Chapter: Medical Surgical Nursing: Management of Patients With Chest and Lower Respiratory Tract Disorders
Pulmonary Tuberculosis
PULMONARY TUBERCULOSIS
Tuberculosis
(TB) is an infectious disease that primarily affects the lung parenchyma. It
also may be transmitted to other parts of the body, including the meninges,
kidneys, bones, and lymph nodes. The primary infectious agent, Mycobacterium tuberculosis, is an
acid-fast aerobic rod that grows slowly and is sensitive to heat and
ultraviolet light. Mycobacterium bovis
and Mycobac-terium avium have rarely
been associated with the developmentof a TB infection.
TB
is a worldwide public health problem, and the mortality and morbidity rates
continue to rise. M. tuberculosis
infects an es-timated one third of the world’s population and remains the
lead-ing cause of death from infectious disease in the world. It is the leading
cause of death among HIV-positive people (World Health Organization, 2000). TB
is closely associated with poverty, mal-nutrition, overcrowding, substandard
housing, and inadequate health care.
In
1952, anti-TB medications were introduced, and the rate of reported cases of TB
in the United States declined an average of 6% each year between 1953 and 1985.
It was thought that by the early part of the 21st century, TB might be
eliminated in the United States. However, since 1985 the trend has reversed and
the number of cases has increased. This change has been attrib-uted to several
factors, including increased immigration, the HIV epidemic, the emergence of
multidrug-resistant strains of TB, in-creased homelessness, decreased interest
and detection by health care providers, and inadequate funding of the U.S.
public health system (Small & Fujiwara, 2001).
Transmission and Risk Factors
TB spreads from person to person by
airborne transmission. An infected person releases droplet nuclei (generally
particles 1 to 5 micrometers in diameter) through talking, coughing, sneezing,
laughing, or singing. Larger droplets settle; smaller droplets re-main
suspended in the air and are inhaled by the susceptible per-son. Risk factors
for TB are listed in Chart 23-4. Chart 23-5 summarizes the CDC’s recommendations
for prevention of TB transmission in health care settings.


Pathophysiology
A
susceptible person inhales mycobacterium bacilli and becomes infected. The
bacteria are transmitted through the airways to the alveoli, where they are
deposited and begin to multiply. The bacilli also are transported via the lymph
system and bloodstream to other parts of the body (kidneys, bones, cerebral
cortex) and other areas of the lungs (upper lobes). The body’s immune sys-tem
responds by initiating an inflammatory reaction. Phagocytes (neutrophils and
macrophages) engulf many of the bacteria, and TB-specific lymphocytes lyse
(destroy) the bacilli and normal tis-sue. This tissue reaction results in the
accumulation of exudate in the alveoli, causing bronchopneumonia. The initial
infection usually occurs 2 to 10 weeks after exposure.
Granulomas,
new tissue masses of live and dead bacilli, are sur-rounded by macrophages,
which form a protective wall around the granulomas. Granulomas are then
transformed to a fibrous tissue mass, the central portion of which is called a
Ghon tubercle. The material (bacteria and macrophages) becomes necrotic,
form-ing a cheesy mass. This mass may become calcified and form a collagenous scar.
At this point, the bacteria become dormant, and there is no further progression
of active disease.
After
initial exposure and infection, the person may develop active disease because
of a compromised or inadequate immune system response. Active disease also may
occur with reinfection and activation of dormant bacteria. In this case, the
Ghon tuber-cle ulcerates, releasing the cheesy material into the bronchi. The
bacteria then become airborne, resulting in further spread of the disease. Then
the ulcerated tubercle heals and forms scar tissue. This causes the infected
lung to become more inflamed, resulting in further development of
bronchopneumonia and tubercle formation.
Unless the process is arrested, it spreads slowly downward to the hilum of the lungs and later extends to adjacent lobes. The process may be prolonged and characterized by long remissions when the disease is arrested, only to be followed by periods of re-newed activity. Approximately 10% of people who are initially infected develop active disease. Some people develop reactivation TB (also called adult-type TB). This type of TB results from a breakdown of the host defenses. It most commonly occurs within the lungs, usually in the apical or posterior segments of the upper lobes, or the superior segments of the lower lobes.
