Chapter: Medical Surgical Nursing: Respiratory Care Modalities
The Patient Undergoing Thoracic Surgery
The Patient Undergoing Thoracic Surgery
Assessment
and management are particularly important in the patient undergoing thoracic
surgery. Frequently, patients under-going such surgery also have obstructive
pulmonary disease with compromised breathing. Preoperative preparation and
careful postoperative management are crucial for successful patient out-comes
because these patients may have a narrow range between their physical tolerance
for certain activities and their limitations, which, if exceeded, can lead to
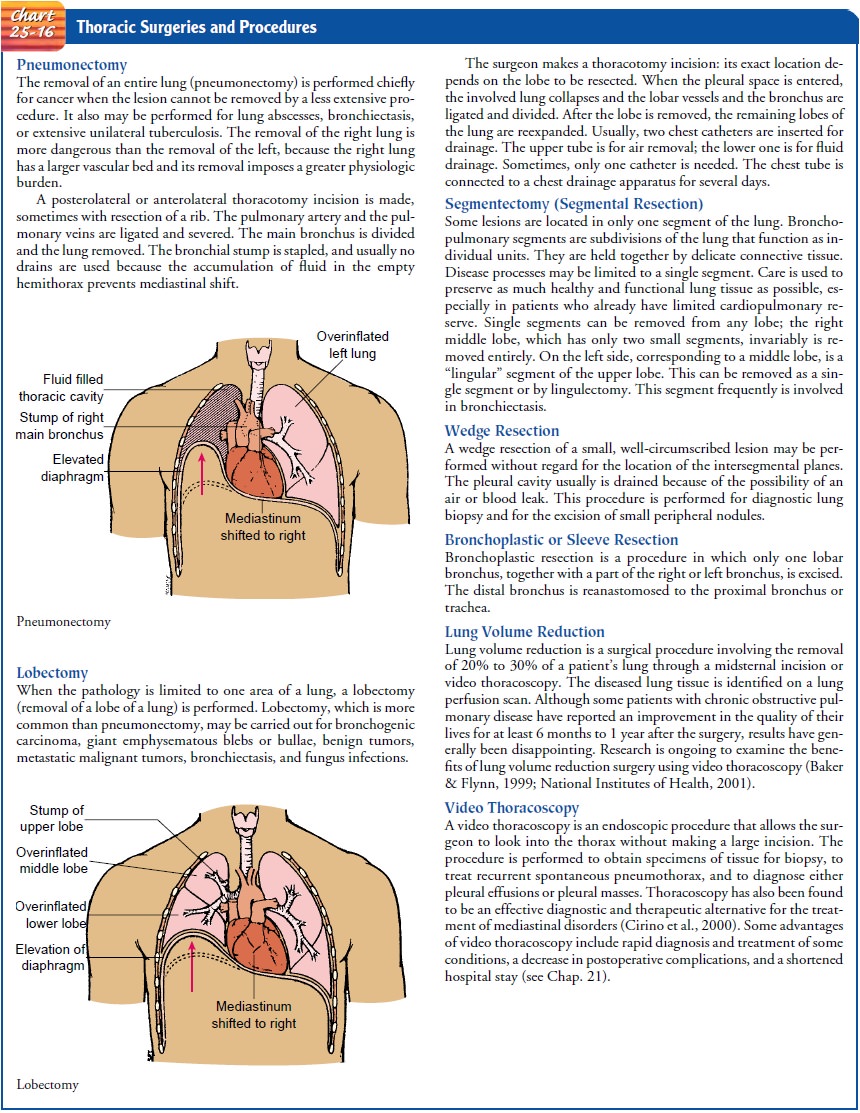
distress. Various types of thoracic surgical procedures are performed to
relieve disease conditions such as lung abscesses, lung cancer, cysts, and
benign tumors (Chart 25-16). An exploratory thoracotomy (creation of a surgi-cal opening into the thoracic
cavity) may be performed to diag-nose lung or chest disease. A biopsy may be
performed in this procedure with a small amount of lung tissue removed for
analy-sis; the chest incision is then closed.
The
objectives of preoperative care for the patient undergoing thoracic surgery are
to ascertain the patient’s functional reserve to determine if the patient can
survive the surgery and to ensure that the patient is in optimal condition for
surgery.
