Chapter: Medical Surgical Nursing: Management of Patients With Chest and Lower Respiratory Tract Disorders
Pulmonary Embolism
Pulmonary Embolism
Pulmonary embolism (PE)
refers to the obstruction of the pul-monary artery or one of its branches by a
thrombus (or thrombi) that originates somewhere in the venous system or in the
right side of the heart. Most commonly, PE is due to a blood clot or throm-bus.
However, there are other types of emboli: air, fat, amniotic fluid, and septic
(from bacterial invasion of the thrombus). It is es-timated that more than half
a million people develop PE yearly, resulting in more than 50,000 deaths. PE is
a common disorder and often is associated with trauma, surgery (orthopedic,
major abdominal, pelvic, gynecologic), pregnancy, heart failure, age older than
50 years, hypercoagulable states, and prolonged immobility. It also may occur
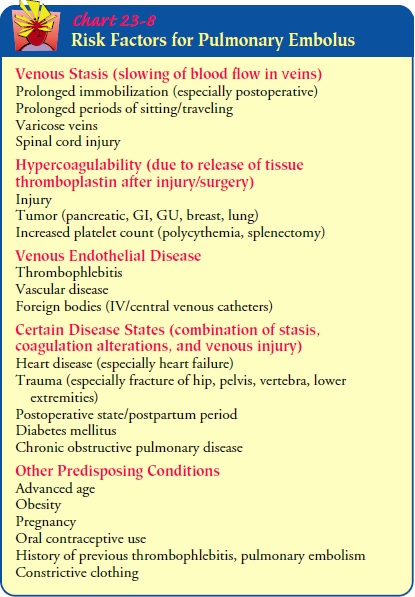
in an apparently healthy person. Risk factors for developing PE are identified
in Chart 23-8.
Although
most thrombi originate in the deep veins of the legs, other sites include the
pelvic veins and the right atrium of the heart. A venous thrombosis can result
from slowing of blood flow (stasis), secondary to damage to the blood vessel
wall (particularly the endothelial lining) or changes in the blood coagulation
mech-anism. Atrial fibrillation is also a cause of pulmonary embolism. An
enlarged right atrium in fibrillation causes blood to stagnate and form clots
in this area. These clots are prone to travel into the pulmonary circulation.
Pathophysiology
When
a thrombus completely or partially obstructs a pulmonary artery or its
branches, the alveolar dead space is increased. The area, although continuing
to be ventilated, receives little or no blood flow. Thus, gas exchange is
impaired or absent in this area. In addition, various substances are released
from the clot and sur-rounding area, causing regional blood vessels and
bronchioles to constrict. This causes an increase in pulmonary vascular
resis-tance. This reaction compounds the ventilation–perfusion imbalance.
The
hemodynamic consequences are increased pulmonary vascular resistance from the
regional vasoconstriction and re-duced size of the pulmonary vascular bed. This
results in an in-crease in pulmonary arterial pressure and, in turn, an
increase in right ventricular work to maintain pulmonary blood flow. When the
work requirements of the right ventricle exceed its capacity, right ventricular
failure occurs, leading to a decrease in cardiac output followed by a decrease
in systemic blood pressure and the development of shock.
Clinical Manifestations
The
symptoms of PE depend on the size of the thrombus and the area of the pulmonary
artery occluded by the thrombus; they may be nonspecific. Dyspnea is the most
frequent symptom; tachypnea (very rapid respiratory rate) is the most frequent
sign (Goldhaber, 1998). The duration and intensity of the dyspnea depend on the
extent of embolization. Chest pain is common and is usually sudden and
pleuritic. It may be substernal and mimic angina pectoris or a myocardial infarction.
Other symptoms include anxiety, fever, tachycardia, apprehension, cough,
diaphoresis, hemoptysis, and syncope.
A
massive embolism is best defined by the degree of hemody-namic instability
rather than the percentage of pulmonary vascu-lature occlusion. It is described
as an occlusion of the outflow tract of the main pulmonary artery or the
bifurcation of the pul-monary arteries that produces pronounced dyspnea, sudden
sub-sternal pain, rapid and weak pulse, shock, syncope, and sudden death.