Clinical Manifestations
The
signs and symptoms of pulmonary TB are insidious. Most patients have a
low-grade fever, cough, night sweats, fatigue, and weight loss. The cough may
be nonproductive, or mucopurulent sputum may be expectorated. Hemoptysis also
may occur. Both the systemic and pulmonary symptoms are usually chronic and may
have been present for weeks to months. The elderly usually present with less
pronounced symptoms than do younger patients. Extrapulmonary disease occurs in
up to 16% of cases in the United States. In patients with AIDS, extrapulmonary
disease is more prevalent and may occur in up to 70% of cases (Niederman &
Sarosi, 2000; Small & Fujiwara, 2001).
Assessment and Diagnostic Findings
A
complete history, physical examination, tuberculin skin test, chest x-ray,
acid-fast bacillus smear, and sputum culture are used to diagnose TB. If the
person is infected with TB, the chest x-ray usually reveals lesions in the upper
lobes and the acid-fast bacil-lus smear contains mycobacterium.
TUBERCULIN SKIN TEST
The
Mantoux test is used to determine if a person has been in-fected with the TB
bacillus. The Mantoux test is a standardized procedure and should be performed
only by those trained in its administration and reading. Tubercle bacillus
extract (tuber-culin), purified protein derivative (PPD), is injected into the
in-tradermal layer of the inner aspect of the forearm, approximately 4 inches
below the elbow (Fig. 23-4). Intermediate-strength (5 TU) PPD in a tuberculin
syringe with a half-inch 26- or 27-gauge needle is used. The needle, with the
bevel facing up, is inserted be-neath the skin. Then 0.1 mL of PPD is injected,
creating an ele-vation in the skin, a wheal or bleb. The site, antigen name,
strength, lot number, date, and time of the test are recorded. The test result
is read 48 to 72 hours after injection. Tests read after 72 hours tend to
underestimate the true size of induration
(hard-ening). A delayed localized reaction indicates that the person is
sensitive to tuberculin.
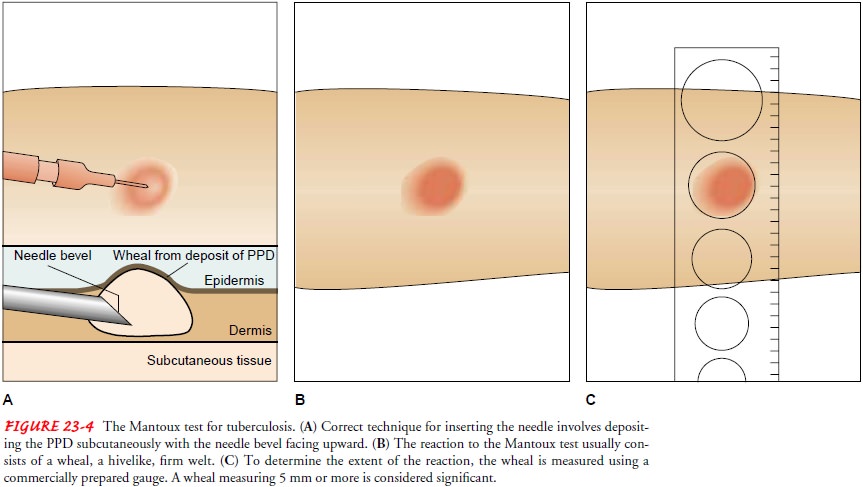
A
reaction occurs when both induration and erythema (red-ness) are noted. After
the area is inspected for induration, it is lightly palpated across the
injection site, from the area of normal skin to the margins of the induration.
The diameter of the in-duration (not erythema) is measured in millimeters at
its widest part (see Fig. 23-4), and the size of the induration is documented.
Erythema without induration is not considered significant.
Interpretation of Results.
The size of the induration deter-mines the significance of the
reaction. A reaction of 0 to 4 mm is considered not significant; a reaction of
5 mm or greater may be significant in individuals who are considered at risk.
An in-duration of 10 mm or greater is usually considered significant in
individuals who have normal or mildly impaired immunity. A significant reaction
indicates that a patient has been exposed to M. tuberculosis recently or in the past or has been vaccinated with
bacille Calmette-Guerin (BCG) vaccine. The BCG vac-cine is given to produce a
greater resistance to developing TB. It is effective in up to 76% of those who
receive it. The vaccine is used in Europe and Latin America but not routinely
in the United States.
A reaction of 5 mm or greater is defined as positive for patients who are HIV-positive or have HIV risk factors and are of un-known HIV status, those who are close contacts with an active case, and those who have chest x-ray results consistent with tuberculosis.
A
significant (positive) reaction does not necessarily mean that active disease
is present in the body. Most (more than 90%) peo-ple who are
tuberculin-significant reactors do not develop clini-cal TB. However, all
significant reactors are candidates for active TB. In general, the more intense
the reaction, the greater the like-lihood of an active infection.
