Chapter: Medical Physiology: Kidney Diseases and Diuretics
Treatment of Renal Failure by Dialysis with an Artificial Kidney
Treatment of Renal Failure by Dialysis with an Artificial Kidney
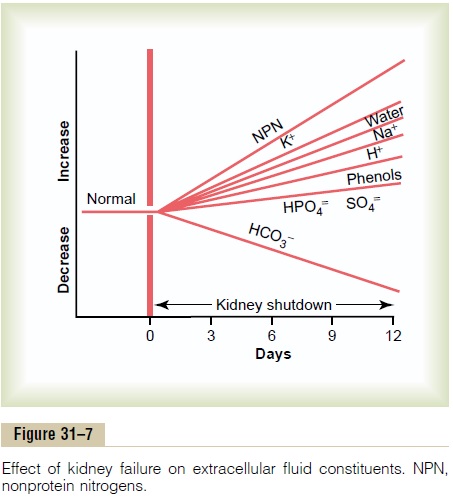
Severe loss of kidney function, either acutely or chron-ically, is a threat to life and requires removal of toxic waste products and restoration of body fluid volume and composition toward normal. This can be accom-plished by dialysis with an artificial kidney. In certain types of acute renal failure, an artificial kidney may be used to tide the patient over until the kidneys resume their function. If the loss of kidney function is irre-versible, it is necessary to perform dialysis chronically to maintain life. In the United States alone, nearly 300,000 people with irreversible renal failure or even total kidney removal are being maintained by dialysis with artificial kidneys. Because dialysis cannot maintain completely normal body fluid composition and cannot replace all the multiple functions performed by the kidneys, the health of patients maintained on artificial kidneys usually remains significantly impaired. A better treatment for permanent loss of kidney function is to restore functional kidney tissue by means of a kidney transplant.
Basic Principles of Dialysis. The basic principle of the arti-ficial kidney is to pass blood through minute blood channels bounded by a thin membrane. On the other side of the membrane is a dialyzing fluid into which unwanted substances in the blood pass by diffusion.
Figure 31–8 shows the components of one type of arti-ficial kidney in which blood flows continually between two thin membranes of cellophane; outside the mem-brane is a dialyzing fluid. The cellophane is porous enough to allow the constituents of the plasma, except the plasma proteins, to diffuse in both directions—from plasma into the dialyzing fluid or from the dialyzing
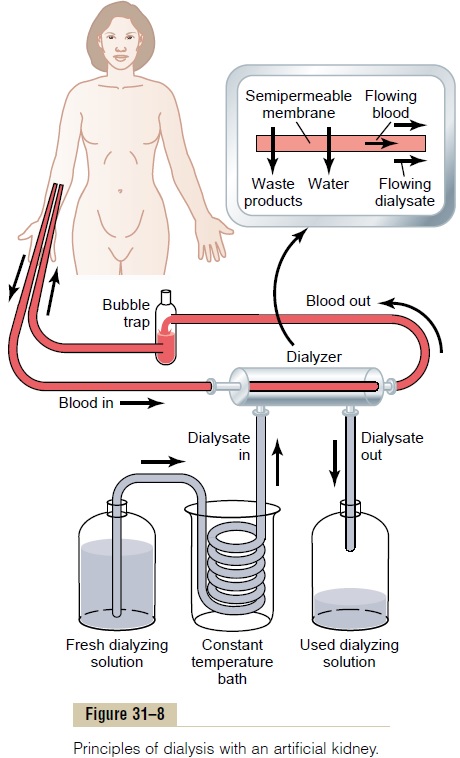
fluid back into the plasma. If the concentration of a sub-stance is greater in the plasma than in the dialyzing fluid, there will be a nettransfer of the substance from the plasma into the dialyzing fluid.
The rate of movement of solute across the dialyzing membrane depends on (1) the concentration gradient of the solute between the two solutions, (2) the per-meability of the membrane to the solute, (3) the surface area of the membrane, and (4) the length of time that the blood and fluid remain in contact with the membrane.
Thus, the maximum rate of solute transfer occurs ini-tially when the concentration gradient is greatest (when dialysis is begun) and slows down as the concentration gradient is dissipated. In a flowing system, as is the case with “hemodialysis,” in which blood and dialysate fluid flow through the artificial kidney, the dissipation of the concentration gradient can be reduced and diffusion of solute across the membrane can be optimized by increasing the flow rate of the blood, the dialyzing fluid, or both.
In normal operation of the artificial kidney, blood flows continually or intermittently back into the vein. The total amount of blood in the artificial kidney at any one time is usually less than 500 milliliters, the rate of flow may be several hundred milliliters per minute, and the total diffusion surface area is between 0.6 and 2.5 square meters. To prevent coagulation of the blood in the artificial kidney, a small amount of heparin is infused into the blood as it enters the artificial kidney. In addi-tion to diffusion of solutes, mass transfer of solutes and water can be produced by applying a hydrostatic pres-sure to force the fluid and solutes across the membranes of the dialyzer; such filtration is called bulk flow.
Dialyzing Fluid. Table 31–7 compares the constituents ina typical dialyzing fluid with those in normal plasma and uremic plasma. Note that the concentrations of ions and other substances in dialyzing fluid are not the same as the concentrations in normal plasma or in uremic plasma. Instead, they are adjusted to levels that are needed to cause appropriate movement of water and solutes through the membrane during dialysis.
Note that there is no phosphate, urea, urate, sulfate, or creatinine in the dialyzing fluid; however, these are present in high concentrations in the uremic blood. Therefore, when a uremic patient is dialyzed, these sub-stances are lost in large quantities into the dialyzing fluid.
The effectiveness of the artificial kidney can be expressed in terms of the amount of plasma that is cleared of different substances each minute, which, is the primary means for expressing the functional effectiveness of the kidneys themselves to rid the body of unwanted substances. Most artificial kidneys can clear urea from the plasma at a rate of 100 to 225 ml/min, which shows that at least for the excretion of urea, the artificial kidney can func-tion about twice as rapidly as two normal kidneys together, whose urea clearance is only 70 ml/min. Yet the artificial kidney is used for only 4 to 6 hours per day, three times a week. Therefore, the overall plasma clear-ance is still considerably limited when the artificial kidney replaces the normal kidneys. Also, it is important to keep in mind that the artificial kidney cannot replace some of the other functions of the kidneys, such as secretion of erythropoietin, which is necessary for red blood cell production.
Related Topics