Chapter: Modern Medical Toxicology: Miscellaneous Drugs and Poisons: Gastrointestinal and Endocrinal Drugs
Laxatives - Gastrointestinal Drugs

Laxatives
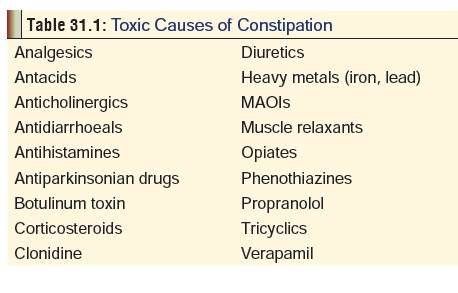
Laxatives are drugs which promote defaecation, and are widely employed in the treatment of constipation. Constipation can result not only from diseases of large intestine, nervous system, and endocrine system, but also from exposure to a number of drugs and toxins (Table 31.1). Laxatives only treat the symptom (constipation) but not the underlying cause. Hence every effort must be made to identify and treat the cause, rather than resort to the indiscriminate use of laxatives which can lead to serious repercussions (vide infra).
![]()
Classification
·
Bulk-forming laxatives
·
Saline and osmotic laxatives
·
Stimulant laxatives
·
Surfactant laxatives
·
Other laxatives : mineral oil,
cisapride.
Bulk-forming Laxatives
Functional constipation is best
treated by a diet rich in plant fibre. Bulk-forming laxatives act as
supplements to dietary fibre and include wheat bran, psyllium (Plantago afra or Plantagoindica), methylcellulose, carboxymethylcellulose, guar gum(Cyanopsis psoraloides), gum tragacanth (Astraplus gummifer), karaya (Sterculia species),
glucomannan (Konjac mannan), malt
soup extract, flaxseed (Linum
usitatissimum), isapghula (Plantago
ovata), and polycarbophil. These laxatives generally swell in water to form
a gel that serves to maintain soft, well-hydrated faeces. They also reflexively
stimulate peristalsis. The laxative effect is usually apparent within 12 to 24
hours and may not become fully apparent for as long as 72 hours.
Glucomannan is a polysaccharide
composed of glucose and mannose. Capsules of the fibre absorb up to 60 times
their weight in water to form a gel.
Systemic toxicity is unlikely since these agents are unab-sorbed from the gastrointestinal tract and excreted in the faeces. Adverse effects are rare and usually mild: flatulence, borbo-rygmi, intestinal impaction. Intestinal obstruction has been occasionally observed. It can be prevented by taking enough water concurrently with the laxative. Obstruction and impac-tions are usually associated with intestinal pathology or lack of adequate hydration. Glucomannan has resulted in decreased blood glucose and serum insulin levels when given in single doses to diabetic patients. Diabetics should be cautioned that decreased insulin or oral hypoglycaemic requirements may occur.
Anaphylactoid reactions have been reported following exposure to psyllium containing products. Moderate-to-severe wheezing, tightness in the chest or
throat, urticaria, and angi-oedema have been reported in some patients. The
seed husk of psyllium mucilloid contains a protein that appears to be the
source of the allergic reactions. Highly purified psyllium mucilloid may
decrease the allergenicity to psyllium-containing products.
Treatment
■■ Immediately
dilute with 4 to 8 ounces (120 to 240 ml) of water or milk (not to exceed 4
ounces or 120 ml in a child).
■■ Monitor fluid and
electrolyte status carefully in sympto-matic patients.
■■ Emesis and activated
charcoal are usually not necessary following ingestions.
■■ Restrict solid food
and maintain high fluid intake until diarrhoea resolves. Oral fluid should
consist of hypotonic solutions containing appropriate electrolytes.
■■ Rigid oesophagoscopy
has been used to relieve oesophageal obstruction.
■■ Mild
to moderate allergic reactions may be treated with antihistamines with or
without inhaled beta agonists, corti-costeroids or adrenaline. Treatment of
severe anaphylaxis also includes oxygen supplementation, aggressive airway
management, adrenaline, ECG monitoring and IV fluids.