Multiple small emboli can lodge in the terminal pul-monary arterioles,
producing multiple small infarctions of the lungs. A pulmonary infarction
causes ischemic necrosis of an area of the lung and occurs in less than 10% of
cases of PE (Arroliga, Matthay & Matthay, 2000). The clinical picture may
mimic that of bronchopneumonia or heart failure. In atypical instances, the
disease causes few signs and symptoms, whereas in other instances it mimics
various other cardiopulmonary disorders.
Assessment and Diagnostic Findings
Death
from PE commonly occurs within 1 hour of symptoms; thus, early recognition and
diagnosis are priorities. Because the symptoms of PE can vary from few to
severe, a diagnostic workup is performed to rule out other diseases. Deep
venous thrombosis is closely associated with the development of PE. Typically,
pa-tients report sudden onset of pain and/or swelling and warmth of the
proximal or distal extremity, skin discoloration, and super-ficial vein
distention. The pain is usually relieved with elevation. The diagnostic workup
includes a ventilation–perfusion scan, pulmonary angiography, chest x-ray, ECG,
peripheral vascular studies, impedance plethysmography, and arterial blood gas
analysis.The chest x-ray is usually normal but may show infiltrates,
at-electasis, elevation of the diaphragm on the affected side, or a pleural
effusion. The chest x-ray is most helpful in excluding other possible causes.
The ECG usually shows sinus tachycardia, PR-interval depression, and
nonspecific T-wave changes. Peripheral vascular studies may include impedance
plethysmography, Doppler ultrasonography, or venography. Test results con-firm
or exclude the diagnosis of PE. Arterial blood gas analysis may show hypoxemia
and hypocapnia (from tachypnea); however, ar-terial blood gas measurements are
normal in up to 20% of patients with PE.
A
ventilation–perfusion scan is the test of choice in patients with suspected PE.
The perfusion portion of the scan may indi-cate areas of diminished or absent
blood flow and is the most use-ful test to rule out clinically important PE. A
ventilation scan may show whether there is also a ventilation abnormality
present. A normal perfusion scan rules out the diagnosis of PE. If there is a
ventilation–perfusion mismatch, the probability of PE is high. Spiral CT of the
chest may also assist in the diagnosis.
If lung scan results are not definitive, pulmonary angiography, considered the gold standard for the diagnosis of PE, can be used. This test is invasive and is performed in the interventional radiol-ogy department. A contrast agent is injected into the pulmonary arterial system, allowing visualization of obstructions to blood flow and abnormalities.
Prevention
For
those at risk, the most effective approach to preventing PE is to prevent deep
venous thrombosis. Active leg exercises to avoid venous stasis, early
ambulation, and use of elastic compression stockings are general preventive
measures. Additional strategies for prevention are listed in the checklist in
Chart 23-9..

Patients
who are older than 40, whose hemostasis is adequate, and who are undergoing
major elective abdominal or thoracic surgery may receive anticoagulant therapy.
Low doses of heparin may be given before surgery to reduce the risk of postoperative
deep venous thrombus and PE. Heparin should be administered subcutaneously 2
hours before surgery and continued every 8 to 12 hours until the patient is
discharged. Low-dose heparin is thought to enhance the activity of antithrombin
III, a major plasma inhibitor of clotting factor X. This regimen is not
recom-mended for patients with an active thrombotic process or for those
undergoing major orthopedic surgery, open prostatectomy, or surgery on the eye
or brain. Low-molecular-weight heparin (eg, enoxaparin [Lovenox]) is an
alternative therapy. It has a longer half-life, enhanced subcutaneous
absorption, a reduced in-cidence of thrombocytopenia, and reduced interaction
with platelets as compared to unfractionated heparin (Ansell, Hickey, Kleinschmidt
et al., 2000).
The
intermittent pneumatic leg compression device is useful in preventing
thromboembolism. The device inflates a bag that in-termittently compresses the
leg from the calf to the thigh, thereby improving venous return. It may be
applied before surgery and continued until the patient is ambulatory. The
device is particu-larly useful for patients who are not candidates for
anticoagulant therapy (Clagett, Anderson, Geerts et al., 1998).
Medical Management
Because
PE is often a medical emergency, emergency management is of primary concern.
