Chapter: Plant Biochemistry: Photosynthesis is an electron transport process
Two photosynthetic reaction centers are arranged in tandem in photosynthesis of algae and plants
Two photosynthetic reaction centers are arranged in tandem in photosynthesis of algae and plants
In green algae about eight photons are required (quantum requirement: pho-tons absorbed per molecule O2 produced) for the photosynthetic water split-ting . Instead of the term quantum requirement, one often uses the reciprocal term quantum yield(molecules of O2 produced per photon absorbed). According to the color of irradiated light (action spectrum) the quantum yield dropped very sharply when algae were illuminated with red light above a wavelength of 680 nm (Fig. 3.14). This effect, named “red drop,” remained unexplained since algae contain chlorophyll, which absorbs light at 700 nm. Robert Emerson and coworkers (USA) solved this problem in 1957 when they observed that the quantum yield in the spectral range above 680 nm increased dramatically when algae were illuminated with orange light (650 nm) and red light simultaneously. Then the quantum yield was higher than the sum of both yields when irradiated separately with the light of each wavelength. This Emerson effect led to the conclusion that two differ-ent reaction centers are involved in photosynthesis of green algae (and also of cyanobacteria and higher plants). In 1960 Robert Hill (Cambridge, UK) postulated a reaction scheme (Fig. 3.15) in which two reaction centers are arranged in tandem and connected by an electron transport chain containing cytochrome-b6 and cytochrome-f (cytochrome-f is a cytochrome of the c type). Light energy of 700 nm was sufficient for the excitation of reaction center I, whereas excitation of the other reaction center II required light of higher energy with a wavelength of 680 nm. The electron flow accord-ing to the redox potentials of the intermediates shows a zigzag, leading to the name Z scheme. The numbering of the two photosystems corresponds to the sequence of their discovery.Photosystem II (PS II) can use light up to a wave-length of 680 nm, whereas photosystem I (PS I) can utilize light with a wave-length up to 700 nm. The sequence of the two photosystems makes it possible that at PS II a very strong oxidant is generated for the oxidation of water and at PS I a very strong reductant is produced for the reduction of NADP+ (see also Fig. 3.3).
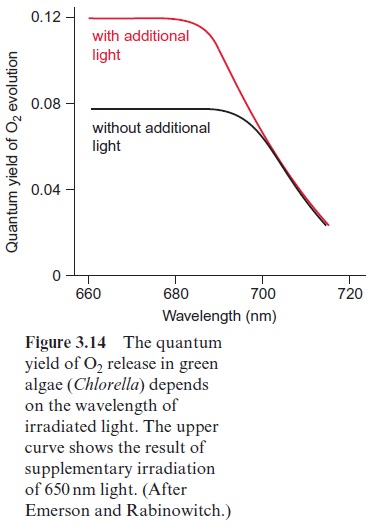
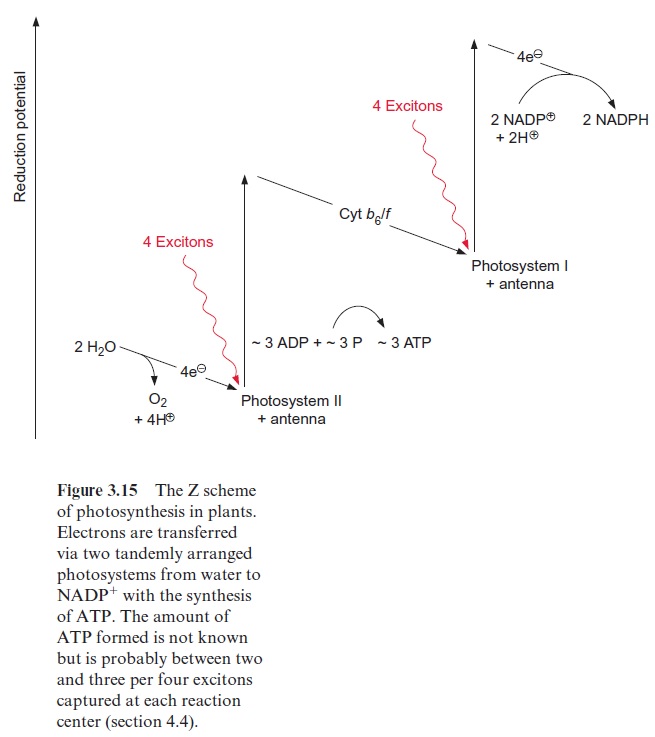
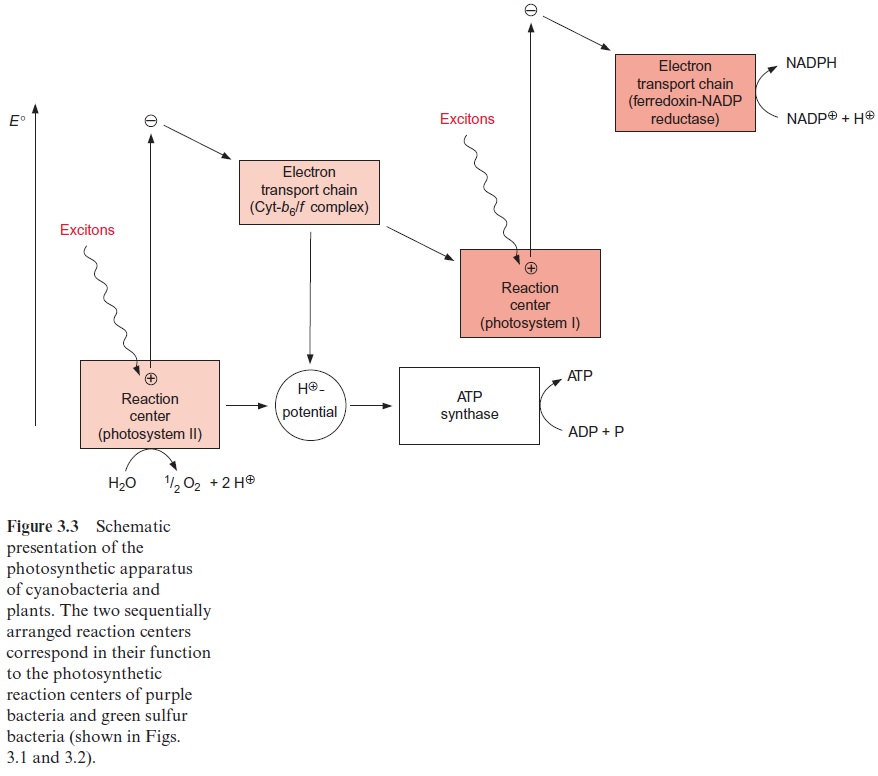
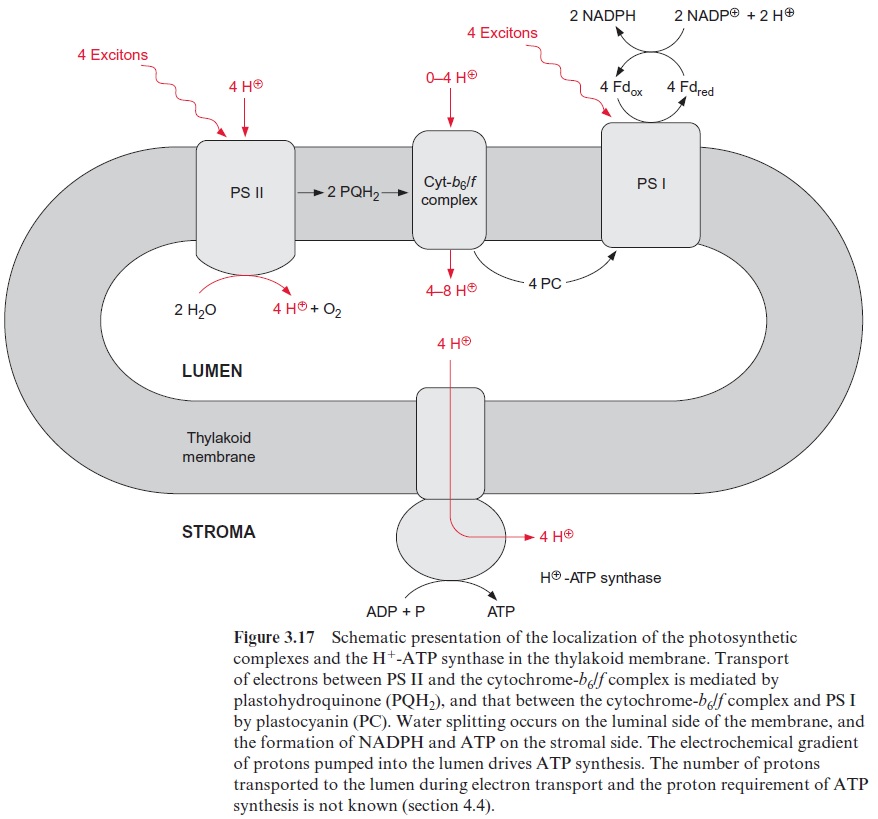