Saline and Osmotic Laxatives
Saline laxatives are generally given
by mouth producing catharsis with soft or fluid stools. The citrates, sulfates,
and tartrates of sodium or potassium are the usual agents used: magnesium
sulfate (epsom salt), magnesium
hydroxide (milkof magnesia),
magnesium citrate, sodium phosphate, potas-sium bitartrate, potassium sodium
tartrate, potassium sulfate, sodium citrate, sodium sulfate, lactulose,
glycerine, sorbitol, mannitol, and polyethylene glycol electrolyte solution.
These agents act by their osmotic properties in the luminal fluid and are
poorly absorbed. However, many of the ions found in saline cathartics may be
absorbed from the gastrointestinal tract. Some of them are utilised as
cathartics in the treatment of poisoning.
Saline cathartics are poorly
absorbed from the gastrointes-tinal tract, hence systemic toxicity is unlikely
unless massive amounts have been ingested. Nausea, vomiting, abdominal pain,
and diarrhoea are frequent findings. Severe diarrhoea may result in excessive
fluid and electrolyte loss. Fluid and electrolyte abnormalities (dehydration
and hypokalaemia) occur secondary to excessive diarrhoea. A mild diuresis may
occur following excessive absorption of these compounds. Magnesium and sodium
salts can cause electrolyte distur-bances and must be used with caution in the
presence of renal impairment. Excessive absorption of sodium may aggravate
congestive heart failure. Seizures may occur with elevated serum sodium levels.
Lactulose can cause flatulence, cramps, nausea, hypernatraemia and
hypokalaemia.
Oedema following saline laxative
withdrawal is not uncommon. The presumed mechanism is persistent
hyperal-dosteronism resulting in sodium retention.
Treatment
·
Monitor serum potassium and sodium
concentrations in symptomatic patients following exposure to one of the saline
cathartics.
·
Fluid and electrolyte status should
be monitored at regular intervals.
·
Emesis is usually not necessary and
may worsen dehydra- tion and electrolyte imbalances after large ingestions; it
is not recommended.
·
Due to molecular size and ionic
dissociation, charcoal is not likely to be beneficial.
·
Restrict solid food and maintain
high fluid intake until diarrhoea resolves. Oral fluids should consist of
hypotonic
·
solution containing appropriate
electrolytes.
·
Patients developing congestive heart
failure from sodium intoxication may be treated with fluid restriction and/or
diuretic therapy, furosemide 1 mg/kg IV to a maximum of 40 mg.
Stimulant Laxatives
These
may be either diphenylmethane derivatives (phenol-phthalein, bisacodyl), or
anthraquinones (danthron, i.e. dihy-droxyanthraquinone, and its derivatives
contained in senna, cascara, rhubarb, and aloe) . Senna is obtained from the
dried pods or leaflets of Cassia
acutifolia or Cassia angustifolia
(Fig 31.1). Cascara sagrada (‘sacred
bark’) is obtained from the bark of the buckthorn tree Rhamnus purshiana (Fig 31.2).
Danthron was removed from the market in the United States in 1987 because of
fears of carcinogenicity. As of August 1997, the FDA has also proposed a ban on
phenolphthalein, based on animal carcinogen data and its potential risk to
humans.


All these drugs stimulate intestinal motility by promoting the accumulation of water and electrolytes in the colonic lumen.
Adverse and Toxic Effects
The therapeutic dose of
diphenylmethane derivatives varies widely between different individuals, and
therefore a particular dose can be ineffective in one, while being excessive in
another.
·
Phenolphthalein discolours urine (and even faeces) pink or
red.
·
Gastric irritation is common with diphenylmethane deriva-
tives.
·
Phenolphthalein use is associated with allergic reactions
(including Stevens-Johnson syndrome), osteomalacia, and protein-losing
gastro-enteropathy. Hypersensitivity reactions may lead to death. Hypotension
and pulmonary oedema, although rare and probably secondary to an allergic
response, have been reported following phenolphthalein intoxication.
·
All stimulant laxatives suffer from a delayed onset of
action (6 to 10 hours).