After emergency measures have been taken and the patient’s condition
stabilizes, the treatment goal is to dis-solve (lyse) the existing emboli and
prevent new ones from form-ing. The treatment of PE may include a variety of
modalities:
General measures to improve
respiratory and vascular status
·
Anticoagulation therapy
·
Thrombolytic therapy
·
Surgical intervention
EMERGENCY MANAGEMENT
Massive
PE is a life-threatening emergency. The immediate ob-jective is to stabilize
the cardiopulmonary system. A sudden rise in pulmonary resistance increases the
work of the right ventricle, which can cause acute right-sided heart failure
with cardiogenic shock. Most patients who die of massive PE do so in the first
1 to 2 hours after the embolic event. Emergency management consists of the
following:
·
Nasal oxygen is administered
immediately to relieve hy-poxemia, respiratory distress, and central cyanosis.
·
Intravenous infusion lines are
started to establish routes for medications or fluids that will be needed.
·
A perfusion scan, hemodynamic
measurements, and arte-rial blood gas determinations are performed. Spiral
(heli-cal) CT or pulmonary angiography may be performed. Spiral CT is more
advanced and quicker than routine to-mography. With spiral CT, the patient
continuously moves as the x-ray tube rotates. With this type of CT, im-ages can
be reconstructed at select levels and locations for diagnostic purposes.
·
Hypotension is treated by a slow
infusion of dobutamine (Dobutrex) (which has a dilating effect on the pulmonary
vessels and bronchi) or dopamine (Intropin).
·
The ECG is monitored continuously for
dysrhythmias and right ventricular failure, which may occur suddenly.
·
Digitalis glycosides, intravenous
diuretics, and antiarrhyth-mic agents are administered when appropriate.
·
Blood is drawn for serum
electrolytes, complete blood count, and hematocrit.
·
If clinical assessment and arterial
blood gas analysis indicate the need, the patient is intubated and placed on a
mechanical ventilator.
·
If the patient has suffered massive
embolism and is hypo-tensive, an indwelling urinary catheter is inserted to
moni-tor urinary output.
·
Small doses of intravenous morphine
or sedatives are ad-ministered to relieve the patient’s anxiety, to alleviate
chest discomfort, to improve tolerance of the endotracheal tube, and to ease
adaptation to the mechanical ventilator.
GENERAL MANAGEMENT
Measures
are initiated to improve the patient’s respiratory and vascular status. Oxygen
therapy is administered to correct the hy-poxemia, relieve the pulmonary
vascular vasoconstriction, and re-duce the pulmonary hypertension. Using
elastic compression stockings or intermittent pneumatic leg compression devices
re-duces venous stasis. These measures compress the superficial veins and
increase the velocity of blood in the deep veins by redirect-ing the blood
through the deep veins. Elevating the leg (above the level of the heart) also
increases venous flow.
PHARMACOLOGIC THERAPY
Anticoagulation Therapy.
Anticoagulant therapy (heparin, war-farin sodium) has
traditionally been the primary method for managing acute deep vein thrombosis
and PE (Goldhaber, 1998). Heparin is used to prevent recurrence of emboli but
has no effect on emboli that are already present. It is administered as an
intravenous bolus of 5,000 to 10,000 units, followed by a con-tinuous infusion
initiated at a dose of 18 U/kg per hour, not to exceed 1,600 U/hour in
otherwise healthy patients (Goldhaber, 1998). The rate is reduced in patients
with a high risk of bleed-ing. The goal is to keep the partial thromboplastin
time 1.5 to 2.5 times normal (or 46 to 70 seconds). Heparin is usually
admin-istered for 5 to 7 days. Low-molecular-weight heparin (eg, enoxa-parin
[Lovenox]) may also be used.
Warfarin
sodium (Coumadin) administration is begun within 24 hours after the start of
heparin therapy because its onset of action is 4 to 5 days. Warfarin is usually
continued for 3 to 6 months. The prothrombin time is maintained at 1.5 to 2.5
times normal (or an INR [international normalized ratio] of 2.0 to 3.0).
Anticoagulation therapy is contraindicated in patients who are at risk for
bleeding (eg, those with gastro-intestinal conditions or with postoperative or
postpartum bleeding).
Thrombolytic Therapy.
