Chapter: Essentials of Anatomy and Physiology: Blood
Preventing Blood Loss
Preventing Blood Loss
When a blood vessel is damaged, blood can leak into other tissues and interfere with normal tissue function, or blood can be lost from the body. The body can tolerate a small amount of blood loss and can produce new blood to replace it. But a large amount of blood loss can lead to death. Fortunately, when a blood vessel is damaged, loss of blood is minimized by three processes: vascular spasm, platelet plug formation, and blood clotting.
Vascular Spasm
Vascular spasm is an immediate but temporary constriction of ablood vessel that results when smooth muscle within the wall of the vessel contracts. This constriction can close small vessels completely and stop the flow of blood through them. Damage to blood vessels can activate nervous system reflexes that cause vascular spasm. Chemicals also produce vascular spasm. For example, platelets releasethromboxanes (throm′ bok-zānz), which are derived from certain prostaglandins, and endothelial (epithelial) cells lining blood vessels release the peptide endothelin (en-dō-thē′ lin).
Platelet Plug Formation
A platelet plug is an accumulation of platelets that can seal up a small break in a blood vessel. Platelet plug formation is very important in maintaining the integrity of the blood vessels of the cardiovascular system because small tears occur in the smaller vessels and capillaries many times each day. People who lack the normal number of platelets tend to develop numerous small hemorrhages in their skin and internal organs.
The formation of a platelet plug can be described as a series of steps, but in actuality many of these steps occur at the same time (figure 11.8). First, platelets stick to the collagen exposed by blood vessel damage; this phenomenon is called platelet adhe-sion. Most platelet adhesion is mediated through von Willebrand factor, a protein produced and secreted by blood vessel endothe-lial cells. Von Willebrand factor forms a bridge between collagen and platelets by binding to platelet surface receptors and collagen. After platelets adhere to collagen, they become activated, change shape, and release chemicals.
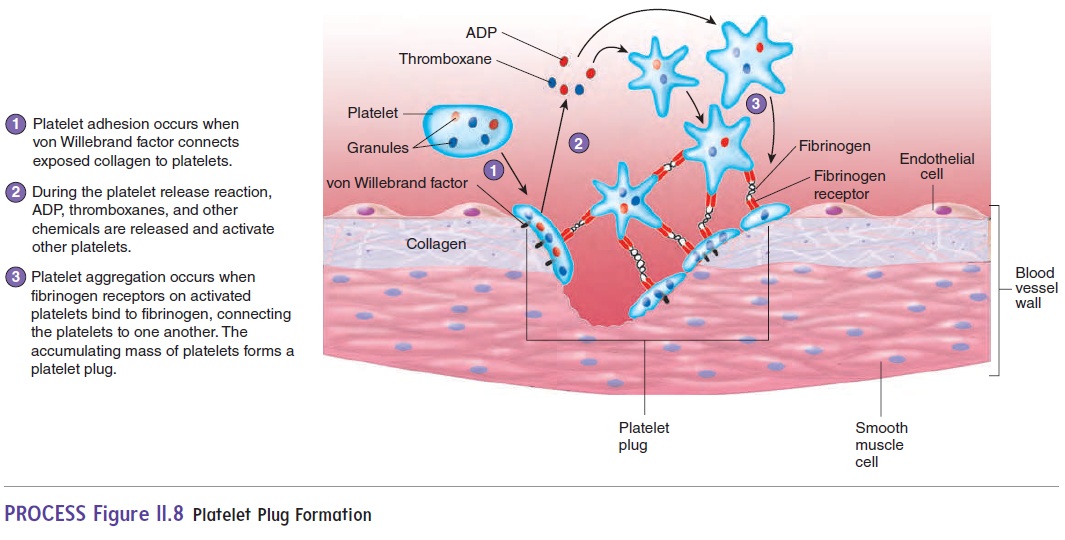
In the platelet release reaction, platelets release chemicals, such as ADP and thromboxane, which bind to their respective receptors on the surfaces of other platelets, activating the platelets. These activated platelets also release ADP and thromboxane, which activates more platelets. Thus, a cascade of chemical release activates many platelets. This is an example of positive feedback. As platelets become activated, they express surface receptors called fibrinogen receptors, which can bind to fibrino-gen, a plasma protein. In platelet aggregation, fibrinogen forms bridges between the fibrinogen receptors of numerous platelets, resulting in a platelet plug
Blood Clotting
Blood vessel constriction and platelet plugs alone are not suf-ficient to close large tears or cuts in blood vessels. When a blood vessel is severely damaged, blood clotting, or coagulation (kō-ag-ū-lā′shŭn), results in the formation of a clot. Aclotis a networkof threadlike protein fibers, called fibrin (f ı̄′ brin), that traps blood cells, platelets, and fluid.