·
Anthraquinones can cause yellowish or brownish coloured
urine. Large doses induce nephritis. Melanosis coli (pigmentation of colonic
mucosa) has been observed on long-term use.
·
Hepatitis and jaundice have been seen with various stimu-
lant laxatives. Chronic abuse of senna has caused toxic hepatitis.
·
Orange vaginal secretions have been reported after use of
large amounts of danthron.
Treatment
■■ Monitor fluid and
electrolyte balance in severely sympto-matic patients.
■■ All persons with
significant toxicity, dehydration, abnormal electrolyte levels, or a history of
poor compli-ance should be admitted for intravenous therapy and consideration
of antibiotic therapy.
■■ Activated charcoal has been shown to reduce the absorp-tion
of phenolphthalein in humans and to decrease the purgative effect.
■■ Restrict solid food
and maintain high fluid intake until diarrhoea resolves. Oral fluid should
consist of polyionic hypotonic solution containing appropriate electrolytes.
Patients with moderate to severe dehydration must be treated with IV fluids.![]()
Surfactant Laxatives
Examples include docusates (docusate
sodium, calcium, and potassium)*, poloxamers (polyoxyethylene-polyoxy-propylene
polymers), bile acids (dehydrocholic acid), and castor oil. Surfactant
laxatives act as stool-wetting and stool-softening agents, allowing the mixing
of water, fats, and faecal material. Docusates elevate alveolar surface tension
by displacement of pulmonary surfactant from the alveolar hypophase. There is
then a marked fall in arterial oxygen tension, an increase in airway pressure,
and an increased alveolar epithelial permeability. Stool softener laxatives are
indicated prophylactically in patients who should not strain during
defaecation, such as those with an episiotomy wound, those with thrombosed
haemorrhoids, fissures, or perianal abscesses, body wall and diaphragmatic
hernias, anorectal stenosis, or postmyocardial infarction.
Docusates while being minimally
effective laxatives, produce only mild side effects—cramps, nausea, skin rash.
Castor oil obtained from the seeds
of the castor plant (Ricinuscommunis)
contains ricin, a very potent toxalbumen, and ricinoleicacid. The oil is a very
powerful and dangerous laxative capable of inducing copious evacuation which
can result in dehydration and electrolyte disturbances. It can reflexly
stimulate uterine musculature and hence is contraindicated in pregnant women.
Mineral Oil
Mineral
oil is a petroleum product and consists of a mixture of aliphatic hydrocarbons.
It is categorised as a lubricant laxative. While being an effective laxative
(penetrating and softening stools), routine use is not advocated owing to the
following adverse effects:
·
Foreign body reaction in intestinal
mucosa.
·
Leakage of oil past the anal
sphincter with rectal irritation. Malabsorption of fat soluble vitamins
(chronic use). Mineral oil acts as a lipid solvent and administration with
meals may interfere with absorption of essential fat-soluble substances.
·
Aspiration can lead to lipid
pneumonitis.
·
A human teratogen by inhalation,
mineral oil has caused testicular tumours in the foetus.
Treatment
·
Monitor for respiratory distress after ingestion of mineral
oil. Monitor ABGs/pulse oximetry and chest radiographs in patients with
respiratory distress.
·
Due to aspiration hazard and generally low toxicity of these
compounds, gastric decontamination is contraindicated.
·
Fluid and electrolyte status should be monitored in patients
demonstrating severe vomiting and diarrhoea.
·
![]() Extracorporeal membrane oxygenation
(ECMO) (a pulmo-nary bypass procedure used in cases of reversible acute
pulmonary and cardiovascular failure) has been successful in the therapy of
paediatric aspiration involving mineral oil or mineral seal oil found initially
unresponsive to standard therapy for hydrocarbon aspiration.
Extracorporeal membrane oxygenation
(ECMO) (a pulmo-nary bypass procedure used in cases of reversible acute
pulmonary and cardiovascular failure) has been successful in the therapy of
paediatric aspiration involving mineral oil or mineral seal oil found initially
unresponsive to standard therapy for hydrocarbon aspiration.