Thrombolytic
therapy (urokinase, strepto-kinase, alteplase, anistreplase, reteplase) also
may be used in treating PE, particularly in patients who are severely
com-promised (eg, those who are hypotensive and have significant hypoxemia
despite oxygen supplementation). Thrombolytic therapy resolves the thrombi or
emboli more quickly and re-stores more normal hemodynamic functioning of the
pul-monary circulation, thereby reducing pulmonary hypertension and
improving perfusion, oxygenation, and cardiac output. Bleeding, however, is a
significant side effect. Contraindications to thrombolytic therapy include a
cerebrovascular accident within the past 2 months, other active intracranial
processes, ac-tive bleeding, surgery within the past 10 days of the thrombotic
event, recent labor and delivery, trauma, or severe hypertension. Consequently,
thrombolytic agents are advocated only for PE affecting a significant area of
blood flow to the lung and caus-ing hemodynamic instability.
Before
thrombolytic therapy is started, prothrombin time, partial thromboplastin time,
hematocrit values, and platelet counts are obtained. Heparin is stopped prior
to administration of a thrombolytic agent. During therapy, all but essential
invasive procedures are avoided because of potential bleeding. If necessary,
fresh whole blood, packed red cells, cryoprecipitate, or frozen plasma is
administered to replace blood loss and reverse the bleed-ing tendency. After
the thrombolytic infusion is completed (which varies in duration according to
the agent used and the condition being treated), the patient is given
anticoagulants.
SURGICAL MANAGEMENT
A
surgical embolectomy is rarely performed but may be indicated if the patient
has a massive PE or hemodynamic instability or if there are contraindications
to thrombolytic therapy. Pulmonary embolectomy requires a thoracotomy with
cardiopulmonary by-pass technique. Transvenous catheter embolectomy is a
technique in which a vacuum-cupped catheter is introduced transvenously into
the affected pulmonary artery. Suction is applied to the end of the embolus and
the embolus is aspirated into the cup. The sur-geon maintains suction to hold
the embolus within the cup, and the entire catheter is withdrawn through the
right side of the heart and out the femoral vein. Catheters are available that
pulverize the clot with high-velocity jets of normal saline solution
(Goldhaber, 1998). An inferior caval filter is usually inserted at the time of
surgery to protect against a recurrence.
Interrupting
the inferior vena cava is another surgical tech-nique used when PE recurs or
when the patient is intolerant of anticoagulant therapy. This approach prevents
dislodged thrombi from being swept into the lungs while allowing ade-quate
blood flow. The preferred approach is the application of Teflon clips to the
inferior vena cava to divide the lumen into small channels without occluding
caval blood flow. Also, the use of transvenous devices that occlude or filter
the blood through the inferior vena cava is a fairly safe way to prevent
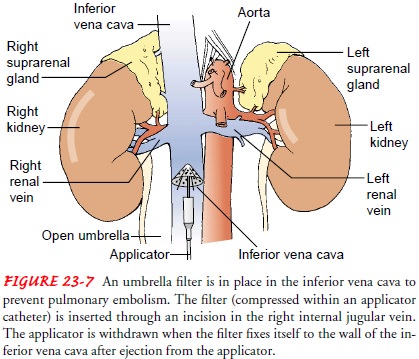
re-current PE. One such technique involves inserting a filter (eg, Greenfield
filter) through the internal jugular vein or common femoral vein (Fig. 23-7).
This filter is advanced into the infe-rior vena cava, where it is opened. The
perforated umbrella per-mits the passage of blood but prevents the passage of
large thrombi. It is recommended that anticoagulation be continued in patients
with a caval filter, if there are no contraindications to its use.
Nursing Management
MINIMIZING THE RISK OF PULMONARY EMBOLISM
A key role of the nurse is to identify patients at high risk for PE and to minimize the risk of PE in all patients. The nurse must have a high degree of suspicion for PE in any patient, but partic-ularly in those with conditions predisposing to a slowing of ve-nous return (see Chart 23-8).
