Chapter: Plant Biochemistry: Photosynthesis is an electron transport process
Regulatory processes control the distribution of the captured photons between the two photosystems
Regulatory processes control the distribution of the captured photons between the two photosystems
Linear photosynthetic electron transport through the two photosystems requires the even distribution of the captured excitons between them. The excitons are transferred preferentially to the chromophore which requires the least energy for excitation. Photosystem I (P700) being on a lower energetic level than PS II (Fig. 3.16) requires less energy for excitation than photosystem II (P680). In an unrestricted compe-tition between the two photosystems, excitons would primarily be directed to PS I. Due to this imbalance, the distribution of the excitons between the two photosystems must be regulated. The spatial separation of PS I and PS II and their antennae in the thylakoid membrane plays an important role in this regulation.
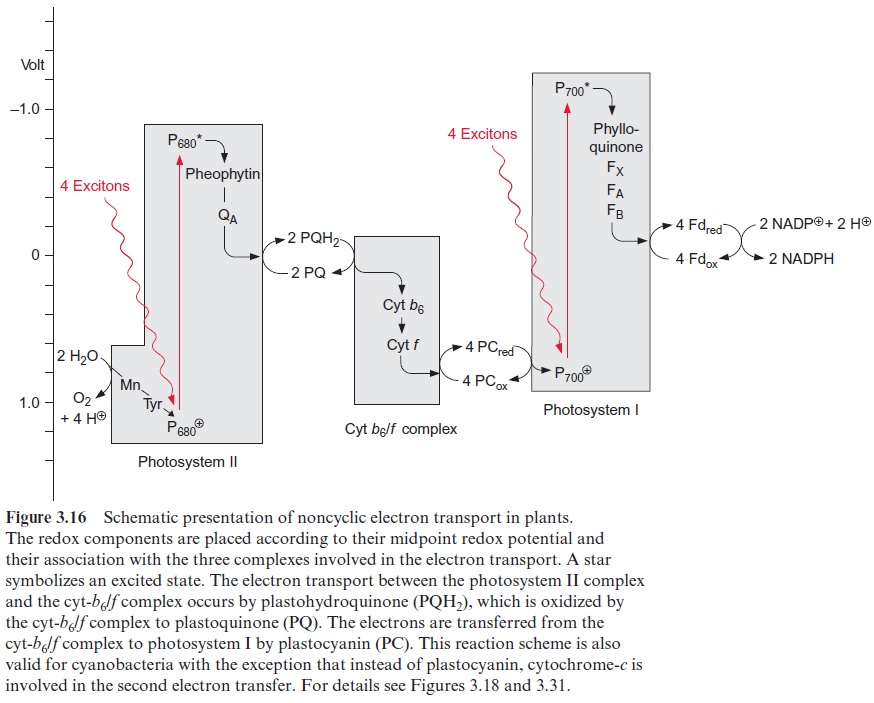
In chloroplasts, the thylakoid membranes are present in two differ-ent arrays, as stacked and unstacked membranes. The outer surface of the unstacked membranes has free access to the stromal space; these mem-branes are called stromal lamellae (Fig. 3.40). In the stacked membranes, the neighboring thylakoid membranes are in direct contact with each other. These membrane stacks can be seen as grains (grana) in light microscopy and are therefore called granal lamellae.
ATP synthase and the PS I complex (including its light harvesting com-plexes, not further discussed here) are located either in the stromal lamellae or in the outer membrane region of the granal lamellae. Therefore, these proteins have free access to ADP and NADP+ in the stroma. The PS II complex, on the other hand, is primarily located in the granal lamellae. Peripheral LHC II subunits attached to the PS II complex contain a protein chain protruding from the membrane, which can prob-ably interact with the LHC II subunit of the adjacent membrane and thus cause tight membrane stacking. The cyt-b6/f complexes are only present in stacked membranes. Since the proteins of PS I and F-ATP-synthase project into the stroma space, they do not fit into the space between the stacked membranes. Thus the PS II complexes in the stacked membranes are sepa-rated spatially from the PS I complexes in the unstacked membranes. It is assumed that this prevents an uncontrolled spillover of excitonsfrom PS II to PS I.
However, the spatial separation of the two photosystems and thus the spillover of excitons from PS II to PS I can be regulated. For example, if the excitation of PS II is greater than that of PS I, plastohydroquinone accumulates, which cannot be oxidized rapidly enough via the cyt-b6/f com-plex by PS I. Under these conditions, a protein kinase is activated, which phosphorylates the hydroxyl groups of threonine residues of peripheral LHC II subunits, causing a conformational change of the LHC protein. As a result of this, the affinity to PS II is decreased and the LHC II subunits dissociate from the PS II complexes. Furthermore, due to the changed con-formation, LHC II subunits can now bind to PS I, mediated by the H sub-unit of PS II. This LHC II-PS I complex purposely increases the spillover of excitons from LHC II to PS I. In this way the accumulation of reduced plastoquinone decreases the excitation of PS II and enhances the excitation of PS I. A protein phosphatase facilitates the reversal of this regulation. This regulatory process, which has been simplified here, enables an opti-mized distribution of the captured photons between the two photosystems, independent of the spectral quality of the absorbed light.