PREOPERATIVE MANAGEMENT
Assessment and Diagnostic Findings
The
nurse performs chest auscultation to assess breath sounds in the different
regions of the lungs. It is important to note if breath sounds are normal,
indicating a free flow of air in and out of the lungs. (In the patient with
emphysema, the breath sounds may be markedly decreased or even absent on
aus-cultation.) The nurse notes crackles and wheezes and assesses for
hyperresonance and decreased diaphragmatic motion. Unilateral diminished breath
sounds and rhonchi can be the result of oc-clusion of the bronchi by mucus
plugs. The nurse assesses for re-tained secretions during auscultation by
asking the patient to cough. It is important to note any signs of rhonchi or
wheezing.
The
patient history and assessment should include the following questions:
·
What signs and symptoms are present
(cough, sputum ex-pectorated [amount and color], hemoptysis, chest pain,
dyspnea)?
·
If there is a smoking history, how
long has the patient smoked? Does the patient smoke currently? How many packs a
day?
·
What is the patient’s
cardiopulmonary tolerance while rest-ing, eating, bathing, and walking?
·
What is the patient’s breathing
pattern? How much exer-tion is required to produce dyspnea?
·
Does the patient need to sleep in an
upright position or with more than two pillows?
·
What is the patient’s physiologic
status (eg, general appear-ance, mental alertness, behavior, nutritional
status)?
·
What other medical conditions exist
(eg, allergies, cardiac disorders, diabetes)?
A
number of tests are performed to determine the patient’s preoperative status
and to assess the patient’s physical assets and limitations. Many patients are
seen by their surgeons in the of-fice, and many tests and examinations are
performed on an out-patient basis. The decision to perform any pulmonary
resection is based on the patient’s cardiovascular status and pulmonary
reserve. Pulmonary function studies (especially lung volume and vital capacity)
are performed to determine whether the planned resection will leave sufficient
functioning lung tissue. Arterial blood gas values are assessed to provide a
more com-plete picture of the functional capacity of the lung. Exercise
tol-erance tests are useful to determine if the patient who is a candidate for
pneumonectomy can tolerate removal of one of the lungs.
Preoperative
studies are performed to provide a baseline for comparison during the
postoperative period and to detect any un-suspected abnormalities. These
studies may include a broncho-scopic examination (a lighted scope is inserted
into the airways to examine the bronchi), chest x-ray, electrocardiogram (for
arte-riosclerotic heart disease, conduction defects), nutritional assess-ment,
determination of blood urea nitrogen and serum creatinine (renal function),
glucose tolerance or blood glucose (diabetes), as-sessment of serum electrolytes
and protein levels, blood volume determinations, and complete blood cell count.
PREOPERATIVE NURSING MANAGEMENT
Improving Airway Clearance
The
underlying lung condition often is associated with increased respiratory
secretions. Before surgery, the airway is cleared of se-cretions to reduce the
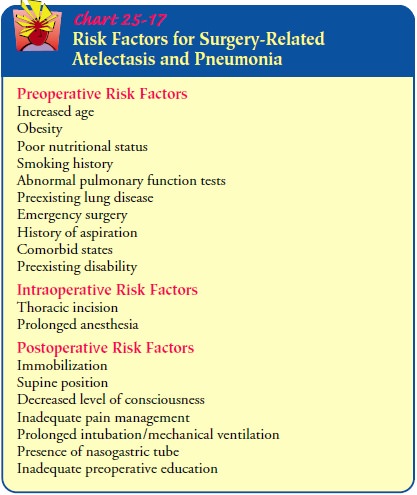
possibility of postoperative atelectasis or in-fection. Risk factors for
postoperative atelectasis and pneumonia are listed in Chart 25-17. Strategies
to reduce the risk for atelec-tasis and infection include humidification,
postural drainage, and chest percussion after bronchodilators are administered,
if pre-scribed. The nurse estimates the volume of sputum if the patient
expectorates large amounts of secretions. Such measurements are carried out to
determine if and when the amount decreases. An-tibiotics are administered as
prescribed for infection, which may be causing the excessive secretions.
