Chapter: Modern Medical Toxicology: Miscellaneous Drugs and Poisons: Gastrointestinal and Endocrinal Drugs
Radiocontrast Agents
RADIOCONTRAST AGENTS
Most
radiocontrast agents in use are iodinated contrast material which may be ionic
or non-ionic compounds.
Classification
·
Higher-osmolality contrast agents (HOCA)—These are mostly
ionic compounds and have been in use for several decades. They ![]() are relatively cheap and safe,
though occasionally severe adverse reactions can occur.
are relatively cheap and safe,
though occasionally severe adverse reactions can occur.
·
Lower-osmolality contrast agents (LOCA)—These are mostly
non-ionic compounds which cause less discomfort and are associated with a lower
incidence of severe adverse reac-tions. However they are quite expensive.
Uses
·
Urography: The agents used for
urography comprise mainly small molecule, water soluble, low protein binding,
high plasma concentration compounds which are given IV.
Examples—
o Ionic monomers: diatrizoates, iothalamates, metrizoates, iodamide,
ioxithalamate.
–– Ionic dimers : ioxaglic acid.
––
Non-ionic monomers : iohexol, iopamidol, ipro-mide, iopentol, metrizamole.
–– Non-ionic dimers : iotralan, iodixanol.
Angiography: These agents are water soluble, with low viscocity and
radiodensity.
·
Examples:
–– Non-ionic monomers: iohexol.
Contrast radiography of GI tract:
These are nonabsorbable agents which form a homogenous coat on the GI mucosa
and do not interact with GI secretions.
·
Examples:
––
Barium sulfate.
Computerised tomography of GI tract:
These are non-absorbable iodinated water-soluble agents with high osmo-lality.
·
Examples:
––
Diatrizoate. Myelography: Agents for this are non-ionic, water soluble,
and miscible with CSF.
·
Examples:
––
Metrizamide, iotralan.
Lymphography, lymphangiography:
These agents are water-insoluble with high radiodensity.
·
Examples:
––
Iodised oil, iotasol.
Magnetic resonance imaging: YYExamples:
–– Gadolinium, manganese, and iron
as the aminopo-lycarboxylate chelates and gadopentetic acid.
Cholecystography, cholangiography:
These agents are preferentially excreted in the bile after absorption from GI
tract.
·
Examples:
–– Ipodates, iocetamic acid,
iopanoic acid, sodium tyropanoate.
Adverse Effects
Contrast media are known for
producing severe reactions, though they are relatively infrequent (1 or 2 per
1000 exami-nations) . Mild to moderate reactions are more common, but subside
on their own.
■■Anaphylactoid reaction:
o Predisposing factors:
–– Cerebral or renal disease in
patient over the age of 50 years.
–– History of allergy (including
asthma). –– History of cardiac disease.
––
History of reaction to contrast material.
–– Multiple myeloma, homocystinuria,
sickle cell anaemia, phaeochromocytoma.
–– Previous study in which large
dose of contrast material was used.
o Categories
of reaction:
–– Mild: nausea, vomiting, cough,
headache, vertigo, itching, pallor, flushing, chills, sweats, rash (hives),
nasal stuffiness, and swelling of face and eyes.
–– Moderate: moderate intensity of
any of the above manifestations, with/without the following—pulse changes,
hypo- or hypertension, dyspnoea, bron-chospasm, and laryngospasm.
–– Severe: life-threatening
manifestations including severe laryngospasm, convulsions, arrhythmias,
unresponsiveness and cardiopulmonary arrest.
Cardiovascular side effects:
o Cardiac ischaemia with pain and
arrhythmias, usually accompanied by dyspnoea. Hypotension with tachy- cardia is
commonly observed.
o ECG changes include sinus
bradycardia, heart block, Q-T prolongation, ventricular
tachycardia/fibrillation, and ST segment and T wave changes.
·
Gastrointestinal side effects: vomiting, abdominal pain.
·
Neurological side effects: Headache (may be associated with
intracerebral haemorrhage), amnesia, visual blurring, cortical blindness,
encephalopathy, vertigo, and convulsions.
·
Pulmonary side effects: Non-cardiogenic pulmonary oedema is
relatively commonly reported.
·
Renal side effects: There have been several reports of acute
renal failure following injection of water soluble contrast media. Table 32.9 lists important predisposing
factors.
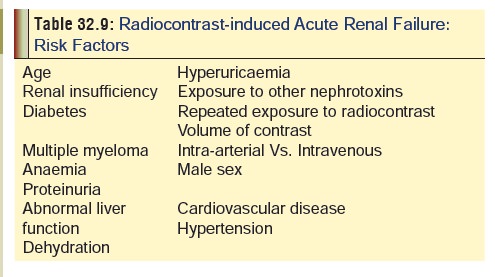
·
Clinically, there is acute tubular necrosis, presenting with
oliguria within 24 hours of exposure to the agent.
A formula has been suggested for calculating the maximum dose of contrast material that can be given safely without compromising renal function:
Contrast agent (maximum limit)
·
Thromboembolic phenomena: Serious thromboembolic events
causing myocardial infarction and stroke have occurred during angiographic
procedures with contrast media. Careful intravascular administration is
necessary to minimise such complications. The following is a list of
precautions:
o Continuous flushing with saline solution to prevent mixing of blood and contrast media, premedication with heparin, and use of plastic syringes are important safety measures when using non-ionic contrast media.
o Non-ionic contrast media should not
be mixed with blood before intravascular injection.
o Extra caution should be exercised when
using non-ionic contrast media in high-risk patients (elderly patients,
patients with coagulation defects, etc.).
