Chapter: Medical Surgical Nursing: Fluid and Electrolytes: Balance and Distribution
Potassium Excess (Hyperkalemia)
POTASSIUM
EXCESS (HYPERKALEMIA)
Hyperkalemia (greater-than-normal serum potassium concentra-tion) seldom
occurs in patients with normal renal function. Like hypokalemia, hyperkalemia
is often due to iatrogenic (treatment-induced) causes. Although less common
than hypokalemia, hyper-kalemia is usually more dangerous because cardiac
arrest is more frequently associated with high serum potassium levels.
A variation of hyperkalemia, pseudohyperkalemia has a num-ber of causes
The most common causes are the use of a tight tourniquet around an exercising
extremity while drawing a blood sample and hemolysis of the sample before
analysis. Other causes include marked leukocytosis (white blood cell count
exceeding 200,000) or thrombocytosis (platelet count exceeding 1 million),
drawing blood above a site where potassium is infusing, and fa-milial
pseudohyperkalemia, where potassium leaks out of the red blood cells while the
blood is awaiting analysis. Failure to be aware of these causes of
pseudohyperkalemia can lead to aggressive treat-ment of a nonexistent
hyperkalemia, resulting in serious lower-ing of serum potassium levels. Thus,
measurements of grossly elevated levels should be verified.
The major cause of hyperkalemia is decreased renal excretion of
potassium. Thus, significant hyperkalemia is commonly seen in patients with
untreated renal failure, particularly those in whom potassium levels rise as a
result of infection or excessive in-take of potassium in food or medications.
In addition, patients with hypoaldosteronism and Addison’s disease are at risk
for hyperkalemia because these conditions are characterized by defi-cient
adrenal hormones, leading to sodium loss and potassium retention.
Medications have been identified as a probable contributing factor in
more than 60% of hyperkalemic episodes. Medications commonly implicated are
potassium chloride, heparin, ACE in-hibitors, captopril, NSAIDs, and potassium-sparing
diuretics. In most such cases, potassium regulation is compromised by renal
insufficiency (Perazella, 2000).
Although a high intake
of potassium can cause severe hyper-kalemia in patients with impaired renal
function, hyperkalemia rarely occurs in people with normal renal function. For
all pa-tients, however, improper use of potassium supplements predis-poses them
to hyperkalemia, especially when salt substitutes are used. Not all patients
receiving potassium-losing diuretics require potassium supplements, and
patients receiving potassium-conserving diuretics should not receive
supplements.
In acidosis, potassium moves
out of the cells into the ECF. This occurs as hydrogen ions enter the cells, a
process that buffers the pH of the ECF. An elevated extracellular potassium
level should be anticipated when extensive tissue trauma has occurred, as in
burns, crushing injuries, or severe infections. Similarly, it can occur with
lysis of malignant cells after chemotherapy.
Clinical Manifestations
The most important consequence of hyperkalemia is its effect on the
myocardium. Cardiac effects of an elevated serum potassium level are usually
not significant below a concentration of 7 mEq/L (7 mmol/L), but they are
almost always present when the level is 8 mEq/L (8 mmol/L) or greater. As the
plasma potassium level rises, disturbances in cardiac conduction occur. The
earliest changes, often occurring at a serum potassium level greater than 6
mEq/L (6 mmol/L), are peaked, narrow T waves; ST-segment depres-sion; and a
shortened QT interval. If the serum potassium level continues to rise, the PR
interval becomes prolonged and is fol-lowed by disappearance of the P waves.
Finally, there is decom-position and prolongation of the QRS complex (see Fig.
14-5). Ventricular dysrhythmias and cardiac arrest may occur at any point in
this progression.
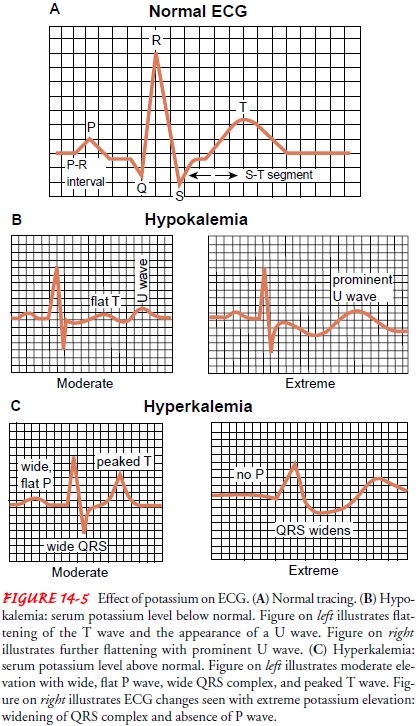
Severe hyperkalemia causes skeletal muscle weakness and even paralysis,
related to a depolarization block in muscle. Similarly, ventricular conduction
is slowed. Although hyperkalemia has marked effects on the peripheral nervous
system, it has little effect on the central nervous system. Rapidly ascending
muscular weak-ness leading to flaccid quadriplegia has been reported in
patients with very high serum potassium levels. Paralysis of respiratory and
speech muscles can also occur. Additionally, GI manifestations, such as nausea,
intermittent intestinal colic, and diarrhea, may occur in hyperkalemic
patients.
Assessment and Diagnostic Findings
Serum potassium levels and ECG changes are crucial to the diag-nosis of
hyperkalemia, as discussed above. Arterial blood gas analy-sis may reveal
metabolic acidosis; in many cases, hyperkalemia occurs with acidosis.