A
nonsignificant (negative) skin test does not exclude TB in-fection or disease
because patients who are immunosuppressed cannot develop an immune response
adequate to produce a pos-itive skin test. This is referred to as anergy.
The
accuracy of the skin test depends on the skill of the person interpreting the
test reaction. One study (Kendig, Kirkpatrick, Carter et al., 1998) revealed
that health care professionals tend to underestimate the size of induration:
only 7% of a sample of 107 health care providers charted the correct size of
induration.
CLASSIFICATION OF TB
Data
from the history, physical examination, skin test, chest x-ray, and
microbiologic studies are used to classify TB into one of five classes. A
classification scheme provides public health officials with a systematic way to
monitor epidemiology and treatment of the disease (American Thoracic Society,
2000).
• Class 0: no exposure; no infection
• Class 1: exposure; no evidence of infection
• Class 2: latent infection; no disease (eg,
positive PPD reac-tion but no clinical evidence of active TB)
• Class 3: disease; clinically active
• Class 4: disease; not clinically active
• Class 5: suspected disease; diagnosis
pending
Gerontologic Considerations
TB
may have atypical manifestations in elderly patients, whose symptoms may
include unusual behavior and altered mental sta-tus, fever, anorexia, and
weight loss. Many elderly patients may have no reaction (loss of immunologic
memory) or delayed reac-tivity for up to a week (recall phenomenon). A second
skin test is performed in 1 to 2 weeks.
Medical Management
Pulmonary
TB is treated primarily with chemotherapeutic agents (antituberculosis agents)
for 6 to 12 months. A prolonged treat-ment duration is necessary to ensure
eradication of the organ-isms and to prevent relapse. A worldwide concern and
challenge in TB therapy is the continuing (since the 1950s) and increasing
resistance of M. tuberculosis to TB
medications. Several types of drug resistance must be considered when planning
effective therapy:
·
Primary drug resistance: resistance
to one of the first-line antituberculosis agents in a person who has not had
previ-ous treatment
·
Secondary or acquired drug
resistance: resistance to one or more antituberculosis agents in a patient
undergoing therapy
·
Multidrug resistance: resistance to
two agents, isoniazid (INH) and rifampin. The populations at highest risk for
multidrug resistance are those who are HIV-positive, insti-tutionalized, or
homeless.
The
increasing prevalence of drug resistance points out the need to begin TB
treatment with four or more medications, to ensure completion of therapy, and
to develop and evaluate new anti-TB medications.
PHARMACOLOGIC THERAPY
In
current TB therapy, five first-line medications are used (Table 23-2): INH,
rifampin, pyrazinamide, and either strep-tomycin or ethambutol.
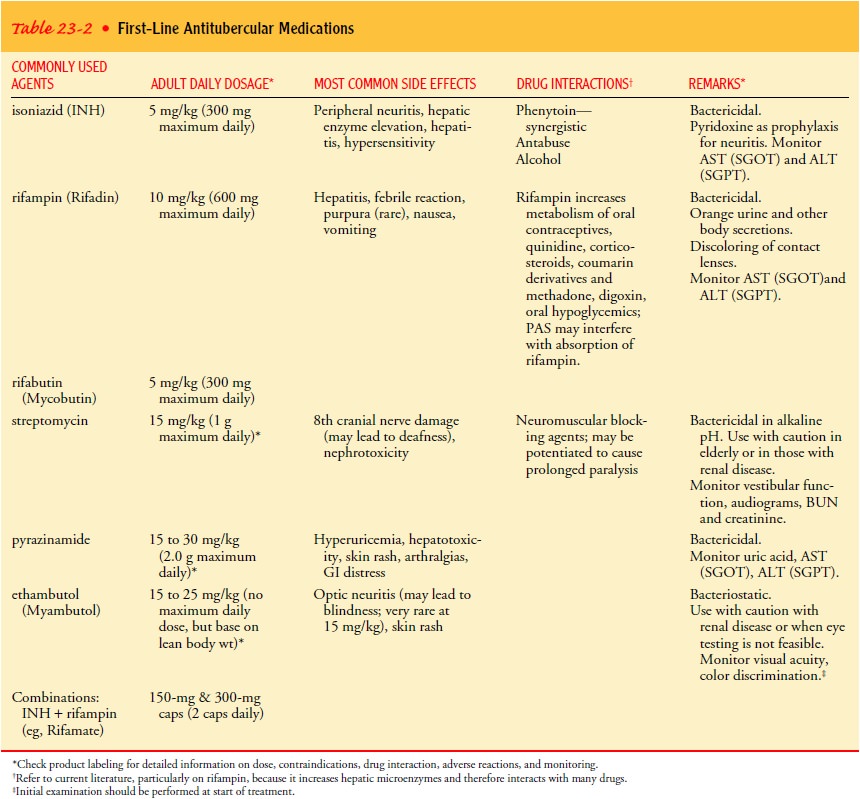
Combination
medications, such as INH and rifampin (Rifa-mate) or INH, pyrazinamide and
rifampin and medications ad-ministered twice a week (eg, rifapentine) are
available to help improve patient adherence. Capreomycin, ethionamide,
para-aminosalicylate sodium, and cycloserine are second-line medica-tions.