·
Thyroid complications: Since radiocontrast agents invar-
iably contain iodides which cannot be metabolised by deiodinating enzymes,
thyrotoxicosis may be induced in susceptible patients. Prophylactic treatment
with sodium perchlorate 1.2 grams administered 30 minutes before, and 6 to 8
hours after exposure, has been suggested.
·
Cancer induction: Thorotrast, a contrast agent which
contains 25% colloidal thorium dioxide has been associated with malignancies.
·
Precipitation of mumps: Iodide
mumps has occasionally been observed in patients who were administered
iodinated contrast agents.
Clinical (Toxic) Features
·
Inadvertent administration of ionic contrast agents such as
diatrizoate or iodamine, instead of iopanidol, by the intrath-ecal route, has
resulted in fatalities.
·
In the past, methiodal sodium was commonly used for
myelography. Because of high incidence of neurotoxic adverse reactions, other
compounds were subsequently introduced, e.g. iothalamate meglumine and
iocarmate.
·
Inadvertent myelography with diatrizoate has led to lumbar
pain, tonic-clonic convulsions, hyperthermia, rhabdomy-olysis, disseminated
intravascular coagulation, renal failure, pulmonary oedema and death.
·
Overdose with iopanoic acid has led to vomiting, diarrhoea,
hypotension, coronary insufficiency, acute hepatic necrosis, renal failure and
death.
·
Overdose with iothalamate meglumine during excretory
urography can lead to cardiopulmonary failure.
Treatment
Adverse Effects
·
Anaphylactoid reaction:
For
urticaria:
–– Mild:
--
Diphenhydramine (50 mg oral/IM/IV), or hydroxyzine (25–50 mg oral/IM/IV).
--Cimetidine
(300 mg oral/slow IV in 10 ml D5W soln), or ranitidine (50 mg oral/slow IV in
10 ml D5W solution).![]()
–– Severe:
-- Adrenaline (1:1000) SC, 0.1 to 0.3 ml.
·
For facial/laryngeal oedema:
–– Mild:
--
Adrenaline (1:1000) SC, 0.1 to 0.3 ml; can be repeated up to 3 times (1 mg
max).
-- Oxygen, 2 to 6 L/min. –– Severe:
--
Intubation.
·
For bronchospasm:
––
Oxygen, 2 to 6 L/min.
–– Adrenaline (1:1000) SC, 0.1 to
0.3 ml, or beta-agonist inhalers.
–– Aminophylline 6 mg/kg, IV, in
D5W, slowly, or terbutaline 0.25 to 0.5 mg, IM/SC.
·
For hypotension with tachycardia:
––
Mild:
-- Trendelenburg position, oxygen,
IV fluids (Ringer lactate > normal saline > D5W).
––
Severe:
-- Adrenaline (1:1000) SC, 0.1 to
0.3 ml ; repeat up to 3 times (1 gm max).
·
For hypotension with bradycardia:
–– Trendelenburg position, oxygen.
–– Atropine 0.6 to 1 mg, slow IV.
––
IV fluids (Ringer lactate > normal saline > D5W).
·
For hypertension:
–– Apresoline 5 mg IV, or sodium
nitroprusside. –– Monitor ECG, pulse oximeter, BP.
·
YY For
convulsions:
–– Oxygen, 2 to 6 L/min. –– Diazepam
5 mg, IV.
––
Phenytoin, 15–18 mg/kg, infusion at 50 mg/min.
·
For pulmonary oedema:
–– Head elevation, rotating
tourniquets. –– Oxygen 2 to 6 L/min.
––
Furosemide 40 mg, slow IV.
––
Corticosteroids (optional).
Neurological side effects:
·
Convulsions must be treated with IV, diazepam,phenytoin,
phenobarbitone, intubation, and intensive medical care.
·
If accidental intrathecal injection of ionic contrastmedia
is suspected, the patient should not be permitted to lie down.
Pulmonary side effects:
·
Patients with a history of radiocontrast medium-related
oedema should be given prophylactic corticosteroids.
Renal side effects:
·
All patients undergoing examination involving contrast media
should be well hydrated, since dehydrationprecipitates renal dysfunction.
· Acute renal failure has to be managed by dialysis.
Toxic Effects
1) Accidental intrathecal
administration of ionic contrast agents must be treated with appropriate
circulatory support, intrathecal lavage, and anticonvulsant therapy.
2) To decrease the incidence of side
effects after myelography, the following have been suggested:
a)
Head elevation for 6 hours after the study.
b)
Avoidance of drugs that lower seizure threshold, e.g.
c)
phenothiazines, MAOIs, tricyclics, alcohol, etc.
3) Overdose with iopanoic acid can be
treated with IV fluids, alkalinisation of urine, and cholestyramine (said to be
a chelator of iopanoic acid). Cardiac and respiratory moni-toring are
mandatory.
4) General recommendations:
a)
Patients with pre-existing renal impairment should be given
0.45% saline IV, for 12 hours before and 12 hours after administration of
contrast agents.
b)
Low-osmolality contrast agents are preferable to high-
osmolality agents.
c)
Pre-treatment with corticosteroids and antihistamines (with
or without adrenaline), has been suggested to minimise reactions to contrast
agents.
Related Topics