Teaching the Patient
Increasingly,
patients are admitted on the day of surgery, which does not provide much time
for the acute care nurse to talk with the patient. Nurses in all settings must
take an active role in ed-ucating the patient and relieving anxiety. The nurse
informs the patient what to expect, from administration of anesthesia to
thoracotomy and the likely use of chest tubes and a drainage system in the
postoperative period. The patient is also informed about the usual
postoperative administration of oxygen to fa-cilitate breathing, and the
possible use of a ventilator. It is es-sential to explain the importance of
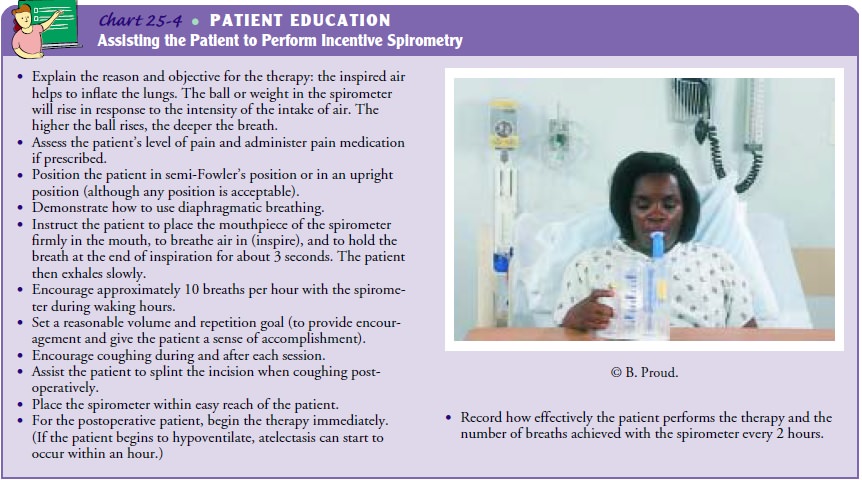
frequent turning to promote drainage of lung secretions. Instruction in the use
of incentive spirometry begins before surgery to familiarize the pa-tient with
its correct use. The nurse should teach diaphragmatic and pursed-lip breathing,
and the patient should begin practic-ing these techniques (see Chart 25-3,
“Breathing Exercises,” and Chart 25-4, “Assisting the Patient to Perform
Incentive Spirometry”).

Because
a coughing schedule will be necessary in the postop-erative period to promote
the clearance or removal of secretions, the nurse instructs the patient in the
technique of coughing and warns the patient that the coughing routine may be
uncomfort-able. The nurse teaches the patient to splint the incision with the
hands, a pillow, or a folded towel (see Chart 25-5).

Another
technique, “huffing,” may be helpful for the patient with diminished expiratory
flow rates or for the patient who re-fuses to cough because of severe pain.
Huffing is the expulsion of air through an open glottis. This type of forced
expiration technique (FET) stimulates pulmonary expansion and assists in
alveolar inflation. The nurse instructs the patient as follows:
·
Take a deep diaphragmatic breath and
exhale forcefully against your hand in a quick, distinct pant, or huff.
·
Practice doing small huffs and
progress to one strong huff during exhalation.
Patients
should be informed preoperatively that blood and other fluids may be administered,
oxygen will be administered, and vital signs will be checked often for several
hours after surgery. If a chest tube is needed, the patient should be informed
that it will drain the fluid and air that normally accumulate after chest
surgery. The patient and family are informed that the pa-tient may be admitted
to the intensive care unit for 1 to 2 days after surgery, that the patient may
experience pain at the incision site, and that medication is available to
relieve pain and discom-fort (Finkelmeier, 2000).
Relieving Anxiety
The
nurse listens to the patient to evaluate his or her feelings about the illness
and proposed treatment. The nurse also deter-mines the patient’s motivation to
return to normal or baseline function. The patient may reveal significant
concerns: fear of hemorrhage because of bloody sputum, fear of discomfort from
a chronic cough and chest pain, fear of ventilator dependence, or fear of death
because of dyspnea and the underlying disease (eg, tumor).
