Chapter: Plant Biochemistry: A leaf cell consists of several metabolic compartments
The endoplasmic reticulum and golgi apparatus form a network for the distribution of biosynthesis products
The endoplasmic reticulum and golgi apparatus form a network for the distribution of biosynthesis products
In an electron micrograph, the endoplasmic reticulum (ER) appears as a labyrinth traversing the cell (Fig. 1.15). Two structural types of ER can be differentiated: the rough and the smooth forms. The rough ER consists of flattened sacs that are sometimes arranged in loose stacks of which the outer side of the membranes is occupied by ribosomes. The smooth ER con-sists primarily of branched tubes without ribosomes. Despite these mor-phological differences, the rough ER and the smooth ER are constituents of a continuous membrane system.

The presence of ribosomes on the outer surface of the ER is tempo-rary. Ribosomes are attached to the ER membrane only when the protein that they synthesize is destined for the ER itself, for the vacuoles, or for export from the cell. These proteins contain an amino acid sequence (signal sequence) that causes the peptide chain in the initial phase of its synthesis to enter the lumen of the ER. A snapshot of the ribosome com-plement of the ER only shows those ribosomes that at the moment of tissue fixation are involved in the synthesis of proteins destined for import into the ER lumen. Membranes of the ER are also the site of membrane lipid synthesis, for which the necessary fatty acids are provided by the plastids.
In seeds and other tissues, oil bodies (also called oleosomes) are present. These are derived from the ER membrane. The oil bodies store triacylglyc-erides and are of great economic importance for oil fruits, such as rape seed or olives. The oil bodies are only enclosed by half of a biomembrane (mono layer), of which the hydrophobic fatty acid residues of the membrane lipids project into the oil and the hydrophilic parts of the lipid layer protrude into the cytosol.
In addition, the ER is a suitable storage site for the production of trans-gene proteins in genetically engineered plants. Native as well as transgene proteins are equipped with a signal sequence and the amino terminal ER-retention signal KDEL (Lys Asp Glu Leu). The ER of leaves is capable of accumulating large amounts of such extraneous proteins (up to 2.5 to 5% of the total leaf protein) without affecting the function of the ER. In the ER lumen, proteins are often modified by N-glycosylation (attachment of hexose chains to amino acid residues, e.g., asparagin).
The transport of proteins into the vacuoles proceeds in different ways. There exists a direct transport via vesicles between the ER and the vacuoles. Most proteins, however, are at first channeled via vesicles to the cis-side of the Golgi apparatus (Fig. 1.16), and only after having been processed in the Golgi apparatus are further transferred into the vacuoles or are excreted from the cell as secretory proteins. Two mechanisms for transporting pro-teins through the Golgi apparatus are under discussion. (1) According to thevesicle shuttle model (Fig. 1.16), the proteins pass through the differ-ent cisternae by enbudding and vesicle transfer, while each cisterna has its fixed position. (2) According to the cisternae progression model, cisternae are constantly being newly formed by vesicle fusion at the cis-side, and they then decompose to vesicles at the trans-side. Present results show that both systems probably function in parallel. The budding of the protein loaded vesicles from the Golgi apparatus occurs at certain regions of the ER mem-brane called ERES (ER export sites). The vesicles are covered at the outer surface of the surrounding membrane with a coat protein (COP II). The fusion of the vesicles with the membrane of the Golgi apparatus is facili-tated by so-called SNARE-proteins (soluble N-ethylmaleinimide sensitive attachment protein receptors). For the backflow of the emptied vesicles to the ER, the vesicles are covered with another coat protein (COP I) and the fusion with the ER again requires a SNARE protein. In Arabidopsis alto-gether 15 genes encoding for SNARE proteins have been identified.
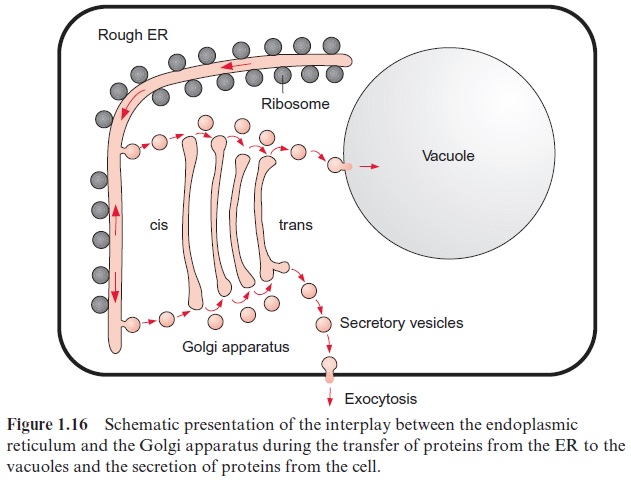
The Golgi apparatus was discovered in 1898 by the Italian Camillo Golgi by using a light microscope. The Golgi system consists of up to 20 curved discs arranged in parallel, the so-called Golgi cisternae or dic-tyosomes, which are surrounded by smooth membranes (not occupied by ribosomes) (Fig. 1.17). At both sides of the discs, vesicles of various sizes can be seen to bud off. The Golgi apparatus consists of the cis-compart-ment, the middle compartment, and the trans-compartment. During transport through the Golgi apparatus, proteins are often modified by O-glycosylation (attachment of hexose chains to serine and threonine residues).

In the Golgi apparatus, proteins are selected either to be removed from the cell by exocytosis (secretion) or to be transferred to lytic vacuoles or to storage vacuoles . Signal sequences of proteins act as sort-ing signals to direct proteins into the vacuolar compartment; the proteins destined for the lytic vacuoles are transferred in clathrin-coated vesicles.
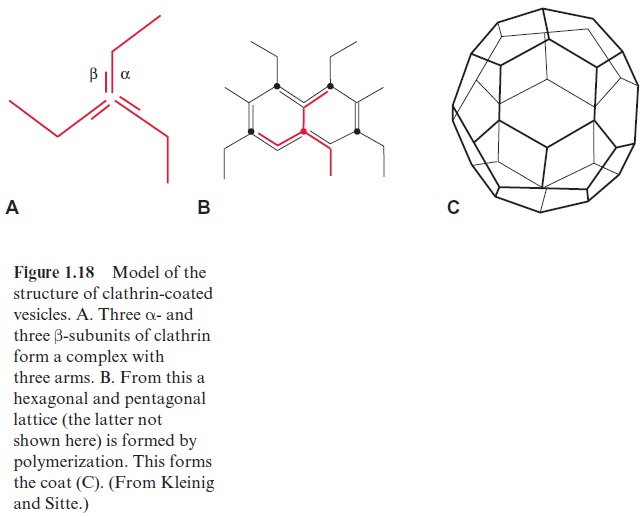
Clathrin is a protein consisting of two different subunits (α-UE 180,000 Dalton, β-UE 35,000 to 40,000 Dalton). Three α- and three β-subunits form a complex with three arms (triskelion), which polymerizes to a hexagonal latticed structure surrounding the vesicle (Fig. 1.18). The transport into the storage vacuoles proceeds via other vesicles without clathrin. Secretion proteins, containing only the signal sequence for entry into the ER, reach the plasma membrane via secretion vesicles without a protein coat and are secreted by exocytosis.
The ER membrane, the membranes of the Golgi apparatus (derived from the ER), the transfer vesicles, and the nuclear envelope are collectively called the endomembrane system.
Related Topics