The formation of a blood clot depends on a number of proteins found within plasma, called clotting factors. Though normally pres-ent in the plasma, the clotting factors are inactive and do not cause clotting. Following injury, however, the clotting factors are activated. Clot formation is a complex process involving many chemical reactions, but it can be summarized in three stages (figure 11.9).
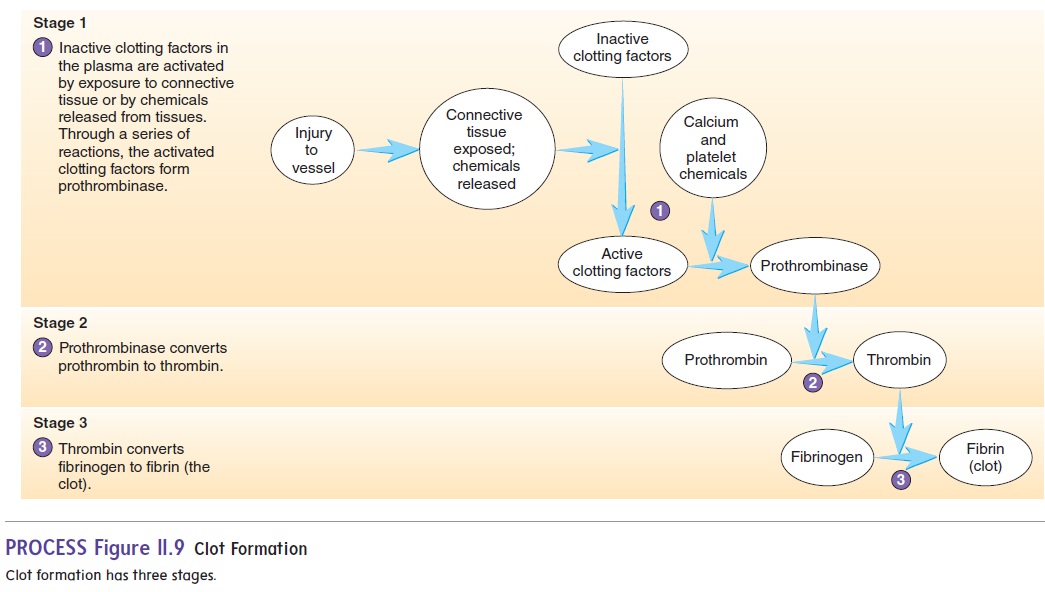
1. The chemical reactions can be started in two ways:Inactive clotting factors come in contact with exposed connective tissue, resulting in their activation, orchemicals, such as thromboplastin, are released frominjured tissues, causing activation of clotting factors. After the initial clotting factors are activated, they in turn activate other clotting factors. A series of reactions results in which each clotting factor activates the next until the clotting factor prothrombinase (prō-throm′ bi-nās), or prothrombinactivator, is formed.
2. Prothrombinase converts an inactive clotting factor called prothrombin (prō-throm′bin) to its active form, thrombin (throm′ bin).
3. Thrombin converts the plasma protein fibrinogen (fı̄-brin′ ō-jen) to fibrin.
At each step of the clotting process, each clotting factor acti-vates many additional clotting factors, resulting in the formation of a clot
Most clotting factors are manufactured in the liver, and many of them require vitamin K for their synthesis. In addition, many of the chemical reactions of clot formation require Ca2+ and the chemicals released from platelets. The clotting process can be severely impaired by low levels of vitamin K, low levels of Ca2+, low numbers of platelets, or reduced synthesis of clotting factors because of liver dysfunction.
