Chapter: Basic & Clinical Pharmacology : Drug Receptors & Pharmacodynamics
Receptor Classes & Drug Development
RECEPTOR CLASSES &DRUG
DEVELOPMENT
The
existence of a specific drug receptor is usually inferred from studying the structure-activity relationship of a
group of struc-turally similar congeners of the drug that mimic or antagonize
its effects. Thus, if a series of related agonists exhibits identical rela-tive
potencies in producing two distinct effects, it is likely that the two effects
are mediated by similar or identical receptor molecules. In addition, if
identical receptors mediate both effects, a competi-tive antagonist will
inhibit both responses with the same Ki; a second competitive
antagonist will inhibit both responses with its own characteristic Ki.
Thus, studies of the relation between struc-ture and activity of a series of
agonists and antagonists can identify a species of receptor that mediates a set
of pharmacologic responses.
Exactly
the same experimental procedure can show that observed effects of a drug are
mediated by different receptors. In
this case, effects mediated by different receptors may exhibit dif-ferent
orders of potency among agonists and different Ki values for each
competitive antagonist.
Wherever
we look, evolution has created many different recep-tors that function to
mediate responses to any individual chemical signal. In some cases, the same
chemical acts on completely differ-ent structural receptor classes. For
example, acetylcholine uses ligand-gated ion channels (nicotinic AChRs) to
initiate a fast (in milliseconds) excitatory postsynaptic potential (EPSP) in
postgan-glionic neurons. Acetylcholine also activates a separate class of G
protein-coupled receptors (muscarinic AChRs), which mediate slower (seconds to
minutes) modulatory effects on the same neurons. In addition, each structural
class usually includes multiple subtypes of receptor, often with significantly
different signaling or regulatory properties. For example, many biogenic amines
(eg, norepinephrine, acetylcholine, and serotonin) activate more than one
receptor, each of which may activate a different G protein, as previously
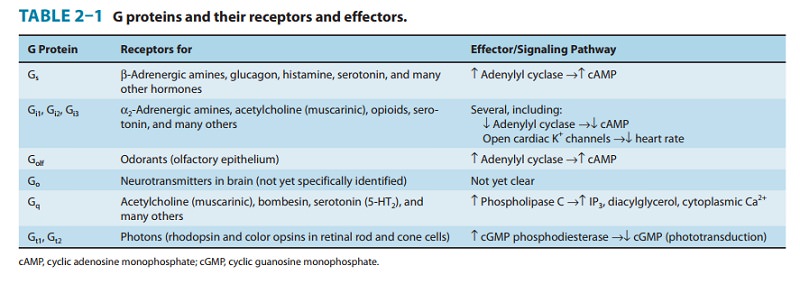
described (see also Table 2–1). The existence of many receptor classes and
subtypes for the same endogenous ligand has created important opportunities for
drug development. For example, propranolol, a selective antago-nist of β adrenoceptors, can
reduce an accelerated heart rate without preventing the sympathetic nervous
system from causing vasocon-striction, an effect mediated by α1 receptors.
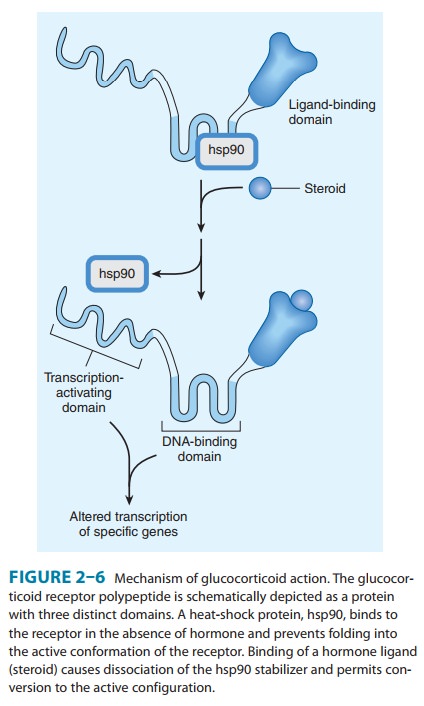
The
principle of drug selectivity may even apply to structurally identical
receptors expressed in different cells, eg, receptors for steroids such as
estrogen (Figure 2–6). Different cell types express different accessory
proteins, which interact with steroid receptors and change the functional
effects of drug-receptor interaction. For example, tamoxifen acts as an antagonist on estrogen receptors
expressed in mammary tissue but as an agonist
on estrogen recep-tors in bone. Consequently, tamoxifen may be useful not only
in the treatment and prophylaxis of breast cancer but also in the prevention of
osteoporosis by increasing bone density. Tamoxifen may also create
complications in postmenopausal women, however, by exerting an agonist action
in the uterus, stimulating endometrial cell proliferation.
New
drug development is not confined to agents that act on receptors for
extracellular chemical signals. Increasingly, pharma-ceutical chemists are
determining whether elements of signaling pathways distal to the receptors may
also serve as targets of selec-tive and useful drugs. We have already discussed
drugs that act on phosphodiesterase and some intracellular kinases. There are
several additional kinase inhibitors presently in clinical trials, as well as
preclinical efforts directed at developing inhibitors of G proteins.
Related Topics