Excess light energy is eliminated as heat
Plants face the general problem that the energy of irradiated light can be much higher than the demand of photosynthetic metabolites such as NADPH and ATP. This is the case when very high light intensities are present and the metabolism cannot keep pace. Such a situation arises at low temperatures, when the metabolism is slowed down because of decreased enzyme activities (cold stress) or at high temperatures, when stomata close to prevent loss of water. Excess excitation of the photosystems could result in an excessive reduction of the components of the photosynthetic electron transport.
Very high excitation of photosystem II, recognized by the accumulation of plastohydroquinone, results in damage to the photosynthetic appara-tus, termedphotoinhibition. A major cause of this damage is an overexcita-tion of the reaction center, by which chlorophyll molecules attain a triplet state, resulting in the formation of aggressive singlet oxygen. The damaging effect of triplet chlorophyll can be demonstrated by placing a small amount of chlorophyll under the human skin, which after illumina-tion causes severe tissue damage. This photodynamic principle is utilized in medicine for the selective therapy of skin cancer.
Carotenoids (e.g., β-carotene, Fig. 2.9) are able to convert the triplet state of chlorophyll and the singlet state of oxygen to the corresponding ground states by forming a triplet carotenoid, which dissipates its energy as heat. In this way carotenoids have an important protective function. If under certain conditions this protective function of carotenoids is una-ble to cope with excessive excitation of PS II, the remaining singlet oxy-gen has a damaging effect on the PS II complex. The site of this damage could be the D1 protein of the photosynthetic reaction center in PS II, which already under normal photosynthetic conditions experiences a high turnover. When the rate of D1-protein damage exceeds the rate of its resynthesis, the rate of photosynthesis is decreased, resulting in photoinhibition.
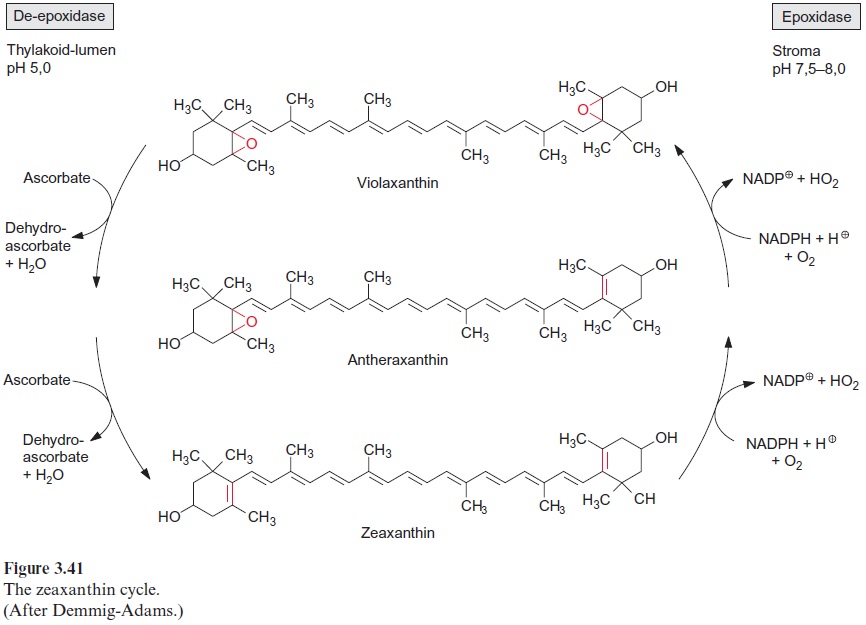
Plants have developed several mechanisms to protect the photosynthetic apparatus from light damage. One mechanism is chloroplast avoidance movement, in which chloroplasts move under high light conditions from the cell surface to the side walls of the cells. Another way is to dissipate the energy arising from an excess of excitons as heat. This process is termed nonphotochemical quenching of exciton energy. Although our knowledge of this quenching process is still incomplete, it is undisputed thatzeaxan-thin plays an important role. Zeaxanthin causes the dissipation of exciton energy to heat by interacting with a chlorophyll-binding protein (CP 22) of photosystem II. Zeaxanthin is formed by the reduction of the diepoxide vio-laxanthin. The reduction proceeds with ascorbate as the reductant and the monoepoxide antheraxanthin is formed as an intermediate. Zeaxanthin can be reconverted to violaxanthin by epoxidation which requires NADPH and O2 (Fig. 3.41). Formation of zeaxanthin by diepoxidase takes place on the luminal side of the thylakoid membrane at an optimum pH of 5.0, whereas the regeneration of violaxanthin by the epoxidase proceeding at the stro-mal side of the thylakoid membrane occurs at about pH 7.6. Therefore, the formation of zeaxanthin requires a high pH gradient across the thylakoid membrane. As discussed in connection with the Mehler reaction , a high pH gradient can be an indicator of the high excitation state of photosystem II. When there is too much excitation energy, an increased pH gradient initiates zeaxanthin synthesis, dissipating excess energy of the PS II complex as heat. This mechanism explains how under strong sunlight most plants convert 50% to 70% of all the absorbed photons to heat. The non-photochemical quenching of excitation energy is the primary way for plants to protect themselves from too much light energy. In comparison, the Mehler reaction and photorespiration under most conditions play only a minor role in the elimination of excess excita-tion energy.
Related Topics