PREVENTING THROMBUS FORMATION
Preventing
thrombus formation is a major nursing responsibility. The nurse encourages
ambulation and active and passive leg ex-ercises to prevent venous stasis in
patients on bed rest. The nurse instructs the patient to move the legs in a
“pumping” exercise so that the leg muscles can help increase venous flow. The
nurse also advises the patient not to sit or lie in bed for prolonged periods,
not to cross the legs, and not to wear constricting clothing. Legs should not
be dangled or feet placed in a dependent position while the patient sits on the
edge of the bed; instead, the patient’s feet should rest on the floor or on a chair.
In addition, intra-venous catheters (for parenteral therapy or measurements of
cen-tral venous pressure) should not be left in place for prolonged periods.
ASSESSING POTENTIAL FOR PULMONARY EMBOLISM
The
nurse examines patients who are at risk for developing PE for a positive
Homans’ sign, which may or may not indicate im-pending thrombosis of the leg
veins. To test for Homans’ sign, the patient assumes a supine position, lifts
the leg, and dorsiflexes the foot. The nurse asks the patient to report whether
calf pain occurs during this maneuver. The occurrence of pain—a positive
Homans’ sign—may indicate deep venous thrombosis.
MONITORING THROMBOLYTIC THERAPY
The
nurse is responsible for monitoring thrombolytic and antico-agulant therapy.
Thrombolytic therapy (streptokinase, urokinase, tissue plasminogen activator)
causes lysis of deep vein thrombi and pulmonary emboli, which helps dissolve
the clots. During throm-bolytic infusion, the patient remains on bed rest,
vital signs are as-sessed every 2 hours, and invasive procedures are limited.
Tests to determine prothrombin time or partial thromboplastin time are
performed 3 to 4 hours after the thrombolytic infusion is started to confirm
that the fibrinolytic systems have been activated. Be-cause of the prolonged
clotting time, only essential arterial punc-tures or venipunctures are
performed, and manual pressure is applied to any puncture site for at least 30
minutes. Pulse oxime-try is used to monitor changes in oxygenation. The nurse
imme-diately discontinues the infusion if uncontrolled bleeding occurs.
MANAGING PAIN
Chest
pain, if present, is usually pleuritic rather than cardiac in origin. A
semi-Fowler’s position provides a more comfortable po-sition for breathing.
However, it is important to continue to turn the patient frequently and
reposition the patient to improve the ventilation–perfusion
ratio in the lung. The nurse administersopioid analgesics as prescribed for
severe pain.
MANAGING OXYGEN THERAPY
Careful
attention is given to the proper use of oxygen. It is im-portant to ensure that
the patient understands the need for con-tinuous oxygen therapy. The nurse
assesses the patient frequently for signs of hypoxemia and monitors the pulse
oximetry values to evaluate the effectiveness of the oxygen therapy. Deep
breathing and incentive spirometry are indicated for all patients to mini-mize
or prevent atelectasis and improve ventilation. Nebulizer therapy or percussion
and postural drainage may be used for management of secretions.
RELIEVING ANXIETY
The
nurse encourages the stabilized patient to talk about any fears or concerns
related to this frightening episode, answers the patient’s and family’s
questions concisely and accurately, explains the therapy, and describes how to
recognize untoward effects early.
MONITORING FOR COMPLICATIONS
When
caring for a patient who has had PE, the nurse must be alert for the potential
complication of cardiogenic shock or right ventricular failure subsequent to
the effect of PE on the cardio-vascular system.
PROVIDING POSTOPERATIVE NURSING CARE
After
surgery, the nurse measures the patient’s pulmonary arterial pressure and
urinary output. The nurse assesses the insertion site of the arterial catheter
for hematoma formation and infection. It is important to maintain the blood
pressure at a level that sup-ports perfusion of vital organs. To prevent
peripheral venous sta-sis and edema of the lower extremities, the nurse
elevates the foot of the bed and encourages isometric exercises, use of elastic
com-pression stockings, and walking when the patient is permitted out of bed.
Sitting is discouraged because hip flexion compresses the large veins in the
legs.
PROMOTING HOME AND COMMUNITY-BASED CARE
Teaching Patients Self-Care.
Before hospital discharge and atfollow-up visits to the clinic or
during home visits, the nurse in-structs the patient about how to prevent
recurrence and what signs and symptoms to report immediately. Patient
instructions, as presented in Chart 23-9, are intended to help prevent
recur-rences and side effects of treatment.
Related Topics