Chapter: Introduction to Human Nutrition: The Vitamins
Assessment of vitamin B6 status
Assessment of vitamin B6 status
Fasting plasma total vitamin B6 (measured microbio-logically), or more specifically pyridoxal phosphate, is widely used as an index of vitamin B6 nutritional status. Despite the fall in plasma pyridoxal phosphate in pregnancy, which has been widely interpreted as indicating vitamin B6 depletion or an increased requirement, the plasma concentration of pyridoxal phosphate plus pyridoxal is unchanged. This suggests that determination of plasma pyridoxal phosphate alone may not be a reliable index of vitamin B6 nutri-tional status.
About half of the normal dietary intake of vitamin B6 is excreted as 4-pyridoxic acid. Urinary excretion of 4-pyridoxic acid will largely reflect the recent intake of the vitamin rather than the underlying nutritional status.
Coenzyme saturation of transaminases
The most widely used method of assessing vitamin B6 status is by the activation of erythrocyte transami-nases by pyridoxal phosphate added in vitro. An acti-vation coefficient for alanine transaminase >1.25, or for aspartate transaminase >1.8, is considered to indi-cate deficiency.
The tryptophan load test
The tryptophan load test for vitamin B6 nutritional status (the ability to metabolize a test dose of trypto-phan) is one of the oldest metabolic tests for func-tional vitamin nutritional status. It was developed as a result of observation of the excretion of an abnor-mal colored compound, later identified as the trypto-phan metabolite xanthurenic acid, in the urine of deficient animals.
Kynureninase (see Figure 8.12) is a pyridoxal phos-phate-dependent enzyme, and its activity falls mark-edly in vitamin B6 deficiency, at least partly because it undergoes a slow mechanism-dependent inactivation that leaves catalytically inactive pyridoxamine phos-phate at the active site of the enzyme. The enzyme can only be reactivated if there is an adequate supply of pyridoxal phosphate. This means that in vitamin B6 deficiency there is a considerable accumulation of both hydroxykynurenine and kynurenine, sufficient to permit greater metabolic flux than usual through kynurenine transaminase, resulting in increased for-mation of kynurenic and xanthurenic acids.
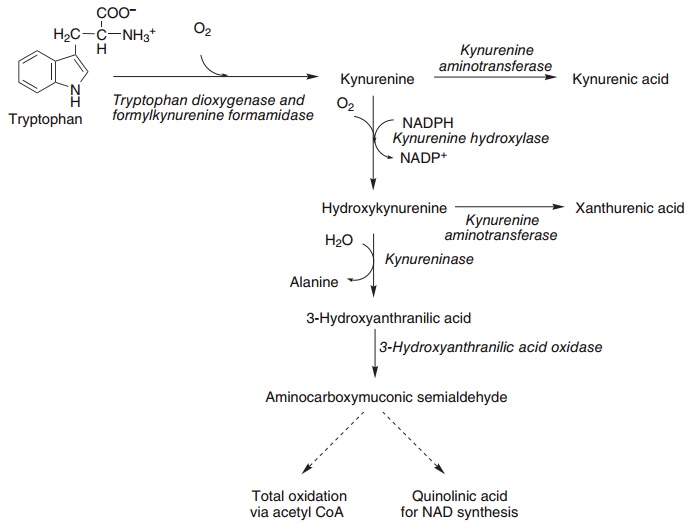
Figure 8.12 Oxidative pathway of tryptophan: the basis of the tryptophan load test. Tryptophan dioxygenase (EC 1.13.11.11), formylkynurenine formamidase (EC 3.5.1.9), kynurenine hydroxylase (EC 1.14.13.9), kynureninase (EC 3.7.1.3).
Xanthurenic and kynurenic acids, and kynurenine and hydroxykynurenine, are easy to measure in urine, so the tryptophan load test [the ability to metabolize a test dose of 2–5 g (150–380 μmol/kg body weight) of tryptophan] has been widely adopted as a con-venient and very sensitive index of vitamin B6 nutritional status. However, because glucocorticoid hormones increase tryptophan dioxygenase activity, abnormal results of the tryptophan load test must be regarded with caution, and cannot necessarily be interpreted as indicating vitamin B6 deficiency. Increased entry of tryptophan into the pathway will overwhelm the capacity of kynureninase, leading to increased formation of xanthurenic and kynurenic acids. Similarly, estrogen metabolites inhibit kyn-ureninase, leading to results that have been misinter-preted as vitamin B6 deficiency.
The methionine load test
The metabolism of methionine includes two pyri-doxal phosphate-dependent steps: cystathionine synthetase and cystathionase (see Figure 8.16). Cystathionase activity falls markedly in vitamin B6 deficiency, and as a result there is an increase in the urinary excretion of homocysteine and cystathionine, both after a loading dose of methionine and under
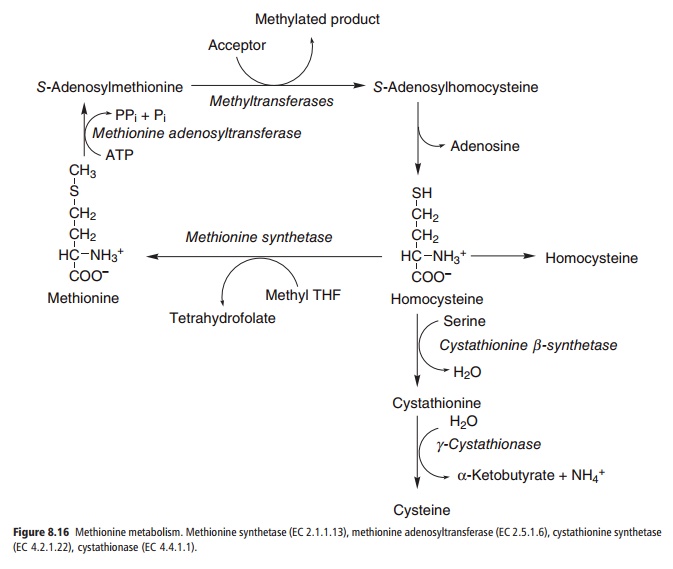
basal conditions. However, as discussed below, homo-cysteine metabolism is more affected by folate status than by vitamin B6 status and, like the tryptophan load test, the methionine load test is probably not reliable as an index of vitamin B6 status in field studies.
Related Topics