Chapter: Aquaculture Principles and Practices: Shrimps and Prawns
Reproduction and larval rearing of fresh-water prawns
Reproduction and larval rearing of fresh-water prawns
Mature M. rosenbergii (Fig. 25.16) easily mates and spawns in captivity throughout the year, even though in nature there are seasonal peaks associated with the onset of the rainy season. Berried females are collected and used for spawning purposes. The general practice is to select gravid females carrying almost ripe (brown-coloured) eggs, from commercial harvests. Brood stock of larger individuals above 45g in weight are preferred as the quantity of eggs spawned is comparatively high. Brood prawns are reared in fresh or slightly brackish water (salinity 2–8ppt) and fed on mussels, cockles, fresh trash fish or compounded feeds.

Where brood ponds are maintained, they are stocked at a lower density of about 12500/ha and the females raised to a weight of 100g to increase the production of eggs and larvae. The eggs change in colour from orange to greyish-brown as they mature. Mature male prawns are considerably larger than the females, their second walking legs much larger and thicker, the cephalothorax proportionately larger and the abdomen narrower. The male can also be distinguished by the presence of a lump or hard point in the centre on the ventral side of the first segment of the abdomen.
Hatchery systems
There are various hatchery systems in use in experimental centres and commercial farms, but the more important ones can be described as belonging to the green water (see fig. 7.3), ‘clear water’ and recirculation systems. The so-called backyard hatcheries use one of these systems, more commonly the green water system. In many hatcheries, mating, spawning and larval rearing occur in the same tank. Larger tanks are required for water storage, post-larval rearing and for hatching Artemia. The tanks may be in the open in tropical areas, but are usually kept under a roof to prevent the water from getting too warm during summer days and too cold at nights. In temperature climates it is necessary to house the tanks in a greenhouse-type construction, to maintain the water temperature at the required level during the cold season.
As mentioned, hatchery tanks can be made in various shapes and sizes using plastic, fibreglass, reinforced concrete, etc. Circular (with flat or conical bottom), rectangular and square-shaped tanks are in use. Though each has its advantages, rectangular tanks are more convenient and space-saving. A 10m3 rectangular tank, with water and air intakes at one end and the drain at the other, has been found to work very efficiently. However, some cultur-ists prefer the circular tanks (fig. 25.17), because they are easy to clean and better water circulation can be maintained in them. Many culturists recommend the tanks to be painted with a dark colour, to enable the larvae to see the feed well. An aeration system at the bottom of the tank helps proper mixing of the water and even distribution of the food particles and the larvae, thereby reducing cannibalism.

Sandifer et al. (1983) described the use of artificial ‘habitat units’ that increase the surface area available to the larvae in rearing systems. In the presence of stacked solid layers, the larvae exhibit what is termed an ‘edge effect’, which is a pronounced preference for the larger edges. The units consist of a rigid frame to which layers of plastic mesh are attached. The mesh layers increase the area of surface edges significantly, both vertically and horizontally. Strips of screening placed on different layers provide feeding spots, where the food settles. Tanks for holding larvae before distribution and for mixing brackish water are of the same design as larval tanks, but are larger, about 50m3. The water supply, as far as possible from bore wells, provides unpolluted fresh and salt water. A pH in the range of 7.0–8.5, a temperature near to the optimum of 28–31°C and nitrite and nitrate levels not higher than 0.1ppm (NO2-N) and 20ppm (NO3-N), have been recommended.
The terms green water, clear water and recirculating systems of larval culture refer largely to the water management procedures. The green water system, as the name implies, involves the production of a mixed phyto-plankton culture dominated by Chlorella at a cell density of 750000–1500000 cells/ml. A solution of a fertilizer mixture of four parts urea to 1 part NPK (15:15:15) in tap water is added to the tank at least once a week, to maintain the plankton bloom. The production of rotifers in the tank is controlled by the application of CuSO4 at the rate of about 0.6ppm. The growth of filamentous algae may be controlled by holding T. mossambica in the tanks at the rate of 1 fish per 400 l. The salinity of the green water is not more than 12ppt, and it is used as replacement water during water exchange of the larval tanks. Only green water less than three days old is used. For a continuous production of such water, without causing phytoplankton ‘crashes’, it is diluted regularly and fertilized as required. The function of the green water is to maintain the water quality in the larval tanks by recycling the waste products of larval metabolism, and to provide food for live food organisms such as Artemia. The effect of the green water is very much dependent on meteorological conditions and the quality of fresh and sea water. So, though basically simple, its efficiency depends very much on local conditions. Furthermore, in this systemonly a lower stocking density of about 30–50 larvae/l, yielding on average 5–25 post-larvae/l, is feasible.