Additional potentially effective medications include other aminoglycosides,
quinolones, rifabutin, clofazimine, and combi-nations of medications.
Recommended
treatment guidelines for newly diagnosed cases of pulmonary TB (CDC, 2000) consist
of a multiple-medication regimen of INH, rifampin, pyrazinamide, and either
streptomycin or ethambutol. This initial intensive-treatment regimen is usually
administered daily for 8 weeks. If cultures demonstrate that the organism is
sensitive to the medications before the 8 weeks of ther-apy have been
completed, either ethambutol or streptomycin can be discontinued. After 8 weeks
of this medication regimen, pyra-zinamide can be discontinued and INH and
rifampin are admin-istered for an additional 4 months. The medication regimen,
however, may continue for 12 months. A person is considered noninfectious after
2 to 3 weeks of continuous medication ther-apy. Vitamin B (pyridoxine) is
usually administered with INH to prevent INH-associated peripheral neuropathy
(see Table 23-2).
INH
also may be used as a prophylactic (preventive) measure for those at risk for
significant disease, including:
·
Household family members of patients
with active disease
·
HIV-infected patients with a PPD
test reaction of 5 mm of induration or more
·
Patients with fibrotic lesions
detected on a chest x-ray, sug-gestive of old TB, and a PPD reaction of 5 mm of
indura-tion or more
·
Patients whose current PPD test
results show a change from former test results, suggesting recent exposure to
TB and possible infection (also called skin test converters)
·
Drug (intravenous or injectable)
users with PPD test results of 10 mm of induration or more
·
Patients with high-risk comorbid
conditions with a PPD result of 10 mm of induration or more
Other
candidates for preventive INH therapy are those age 35 years or younger with
PPD test results of 10 mm of indura-tion or more and one of the following
criteria:
·
Foreign-born individuals from
countries with a high preva-lence of TB
·
High-risk, medically underserved
populations
·
Institutionalized patients
Prophylactic
INH treatment involves taking daily doses for 6 to 12 months. Liver enzyme,
blood urea nitrogen, and creati-nine levels are monitored monthly. Sputum
culture results are monitored for acid-fast bacillus to evaluate the
effectiveness of treatment and the patient’s compliance with therapy.
In 1998, the federal Advisory Council for the Elimination of Tuberculosis published recommendations for the development of TB vaccines. The recommendations include a focus on a “postinfection vaccine” to prevent people infected with TB from developing active disease (CDC, 1998).
To date, this vaccine has not
become clinically available. In 2000, recommendations were released regarding
the treatment of latent TB infection (Ameri-can Thoracic Society and CDC,
2000). Isoniazid (INH) for 6 to 12 months has been the mainstay of treatment
for latent TB in-fection. However, this long duration of treatment has been
limited due to poor adherence and concerns of toxicity. The American Thoracic
Society and CDC released newer guidelines in the 2000 document, which focused
on treating a latent infection over a shorter period of time. The CDC released
case reports of liver injury associated with the 2-month rifampin-pyrazinamide
(RIF-PZA) dosing regimen in August 2001 (MMWR, 2001). This prompted a review
and changes to the 2000 guidelines. In sum-mary, a 2-month RIF-PZA treatment
regimen for latent TB in-fection should be used with caution, especially in
patients who are concurrently taking medications for liver disease or those
with a history of alcoholism. For patients not infected with HIV, 9 months of
daily INH remains the preferred treatment, and 4 months of daily RIF is an acceptable
alternative. No more than a 2-week supply of RIF-PZA should be dispensed at any
one time to facilitate periodic clinical assessments. Lastly, serum
aminotrans-ferase and bilirubin should be measured at baseline and at 2, 4, and
6 weeks of treatment in patients taking RIF-PZA (MMWR, 2001).
Related Topics