Chapter: Plant Biochemistry: A leaf cell consists of several metabolic compartments
Functionally intact cell organelles can be isolated from plant cells
Functionally intact cell organelles can be isolated from plant cells
In order to isolate cell organelles, the cell has to be disrupted only to such an extent that its intact organelles are released into the isolation medium, resulting in a cell homogenate. To prevent the liberated organelles from swelling and disruption, the isolation medium must be isotonic. The pres-ence of an osmotic compound (e.g., sucrose) generates an osmotic pressure in the medium, which should correspond to the osmotic pressure of the aqueous phase within the organelle. Media containing 0.3 mol/L sucrose or sorbitol usually are used for such cell homogenizations.
Figure 1.19 shows the protocol of the isolation scheme for chloroplasts. Small leaf pieces are homogenized by cutting them up within seconds using blades rotating at high speed, such as in a food mixer. It is important that the homogenization time is short; otherwise the cell organelles released into the isolation medium would also be destroyed. Such homogenization is only applicable for leaves that have soft cell walls, e.g., spinach. In the case of leaves with more rigid cell walls (e.g., cereal plants), protoplasts are first prepared from leaf pieces. These protoplasts are then ruptured by forcing the protoplast suspension through a net with a mesh smaller than the size of the protoplasts.
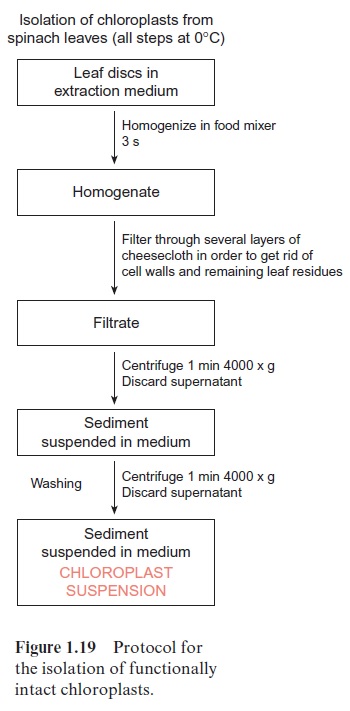
The desired organelles can be separated and purified from the rest of the cell homogenate by differential or density gradient centrifugation. In the case of differential centrifugation, the homogenate is suspended in a medium with a density much lower than that of the cell organelles. In the gravitational field of the centrifuge, the sedimentation velocity of the par-ticles depends primarily on the particle size (the large particles sediment faster than the small particles). As shown in Figure 1.19, taking the isola-tion of chloroplasts as an example, relatively pure organelle preparations can be obtained within a short time by a sequence of centrifugation steps at increasing speeds.
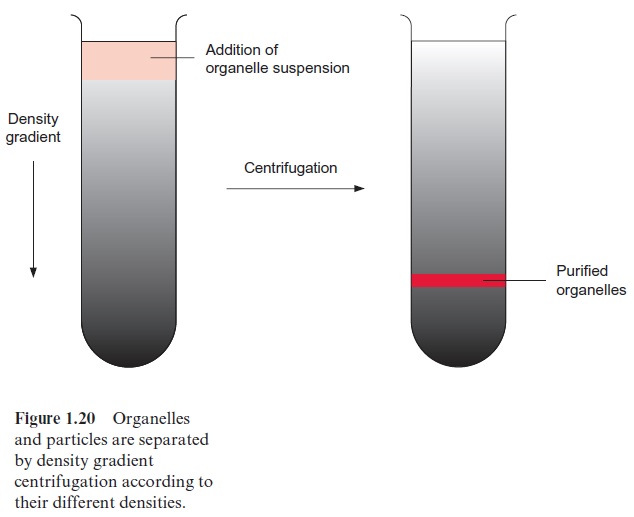
In the case of density gradient centrifugation (Fig. 1.20), the organelles are separated according to their density. Media of differing densities are assembled in a centrifuge tube so that the density increases from top to bot-tom. To prevent alterations of the osmolarity of the medium, heavy mac-romolecules (e.g., Percoll = silica gel) are used to achieve a high density. The cell homogenate is layered on the density gradient prepared in the cen-trifuge tube and centrifuged until all the particles of the homogenate have reached their zone of equal density in the gradient. As this density gradient centrifugation requires high centrifugation speed and long running times, it is often used as the final purification step after preliminary separation by differential centrifugation.
By using these techniques it is possible to obtain functionally intact chloroplasts, mitochondria, peroxisomes, and vacuoles of high purity with the option to study their metabolic properties thereafter in the test tube.
Related Topics