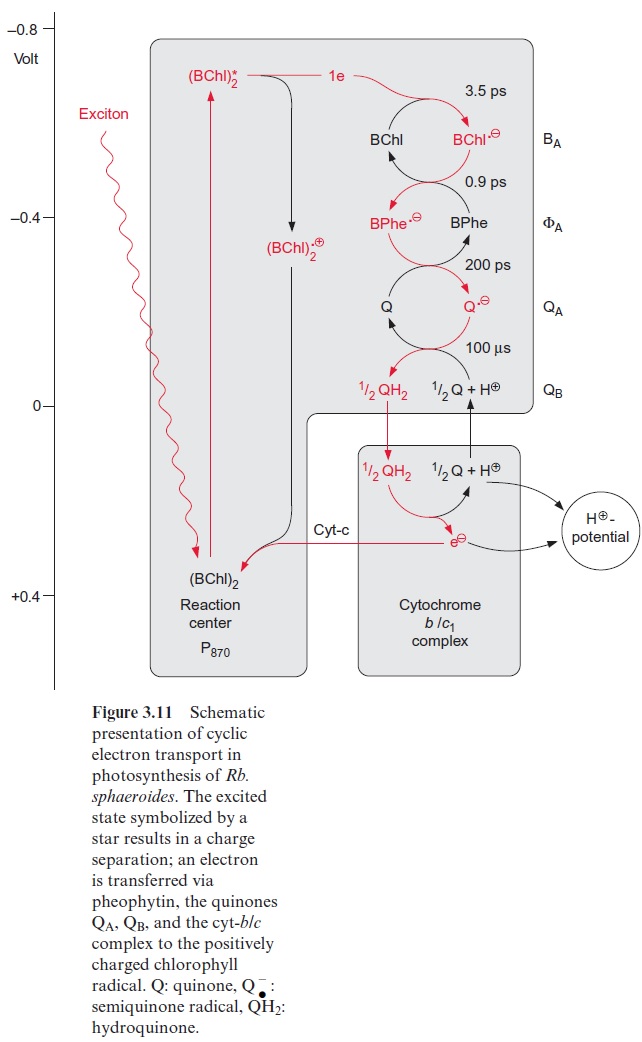
Figure 3.16 gives an overview of electron transport through the photo-synthetic complexes; the carriers of electron transport are drawn according to their electric potential (see also Fig. 3.11). Figure 3.17 shows how the photosynthetic complexes are arranged in the thylakoid membrane. There is a potential difference of about 1.2 volt between the process of water oxi-dation and NADP+reduction. The absorbed photons of 680 and 700 nm together correspond to a total potential difference of 3.45 volt. Thus, only about one-third of the energy of the pho-tons absorbed by the two photosystems is used to transfer electrons from water to NADP+ . In addition to this, about one-eighth of the light energy absorbed by the two photosystems is conserved by pumping protons into the lumen of the thylakoids via PS II and the cytochrome-b6/f complex (Fig. 3.17). This proton transport leads to the formation of a proton gradi-ent between the lumen and the stroma space. An H+ -ATP synthase, also located in the thylakoid membrane, uses the energy of the proton gradient to synthesize ATP.
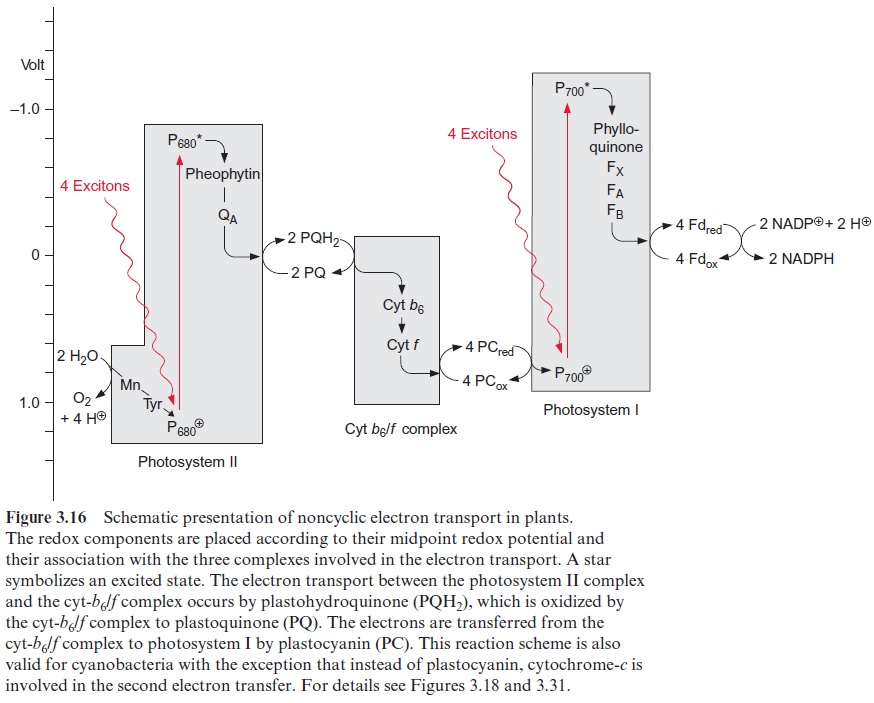
Thus about half the absorbed light energy of the two photosystems is not used for chemical work but is dissipated as heat.
Related Topics