The clear water system can be adopted for different levels of larval production. Circular tanks with conical bottoms or rectangular tanks are used. The system developed by Aquacop (1977a) for high-density larval culture involves the use of conical-bottomed tanks of 800 l capacity with total water exchange and environmental control, including temperature and light intensity. The water quality is maintained by chlorination, followed by dechlorination with UV light, strong aeration and the use of antibiotics. Higher stocking levels of 100–200 larvae/l yielding 25–100 post-larvae/l are feasible, but require very careful management.
Recirculation systems are valuable in water conservation and energy use. The simplest type of this system recirculates the tank water through a graded sand/gravel filter, using mechanical or air lift pumps. More complicated, completely closed systems involving mechanical and biological filtration, chlorination and dechlorination or the use of ozone, have also been developed. Though the value of such a system in controlling the water quality is recognized and is utilized in experimental work, commercial application on any significant scale has yet to be realized. On the other hand, efforts are made in commercial hatcheries to operate both green water and clear water systems with only limited water exchange.
Spawning and larval rearing
Mating takes place between hard-shelled males and ripe soft-shelled females which have completed their premating moult. Semen is deposited in a gelatinous mass on the underside of the thoracic region of the female, between the walking legs. Within a few hours, the female extrudes eggs through the gonopore and the eggs are fertilized by the semen attached to the abdomen and transferred to a brood chamber located under the abdomen between the pleopods. It is believed that an enzymatic reaction is involved in the release of sperm cells from the spermatophore. The fertilized eggs become attached to each other and the setae of the first four pleopods by a cementing
substance, which hardens into an attachment membrane. The vigorous movements of the pleopods keep the eggs well aerated. The number of eggs laid varies with the size of the prawn, but it is reported to be up to 80000– 100000 per spawn. However, a one-year-old female may spawn only 5000–20000 eggs during its first spawning. The incubation period ranges from 18 to 23 days at temperatures of about 28°C. Even during the incubation period of fertilized eggs, ovarian eggs start maturing and a second pre-mating moult can occur within a period of about three weeks.
Selected healthy berried females from natural sources, rearing ponds, brood tanks or aquaria are introduced into the larval tanks. In community rearing tanks, it is necessary to ensure that all of them have black or grey eggs, so that they will all hatch out within two or three days and the larvae in the tanks will be of the same age. This helps in appropriate feeding and reducing cannibalism among the larvae.
Brackish water with a salinity of about 5ppt is provided in the tanks, as hatching rates are lower in fresh water. Water of the required salinity is obtained from mixing tanks in the hatchery. It is, however, possible to hatch the eggs in fresh water and raise the salinity after hatching.After the eggs have hatched, the spent females are removed from the tank with a coarse-mesh dip net. Usually larval release is over a four-day period, with a peak between 24 and 72 hours. So the females have to remain in the tanks for up to four days. The salinity of the water can then be raised to around 12ppt. The optimum temperature range is 26–31°C, and sudden changes of salinity or temperature are avoided. Aeration is maintained in the tanks in order to maintain oxygen levels near saturation. In clear water and recirculating system hatcheries the tanks are not exposed to direct sunlight, and at least part of the tanks are under cover, providing some suitable source of indirect light.
Macrobrachium undergoes 11 stages duringlarval development (fig. 25.18) and the time taken for a batch of larvae to metamorphose varies according to temperature and feeding conditions. Under favourable conditions, the post-larval stage is reached in 16–28 days. The past-larvae resemble juvenile prawns and rest or crawl on the tank surfaces. At this stage they are harvested from the tanks for nursing or on-growing.