The
nurse helps the patient to overcome these fears and to cope with the stress of
surgery by correcting any misconceptions, supporting the patient’s decision to
undergo surgery, reassuring the patient that the incision will “hold,” and
dealing honestly with questions about pain and discomfort and their treatment.
The management and control of pain begin before surgery, when the nurse informs
the patient that many postoperative problems can be overcome by following
certain routines related to deep breathing, coughing, turning, and moving. If
patient-controlled analgesia or epidural analgesia is to be used after surgery,
the nurse instructs the patient in its use.
POSTOPERATIVE MANAGEMENT
After
surgery the vital signs are checked frequently. Oxygen is administered by a
mechanical ventilator, nasal cannula, or mask for as long as necessary. A
reduction in lung capacity requires a period of physiologic adjustment, and
fluids may be given at a low hourly rate to prevent fluid overload and
pulmonary edema. When the patient is conscious and the vital signs have
stabilized, the head of the bed may be elevated 30 to 45 degrees. Careful
positioning of the patient is important. Following pneumonectomy, a patient is
usually turned every hour from the back to the operative side and should not be
completely turned to the unoperated side. This allows the fluid left in the
space to consolidate and prevents the remaining lung and the heart from
shifting (mediastinal shift) toward the operative side. The patient with a
lobectomy may be turned to either side, and a patient with a segmental
resection usually is not turned onto the operative side unless the surgeon
prescribes this position (Finkelmeier, 2000).
Medication
for pain is needed for several days after surgery. Because coughing can be
painful, patients should be taught to splint the chest. Exercises are resumed
early in the postoperative period to facilitate lung ventilation. The nurse
assesses for signs of complications, including cyanosis, dyspnea, and acute
chest pain. These may indicate atelectasis and should be reported im-mediately.
Increased temperature or white blood cell count may indicate an infection, and
pallor and increased pulse may indi-cate internal hemorrhage. Dressings should
be assessed for fresh bleeding.
Mechanical Ventilation
Depending
on the nature of the surgery, the patient’s underly-ing condition, the
intraoperative course, and the depth of anes-thesia, the patient may require
mechanical ventilation after surgery. The physician is responsible for
determining the venti-lator settings and modes, as well as determining the
overall method and pace of weaning. However, the physician, nurse, and
respiratory therapist work together closely to assess the pa-tient’s tolerance
and weaning progress. Early extubation from mechanical ventilation can also
lead to earlier removal of arter-ial lines (Zevola & Maier, 1999).
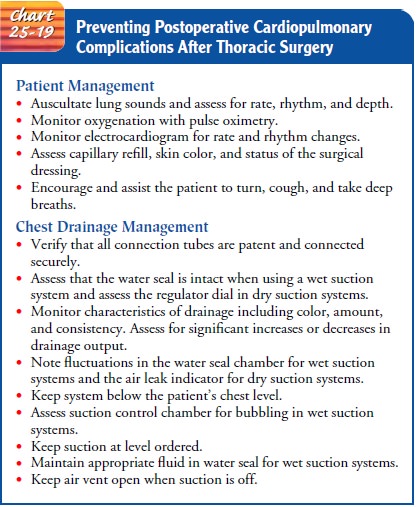
Chest Drainage
A
crucial intervention for improving gas exchange and breathing in the
postoperative period is the proper management of chest drainage and the chest drainage system. After thoracic
surgery, chest tubes and a closed drainage system are used to re-expand the
involved lung and to remove excess air, fluid, and blood. Chest drainage
systems also are used in treatment of spontaneous pneu-mothorax and trauma
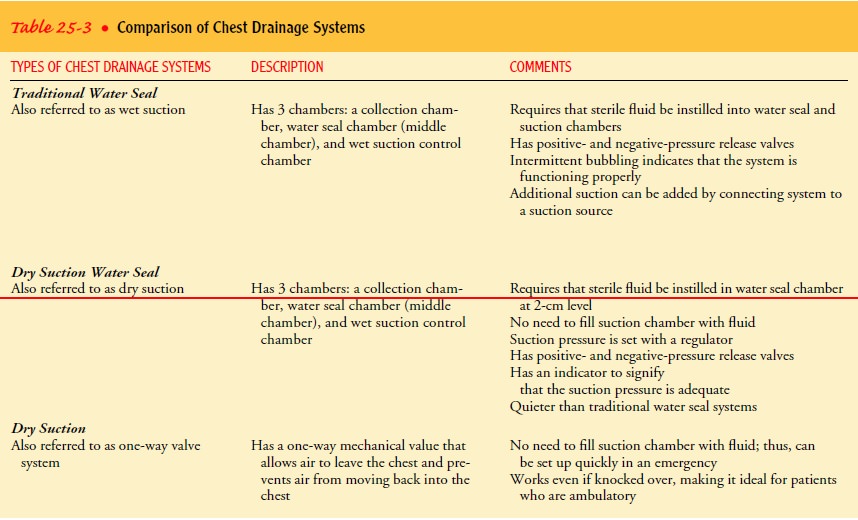
resulting in pneumothorax. Table 25-3 describes and compares the main features
of these systems. Management of chest drainage systems is explained in Chart
25-18. Prevention of cardiopulmonary complications following tho-racic surgery
is discussed in Chart 25-19.