Humans rely on two sources of vitamin K. About half comes from the diet, and the other half comes from bacteria within the large intestine. Antibiotics taken to fight bacterial infections sometimes kill these intestinal bacteria, reducing vitamin K levels and causing bleeding problems. Vitamin K supplements may be necessary for patients on prolonged antibiotic therapy. Newborns lack these intestinal bacteria and thus routinely receive a vita-min K injection at birth. Infants can also obtain vitamin K from food, such as milk. Because cow’s milk contains more vitamin K than does human milk, breast-fed infants are more susceptible to bleeding than are bottle-fed infants. However, maternal supple-mentation with vitamin K, such as with oral vitamins, adequately elevates breast-fed infant vitamin K levels and decreases the risk of bleeding.
Control of Clot Formation
Without control, clotting would spread from the point of its initiation throughout the blood vessels. Fortunately, the blood contains several anticoagulants (an′ t ē-k ō-ag′ ū-lantz), which prevent clotting factors from forming clots under normal condi-tions. For example, antithrombin (an-tē-throm′ bin) and heparin (hep′ ă-rin) inactivate thrombin. Without thrombin, fibrinogen is not converted to fibrin, and no clot forms. At an injury site, however, the activation of clotting factors is very rapid. Enough clotting factors are activated that the anticoagulants can no longer prevent a clot from forming. Away from the injury site, there are enough anticoagulants to prevent clot formation from spreading.
Clot Retraction and Fibrinolysis
After a clot has formed, it begins to condense into a more compact structure through a process known as clot retraction. Platelets contain the contractile proteins actin and myosin, which operate in a fashion similar to that of the actin and myosin in muscle . Platelets form small extensions that attach to fibrin through surface receptors. Contraction of the extensions pulls on the fibrin and leads to clot retraction. During clot retraction, serum, which is plasma without the clotting factors, is squeezed out of the clot.
Retraction of the clot pulls the edges of the damaged blood vessel together, helping stop the flow of blood, reducing the prob-ability of infection, and enhancing healing. The vessel is repairedas fibroblasts move into the damaged area and new connective tissue forms. In addition, epithelial cells around the wound divide and fill in the torn area .
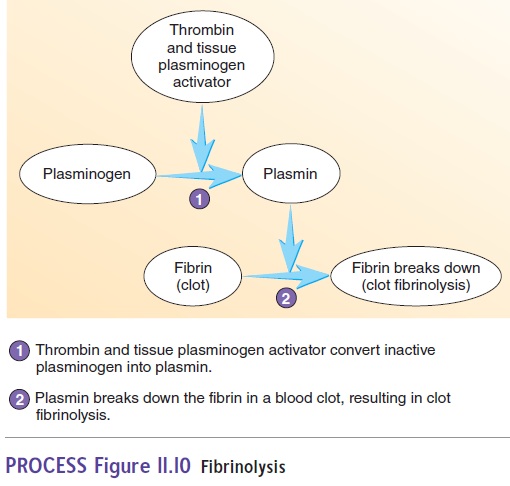
Clots are dissolved by a process called fibrinolysis (f - bri-nol′ -i-sis) (figure 11.10). An inactive plasma protein called plasminogen (plaz-min′̄o-jen) is converted to its active form, plasmin (plaz′min). Thrombin, other clotting factors activatedduring clot formation, and tissue plasminogen activator (t-PA) released from surrounding tissues can stimulate the conversion of plasminogen to plasmin. Over a few days, plasmin slowly breaks down the fibrin.
A heart attack can result when a clot blocks blood vessels that supply the heart. One treatment for a heart attack is to inject certain chemicals into the blood that activate plasmin. Unlike aspirin and anticoagulant therapies, which are used to prevent heart attacks, plasmin activators quickly dissolve the clot and restore blood flow
Related Topics