Medical Management
An immediate ECG should
be obtained to detect changes. Shortened repolarization and peaked T waves are
seen initially.
It is prudent as well to obtain a repeat serum potassium level from a
vein without an IV infusion containing potassium to verify results.
In nonacute situations,
restriction of dietary potassium and potassium-containing medications may
suffice. For example, elim-inating the use of potassium-containing salt
substitutes in the pa-tient taking a potassium-conserving diuretic may be all
that is needed to deal with mild hyperkalemia.
Prevention of serious hyperkalemia by the administration, either orally
or by retention enema, of cation exchange resins (eg, Kayexalate) may be
necessary in patients with renal impairment. Cation exchange resins cannot be
used if the patient has a para-lytic ileus because intestinal perforation can
occur. Kayexalate can bind with other cations in the GI tract and contribute to
the de-velopment of hypomagnesemia and hypocalcemia; it may also cause sodium
retention and fluid overload (Karch, 2002).
EMERGENCY PHARMACOLOGIC THERAPY
When serum potassium
levels are dangerously elevated, it may be necessary to administer IV calcium
gluconate. Within minutes after administration, calcium antagonizes the action
of hyper-kalemia on the heart. Infusion of calcium does not reduce the serum
potassium concentration but immediately antagonizes the adverse cardiac
conduction abnormalities. Calcium chloride and calcium gluconate are not
interchangeable: calcium gluconate con-tains 4.5 mEq of calcium and calcium
chloride contains 13.6 mEq of calcium; therefore, caution must be used.
Monitoring the blood
pressure is essential to detect hypoten-sion, which may result from the rapid
IV administration of cal-cium gluconate. The ECG should be continuously
monitored during administration; the appearance of bradycardia is an
indi-cation to stop the infusion. The myocardial protective effects of calcium
are transient, lasting about 30 minutes. Extra caution is required if the
patient has been “digitalized” (received accelerated dosages of a digitalis-based
cardiac glycoside to reach a desired serum digitalis level rapidly) because
parenteral administration of calcium sensitizes the heart to digitalis and may
precipitate digi-talis toxicity.
IV administration of
sodium bicarbonate may be necessary to alkalinize the plasma and cause a
temporary shift of potassium into the cells. Also, sodium bicarbonate furnishes
sodium to an-tagonize the cardiac effects of potassium. Effects of this therapy
begin within 30 to 60 minutes and may persist for hours; how-ever, they are
temporary.
IV administration of
regular insulin and a hypertonic dextrose solution causes a temporary shift of
potassium into the cells. Glu-cose and insulin therapy has an onset of action
within 30 minutes and lasts for several hours.
Beta-2 agonists also
move potassium into the cells and may be used in the absence of ischemic
cardiac disease. These stop-gap measures only temporarily protect the patient
from hyper-kalemia. If the hyperkalemic condition is not transient, actual
removal of potassium from the body is required; this may be ac-complished by
using cation exchange resins, peritoneal dialysis, hemodialysis or other forms
of renal replacement therapy.
Nursing Management
Patients at risk for potassium excess, for example those with renal
failure, should be identified so they can be monitored closely for signs of
hyperkalemia. The nurse observes for signs of muscle weakness and dysrhythmias.
The presence of paresthesias is noted, as are GI symptoms such as nausea and
intestinal colic. For patients at risk, serum potassium levels are measured
periodically.Elevated serum potassium levels may be erroneous; thus, highly
abnormal levels should always be verified. To avoid false reports of
hyperkalemia, prolonged use of a tourniquet while drawing the blood sample is
avoided, and the patient is cautioned not to ex-ercise the extremity
immediately before the blood sample is ob-tained. The blood sample is delivered
to the laboratory as soon as possible, because hemolysis of the sample results in
a falsely ele-vated serum potassium level.
PREVENTING HYPERKALEMIA
Measures are taken to prevent hyperkalemia in patients at risk, when
possible, by encouraging the patient to adhere to the pre-scribed potassium
restriction. Potassium-rich foods to be avoided include coffee, cocoa, tea,
dried fruits, dried beans, and whole-grain breads. Milk and eggs also contain
substantial amounts of potassium. Conversely, foods with minimal potassium
content include butter, margarine, cranberry juice or sauce, ginger ale,
gumdrops or jellybeans, hard candy, root beer, sugar, and honey.
CORRECTING HYPERKALEMIA
As stated earlier, it is
possible to exceed the tolerance for potas-sium in any person if it is
administered rapidly by the IV route. Therefore, great care should be taken to
monitor potassium so-lutions closely, paying close attention to the solution’s
concen-tration and rate of administration. When potassium is added to
parenteral solutions, the potassium is mixed with the fluid by in-verting the
bottle several times. Potassium chloride should never be added to a hanging
bottle because the potassium might be ad-ministered as a bolus (potassium
chloride is heavy and settles to the bottom of the container).
It is important to caution patients to use salt substitutes spar-ingly
if they are taking other supplementary forms of potassium or
potassium-conserving diuretics. Also, potassium-conserving diuretics, such as
spironolactone (Aldactone), triamterene (Dyre-nium), and amiloride (Midamor);
potassium supplements; and salt substitutes should not be administered to
patients with renal dys-function. Most salt substitutes contain approximately
50–60 mEq of potassium per teaspoon.
Related Topics