Chapter: Biotechnology Applying the Genetic Revolution: Molecular Biology of Cancer
The Ras Oncogene-Hyperactive Protein
THE RAS ONCOGENE—HYPERACTIVE PROTEIN
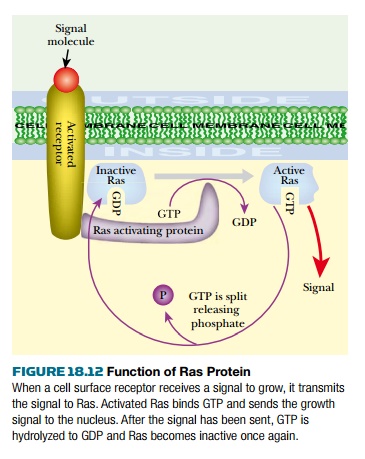
Ras protein transmits signals concerning cell division in humans, flies, and even yeastcells (Fig. 18.12). Growth signals are received from outside the cell by receptors at the cell surface. The activated receptor transfers the signal to intracellular Ras protein. After receiving a signal, normal Ras protein binds guanosine triphosphate (GTP) and enters signal-emitting mode. After transmitting a brief pulse of signals, Ras then splits the GTP into guanosine diphosphate (GDP) plus phosphate and relapses into standby mode again. The cancer-causing form of the Ras protein is locked permanently into the signal-emitting mode and never splits the GTP. Therefore it constantly floods the cell with signals urging cell division, even when none is received from outside.
The rasoncogene is the result
of a single base change in the structural region of the gene. This causes an
alteration in a single amino acid in the encoded protein (Fig. 18.13). Most ras mutations alter the amino acid at
position 12; others affect position 13 or 61. Only a few very specific
mutations can create a ras oncogene
from the proto-oncogene. The 3D structure of the Ras protein has been solved by
x-ray crystallography. Those few amino acid residues that are changed by
oncogenic mutations are all directly involved in the binding and splitting of
the GTP. The consequence of hyperactivation of Ras is uncontrolled cell
division and the beginnings of a possible cancer. Mutations of ras are frequently detected and have
been analyzed in detail, in cancers of the lung, colon, pancreas, and thyroid.

Related Topics