Chapter: Genetics and Molecular Biology: RNA Polymerase and RNA Initiation
Three RNA Polymerases in Eukaryotic Cells
Three RNA Polymerases in Eukaryotic Cells
Investigation of eukaryotic cells shows that they
do contain more than one type of RNA polymerase. The typical protein
fractionation schemes used by biochemists to purify proteins yield three
distinct types of RNA polymerase from a variety of higher cells. These three
species are called RNA polymerases I, II, and III for the order in which they
elute from an ion exchange column during their purification.
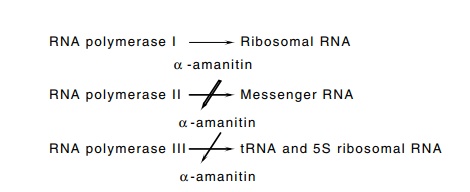
Figure
4.7 Three eukaryotic RNA polymerases,
their sensitivities toα-amani-tin,
and the products they synthesize.
RNA polymerase I synthesizes ribosomal RNA. It is
found in the nucleolus, an organelle in which ribosomal RNA is synthesized.
Further confirmation of this conclusion is the finding that only purified RNA
polymerase I is capable of correctly initiating transcription of ribosomal RNA in vitro. A simple experiment
demonstrating this is to use DNA containing ribosomal RNA genes as a template
and then to assay the strand-specificity of the resulting product. RNA polymerase
I synthe-sizes RNA predominantly from the correct strand of DNA, whereas RNA
polymerases II and III do not. RNA polymerase II is the polymerase responsible
for most synthesis of messenger RNA. In
vitro experiments show that, of the three, this polymerase is the most
sensitive to a toxin from mushrooms, α-amanitin
(Fig. 4.7). The addition of low concentrations of α-amanitin to cells or to isolated nuclei blocks additional synthesis of
just messenger RNA. Further, α-amanitin-resistant
cells possess a toxin-resistant RNA polym-erase II.
RNA polymerase III is less sensitive to α-amanitin than is RNA polymerase II, but it is
sufficiently sensitive that its in vivo
function can be probed. The sensitivity profile for synthesis of tRNA and 5S
RNA parallels the sensitivity of RNA polymerase III. In vitro transcription experiments with purified polymerase III
also show that this enzyme synthesizes tRNA and 5S ribosomal RNA as well as
some of the RNAs found in spliceosomes as discussed.
Related Topics