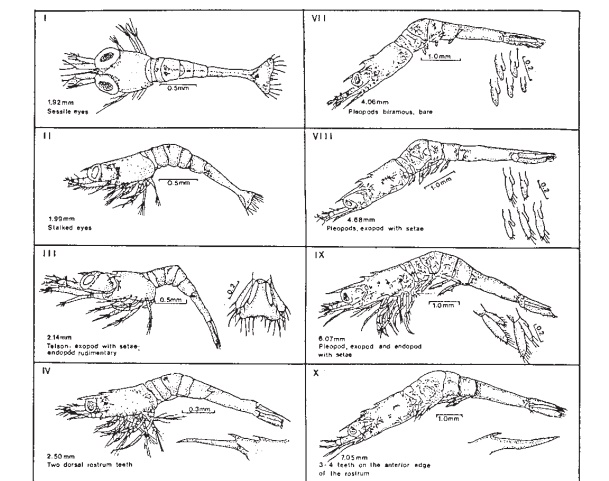
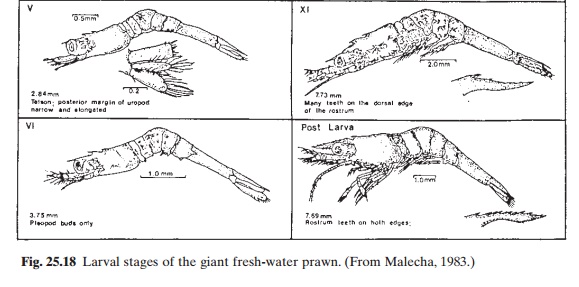
There is considerable variation in the density of larvae reared in larval tanks. A final density of 40/l is recommended; but the culture can start with a higher density of 80–120/l, and the stock can be split on about the twelfth day (stages V–VIII) in order to reach the final desired density. In small rearing ponds, it may be possible to carry out the rearing without any division of stock. In hatcheries following the green water system, water in the tanks is lowered daily to a 50 per cent level and replaced with green water. In clean water systems, after removal of about 50 per cent of the water clean water is used for refilling. Some hatcheries in Hawaii practise a combination of these systems. They follow the green water system during the early part of the culture (three to six days) and then switch to clean water.
Appropriate feeding is a key factor in successful larval rearing. Different types of feeds are in use in hatcheries, the more important of which are nauplii of Artemia, fish flesh, fish roe, egg custard, egg and mussel mixtures and compound feeds. As Macrobrachium larvae do not actively search for food, feeding is generally started with swimming nauplii of Artemia twice a day and continued until the fifth day after hatching. The actual quantity to be fed is determined by visual examination of the tank, the main consideration being to provide an adequate distribution of the nauplii near each larva, to facilitate feeding. New and Singholka (1985) roughly calculated the requirement of nauplii for a 10m3 larval tank to be about 10–50 million per day. Prepared feeds are generally given only from the fifth day, even though weaning can be started a couple of days earlier. These are given four or five times during the day, and it is recommended that the night feeding should be with Artemia nauplii only. The quantity of prepared feed is also determined by the need for a suspension of it in the tank, to make it easy for the larvae to locate it. The quantity needed at each feeding has been calculated to be about 30–60g per tank initially, gradually increasing to 200g. Vigorous aeration helps to keep the feed in suspension near the larvae.
Fish such as skipjack tuna, bonito and pollock, used as feed for the larvae, are filleted, ground and forced through stainless steel sieves with strong jets of water to obtain particles of the required size. Particles of 0.3mm size are suitable for feeding until the tenth day and from then on 0.3–1mm size until metamorphosis.
Egg custard for feeding larvae is prepared by cooking scrambled whole chicken eggs andpassing it through stainless steel sieves of the required mesh size. A mixture of eggs and mussels has been found to be an efficient feed for larvae. Shelled mussels are minced to fine particles, strained through a coarse sieve and blended with whole beaten egg. The mixture is steamed until it solidifies and then sieved to produce particles of the required size.
As mentioned earlier, the time taken for a larval batch to finish metamorphosis depends on the environmental conditions, but it is usually completed in less than 28 days and it is best to harvest them at this stage. The post-larvae can then be transferred to fresh water, but it is advisable to acclimatize them through a gradual lowering of the salinity by draining part of the brackish water and replacing it with fresh water. The production from a 10m3 tank is between 100000 and 200000 larvae.
The post-larvae are stocked directly in production ponds or grown for a period of one or two months before on-growing to market size. The nursery ponds are stocked at higher densities of about 1000/m2. Besides the normal advantages of nursery rearing, this practice helps the farmer to count the number of juve-niles and assess their state of health before release into production ponds. Nursery rearing is considered essential in temperate climates, but even in tropical climates this practice can be beneficial. An increasing number of farms seem to be incorporating a nursery stage in the culture operations.
Related Topics