The
normal breathing mechanism operates on the principle of negative pressure; that
is, the pressure in the chest cavity normally is lower than the pressure of the
atmosphere, causing air to move into the lungs during inspiration. Whenever the
chest is opened, there is a loss of negative pressure, which can result in the
collapse of the lung. The collection of air, fluid, or other substances in the
chest can compromise cardiopulmonary function and can also cause the lung to
collapse. Pathologic substances that collect in the pleural space include
fibrin, or clotted blood; liquids (serous fluids, blood, pus, chyle); and gases
(air from the lung, tracheo-bronchial tree, or esophagus).
Chest tubes may be inserted to drain fluid or air from any of the three compartments of the thorax (the right and left pleural spaces and the mediastinum). The pleural space, located between the visceral and parietal pleura, normally contains 20 mL or less of fluid, which helps to lubricate the visceral and parietal pleura. Surgical incision of the chest wall almost always causes some de-gree of pneumothorax (air accumulating in the pleural space) or hemothorax (build-up of serous fluid or blood in the pleural space). Air and fluid collect in the pleural space, restricting lung expansion and reducing gas exchange. Placement of a chest tube in the pleural space restores the negative intrathoracic pressure needed for lung re-expansion following surgery or trauma.
The
mediastinal space is an extrapleural space that lies be-tween the right and
left thoracic cavities. Mediastinal chest tubes promote the removal of blood or
other fluid from around the heart (Finkelmeier, 2000). Accumulating fluid can
stop the heart from beating if it is not drained. A mediastinal tube can be
in-serted either anteriorly or posteriorly to the heart to drain blood after
surgery or trauma. Without a tube, compression of the heart could occur,
leading to death (Carroll, 2000).
There
are two types of chest tubes: small-bore and large-bore catheters. Small-bore
catheters (7F to 12F) have a one-way valve apparatus to prevent air from moving
back into the patient. They can be inserted through a small skin incision.
Large-bore catheters,which range in size up to 40F, are usually connected to a
chest drainage system to collect any pleural fluid and monitor for air leaks
(Scanlan, Wilkins & Stoller, 1999). After the chest tube is positioned, it
is sutured to the skin and connected to a drainage apparatus to remove the
residual air and fluid from the pleural or mediastinal space. This results in
the re-expansion of remaining lung tissue.
CHEST DRAINAGE SYSTEMS
Chest drainage systems have a suction source, a collection chamber for pleural drainage, and a mechanism to prevent air from reentering the chest with inhalation.
Various types of chest
drainage systems are available for use in removal of air and fluid from the
pleural space and re-expansion of the lungs. Chest drainage systems come with
either wet (water seal) or dry suction control. In wet suction systems, the
amount of suction is determined by the amount of water instilled in the suction
chamber. The amount of bubbling in the suction chamber in-dicates how strong
the suction is. Wet systems use a water seal to prevent air from moving back
into the chest on inspiration. Dry systems use a one-way valve and a suction
control dial in place of the water needed with wet or water seal system. Both
systems can operate by gravity drainage, without a suction source.
