Chapter: Biotechnology Applying the Genetic Revolution: Molecular Biology of Cancer
Inherited Susceptibility to Cancer
INHERITED SUSCEPTIBILITY TO CANCER
It
is thought that 5% to 10% of cancers may be largely due to inherited defects.
Many of the genes involved in this are poorly understood, but they may be
divided into three general categories.
First,
as we have already discussed, it is possible to inherit one defective copy of
an anti-oncogene. In this case every somatic cell starts life with one faulty
copy and only a single somatic mutation is needed to completely inactivate the
pair of anti-oncogenes. (Note: Inheriting two defective copies of an
anti-oncogene is normally lethal. Artificially engineered mice that are doubly
negative for such genes generally die before birth.)
Secondly,
mutations in certain special genes affect the rate at which further mutations occur
during cell division. Such genes are known as mutator genes . These include
genes involved in DNA synthesis, such as the genes encoding DNA polymerase.
Some mutator genes are more subtle and are involved in DNA repair. These have
been analyzed in detail in bacteria, although less is known for humans.
Nonetheless, it appears that certain inherited forms of colon cancer are due to
defects in genes involved in mismatch repair.
This,
in turn, increases the rate of mutation in all other genes including the
tumor-suppressor
genes. Defects in mutator gene are generally recessive, like those in other
tumor-suppressor genes.
Inherited
breast cancer falls into this category. Inheriting a single defective copy of
either the BRCA1 or BRCA2 genes (breast cancer A genes)
predisposes women to breast cancer. About 0.5% of U.S. women carry mutations in
BRCA1 , which also predisposes
to cancer of the ovary. As with other tumor-suppressorgenes, the second copy
must mutate during division of somatic cells for a cancer to arise. Individuals
with two defective BRCA alleles
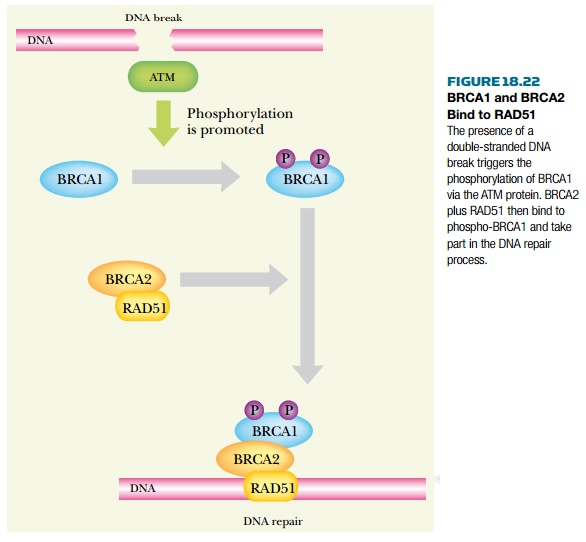
die as embryos. Both BRCA1 and BRCA2 proteins are involved in DNA repair. Both
bind to RAD51, which takes part in mending double-stranded DNA breaks ( Fig.
18.22 ). In addition BRCA1 has a dual role as a transcriptional regulator of
other components needed for DNA repair.
Finally,
there are indirect effects on cancer frequency due to genetic differences
between races or within populations. For example, some skin cancers result from
mutations caused by ultraviolet radiation from the sun. White people,
especially those exposed to high levels of sunshine in the tropics or
Australia, develop skin cancer much more often than darker-skinned people. The
reason is obvious: the more pigment, the less UV radiation penetrating to your
DNA.
Related Topics