Cisapride
Cisapride and renzapride (a related
drug) are oral gastrointes-tinal prokinetic agents that stimulate
gastrointestinal motility and are used in the treatment of gastroesophageal
reflux disease. Cisapride is a benzamide, and is mainly employed in the
treatment of gastric retention and other gastroparetic condi-tions. Since it
also increases colonic motility, it is sometimes recommended as a laxative,
especially for chronic constipa-tion. Renzapride has been shown in clinical
trials to be useful in the treatment of diabetic gastroparesis and
constipation-predominant irritable bowel syndrome. In 2000, cisapride was
withdrawn from the market in both the United States and the United Kingdom due
to increasing numbers of reported adverse events of heart rhythm abnormalities.
Cisapride increases the release of
acetylcholine at the myenteric plexus level of the gastrointestinal tract,
increases the lower oesophageal resting tone, and increases the ampli-tude of
lower oesophageal contractions. Gastrointestinal motility is then stimulated,
gastric emptying is accelerated, and colonic peristalsis is increased.
Renzapride is a type 3 serotonin antagonist and a type 4 serotonin agonist.
Following a therapeutic dose, renzapride blocks serotonin 3 receptors and
stimulates serotonin 4 receptors; it does not exhibit dopamine D(2) receptor
antagonism. It also has been shown to stimulate gastric acid secretion during
low-dose pentagastrin infusion, and block secretion during high-dose
pentagastrin stimula-tion. Due to this interchange of mechanisms, this drug
lowers oesophageal sphincter pressure and increases gastric emptying.
Cisapride should be taken prior to a
meal, since food increases its absorption. Adverse effects include headache,
vertigo, abdominal cramps, borborygmi, gastrointestinal distress, urinary
frequency. Tachycardia has been reported. ECG abnormalities, QT prolongation,
and arrhythmias including torsades de pointes have also been reported.
Akathisias have also occurred rarely. Urinary incontinence and vaginitis have
developed in a few patients.
Overdose experience with cisapride and
renzapride are limited. Retching, borborygmi, flatulence, stool frequency, and
urinary frequency have been reported. Drugs that inhibit the cytochrome P450
3A4 enzymes may increase blood levels of cisapride, resulting in cardiac
arrhythmias. Concurrent use of erythromycin, clarithromycin, troleandomycin,
nefazodone, indinavir, ritonavir, and azole antifungals can induce cardiac
arrhythmias. Drugs known to prolong the QT interval and increase the risk of
arrhythmia should also not be taken concur-rently with cisapride. Since
cisapride increases gastric emptying and rate of absorption, the effect of
sedative drugs and alcohol may be significantly pronounced.
Treatment
·
Monitor electrolyte levels if the
patient experiences severe and prolonged diarrhoea.
·
Obtain an ECG and institute
continuous cardiac monitoring for possible QT prolongation and arrhythmias.
·
Activated charcoal may be
administered.
·
For torsades de pointes: Withdraw
the causative agent. Haemodynamically unstable patients require electrical
cardioversion. Emergent treatment with magnesium sulfate, isoproterenol, or
atrial overdrive pacing is indicated. Detect and correct underlying electrolyte
abnormalities (hypomag- nesaemia, hypokalaemia, hypocalcaemia).
·
Despite its prolongation of the QT
interval, amiodarone has been reported to be effective in treating both acute
episodes of torsades de pointes, as well as preventing recurrences.
·
Avoid class Ia antiarrhythmics
(quinidine, disopyramide, procainamide, aprindine) and most class III
antiarrhythmics (N- acetylprocainamide, sotalol) since they may further prolong
the QT interval, and have been associated with torsades de pointes.
·
Mexiletine may be useful in the
treatment of cisapride- induced long QT syndrome. Caution should however be
observed in the presence of pre-existing impaired cardiac function, since
mexiletine could suppress the ventricular contraction together with a decrease
of cardiac output, leading to potential cardiovascular collapse.
·
Sinus tachyarrhythmias do not need
to be routinely treated unless the patient demonstrates signs and/or symptoms
of haemodynamic instability. In those cases, tachyarrhythmias may respond to IV
esmolol.
Related Topics