Water Seal Chest Drainage Systems. The traditional water sealchest drainage system (or wet suction)
has three chambers: a collection chamber, a water seal chamber, and a wet
suction control chamber. The collection chamber acts as a reservoir for fluid
draining from the chest tube. It is graduated to permit easy measurement of
drainage. Suction may be added to create neg-ative pressure and promote
drainage of fluid and removal of air. The suction control chamber regulates the
amount of negative pressure applied to the chest. The amount of suction is
deter-mined by the water level. It is generally set at 20-cm water; adding more
fluid results in more suction. After the suction is turned on, bubbling appears
in the suction chamber. A positive-pressure valve is located at the top of the
suction chamber that automatically opens with increases in positive pressure
within the system. Air will automatically be released through a
posi-tive-pressure relief valve if the suction tubing is inadvertently clamped
or kinked.
The
water seal chamber has a one-way valve or water seal that prevents air from
moving back into the chest when the patient inhales. There will be an increase
in the water level with inspiration and a return to the baseline level during
exhalation; this is referred to as tidaling. Intermittent bubbling in the water
seal chamber is normal, but continuous bubbling can indicate an air leak. Bub-bling
and tidaling do not occur when the tube is placed in the me-diastinal space;
however, fluid may pulsate with the patient’s heartbeat. If the chest tube is
connected to gravity drainage only, suction is not used. The pressure is equal
to the water seal only. Two-chamber chest drainage systems (water seal chamber
and collection chamber) are available for use with patients who need only
gravity drainage.
The
water level in the water seal chamber reflects the negative pressure present in
the intrathoracic cavity. A rise in the water level indicates negative pressure
in the pleural or mediastinal space. Excessive negative pressure can cause
trauma to tissue (Bar-El, Ross, Kablawi & Egenburg, 2001). Most chest
drainage systems have an automatic means to prevent excessive negative
pressure. By pressing and holding a manual high-negativity vent (usually
located on the top of the chest drainage system) until the water level in the
water seal chamber returns to the 2-cm mark, excessive negative pressure is avoided,
preventing damage to tissue.
Dry Suction Water Seal Systems. Dry suction water seal systems,also referred to as dry suction,
have a collection chamber for drainage, a water seal chamber, and a dry suction
control cham-ber. The water seal chamber is filled with water to the 2-cm
level. Bubbling in this area can indicate an air leak. The dry suction control
chamber contains a regulator dial that conveniently reg-ulates vacuum to the
chest drain. Water is not needed for suction as it is in the wet system.
Without the bubbling in the suction chamber, the machine is quieter.
Once
the tube is connected to the suction source, the regula-tor dial allows the
desired level of suction to be dialed in; the suc-tion is increased until an
indicator appears. The indicator has the same function as the bubbling in the
traditional water seal sys-tem; that is, it indicates that the vacuum is
adequate to maintain the desired level of suction. Some drainage systems use a
bellows (a chamber that can be expanded or contracted) or an orange-colored
float device as an indicator of when the suction control regulator is set.
When the water in the water seal
rises above the 2-cm level, intrathoracic pressure increases. Dry suction water
seal systems have a manual high-negativity vent located on top of the drain.
Pressing the manual high-negativity vent until the indicator ap-pears (either a
float device or bellows) and the water level in the water seal returns to the
desired level, intrathoracic pressure is decreased.
Dry Suction with a One-Way Valve System. A third type of
chestdrainage system is dry suction with a one-way mechanical valve. This
system has a collection chamber, a one-way mechanical valve, and a dry suction
control chamber. The valve acts in the same way as a water seal and permits air
to leave the chest but prevents it from moving back into the pleural space.
This model lacks a water seal chamber and therefore has the advantage of a
system that operates without water. For example, it can be set up quickly in
emergency situations, and the dry control drain will still work even if it is
knocked over. If the wet suction drain is knocked over, the water seal could be
lost. This makes the dry suction systems useful for the patient who is
ambulating or being transported. However, without the water seal chamber, there
is no way to tell by inspection if the pressure in the chest has changed. An
air leak indicator is present so that the system can be checked for air leaks.
If an air leak is sus-pected, 30 mL of water are injected into the air leak
indicator. Bub-bles will appear if a leak is present (Carroll, 2000).
Related Topics