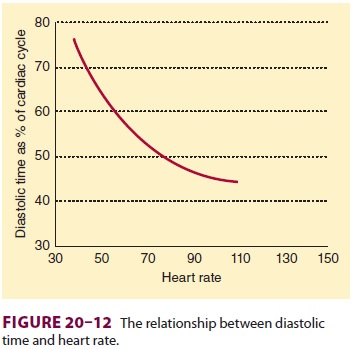
Decreases in aortic pressure or increases in ven-tricular end-diastolic pressure can reduce coronary perfusion pressure. Increases in heart rate also decrease coronary perfusion because of the dispro-portionately greater reduction in diastolic time as heart rate increases (Figure 20–12). Because it is subjected to the greatest intramural pressures during systole, the endocardium tends to be most vulnerable to ischemia during decreases in coronary perfusion pressure.
Control of Coronary Blood Flow
Coronary blood flow normally parallels myocar-dial metabolic demand. In the average adult man, coronary blood flow is approximately 250 mL/min at rest. The myocardium regulates its own blood flow closely between perfusion pressures of 50 and 120 mm Hg. Beyond this range, blood flow becomes increasingly pressure dependent.
Under normal conditions, changes in blood flow are entirely due to variations in coronary arterial tone (resistance) in response to metabolic demand. Hypoxia—either directly, or indirectly through the release of adenosine—causes coronary vasodilation. Autonomic influences are generally weak. Both α1- and β2-adrenergic receptors are present in the coronaryarteries. The α1-receptors are primarily located on larger epicardial vessels, whereas the β2-receptors are mainly found on the smaller intramuscular and subendocardial vessels. Sympathetic stimulation generally increases myo-cardial blood flow because of an increase in meta-bolic demand and a predominance ofβ2-receptor activation. Parasympathetic effects on the coro-nary vasculature are generally minor and weakly vasodilatory.
3. Myocardial Oxygen Balance
Myocardial oxygen demand is usually the most important determinant of myocardial blood flow. Relative contributions to oxygen requirements include basal requirements (20%), electrical activ-ity (1%), volume work (15%), and pressure work (64%). The myocardium usually extracts 65% of the oxygen in arterial blood, compared with 25% in most other tissues. Coronary sinus oxygen satu-ration is usually 30%. Therefore, the myocardium (unlike other tissues) cannot compensate for reduc-tions in blood flow by extracting more oxygen from hemoglobin. Any increases in myocardial metabolic demand must be met by an increase in coronary blood flow. Table 20–6 lists the most important fac-tors in myocardial oxygen demand and supply. Note that the heart rate and, to a lesser extent, ventricular

end-diastolic pressure are important determinants of both supply and demand.
Related Topics