Chapter: Surgical Pathology Dissection : The Digestive System
Non-Neoplastic Intestinal Disease: Surgical Pathology Dissection
Non-Neoplastic Intestinal Disease
Small Biopsies
Proper
tissue orientation is a critical part of the histologic evaluation of biopsies
of the gastroin-testinal tract. Tissue orientation is a two-step pro-cess that
involves the coordinated actions of the endoscopist and the histotechnologist.
The en-doscopist should mount the biopsy mucosal-side up on an appropriate
solid surface (e.g., filter paper) and place it in fixative. This first step
should be done immediately, in the endoscopy suite, so that the specimen does
not dry out en route to the surgical pathology laboratory. The
histotechnologist can then embed and cut the biopsy specimen perpendicular to
the mounting surface. If the specimen is free-floating, great care must be taken
to identify the mucosal surface for proper embedding. Multiple sections should
be cut from each tissue block for histologic evalua-tion. Step sections are
preferred to serial sections so that intervening unstained sections are
avail-able for special stains as needed.
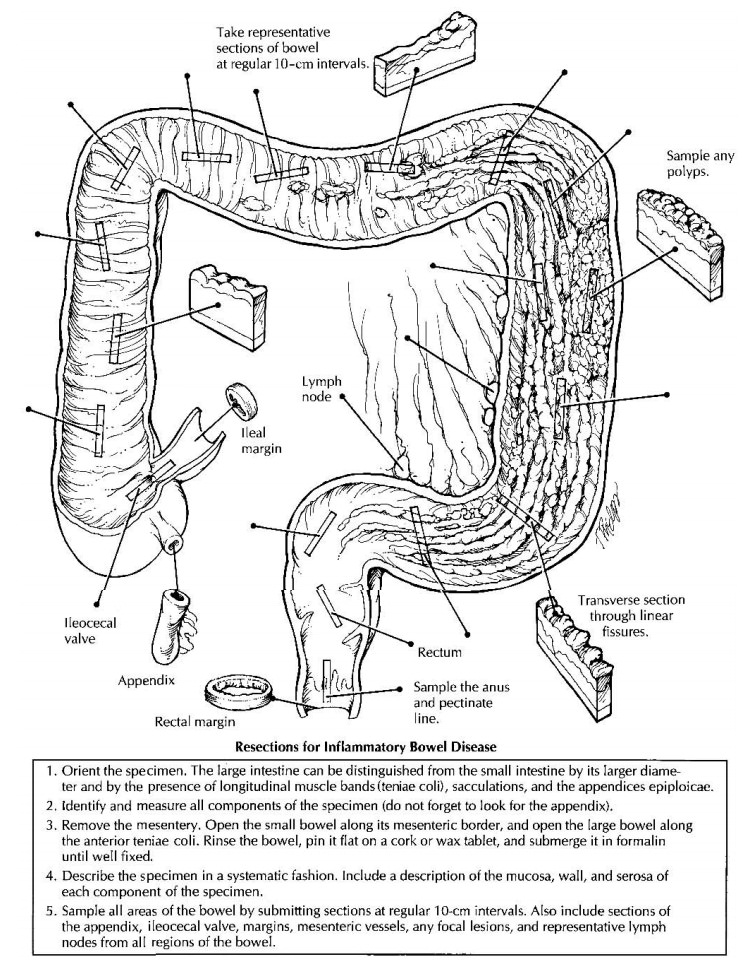
Resections of Small and Large Intestine for Inflammatory Bowel Disease
Given
the structural simplicity of the intestinal tract and the ease with which the
bowel can be opened, there is a strong tendency to rush into these dissections without
thinking ahead. The ap-proach to the non-neoplastic bowel specimen requires an
effective strategy that gives careful consideration to an organized gross
description, specimen photography and fixation, and details of dissection and
tissue sampling.
The Organized Gross Description
A good
gross description not only describes all the relevant gross findings but
presents these findings in an organized fashion. This can be a difficult task
in bowel resections, where the speci-men may consist of more than one structure
(e.g., ileum, appendix, cecum, and colon). Organize your gross description.
First, describe the speci-men after it has been examined and at least
par-tially dissected. This will make it possible to collect all of the gross
findings and integrate them into an organized statement. Second, always
de-scribe each component of the resection as an indi-vidual unit. For example,
describe the mucosa, wall, and serosa of the ileum and then move on to the
appendix, cecum, and finally the colon. Third, focus on the mucosa. Begin by
describ-ing the distribution of mucosal alterations (e.g., diffuse,
discontinuous) and then describe the specific characteristics of these changes
(e.g., ul-cerated, granular). Of course, no gross descrip-tion is complete without
a description of the wall, serosa, and mesentery; but for inflammatory bowel
disease, a less detailed description of these layers will generally suffice.
Specimen Dissection
Given
the structural simplicity of the bowel, opening these specimens is generally
straightfor-ward. When possible, the small intestine should be opened adjacent
to the mesentery. In contrast, the large intestine should be opened on the
anti-mesenteric border along the anterior (free) teniae coli. Remove the
mesentery before fixing the bowel. Treat the mesenteric soft tissues as though
For total colectomies, remove the mesentery as six separate portions,
and designate these as proximal ascending, distal ascending, proximal
transverse, distal transverse, proximal descending, and distal descending.
Although only representative lymph nodes need to be submitted, the six portions
should be clearly labeled and saved for easy retrieval if more ex-tensive lymph
node sampling is later required.
Specimen Fixation
In
general, the bowel should be fixed before it is photographed, described, and
sectioned. Subtle mucosal alterations (e.g., erosions, ulcerations, areas of
hemorrhage) that may not be apparent in the fresh specimen are often well
defined once the specimen is fixed. In most cases, the specimen can be opened
and pinned on a solid surface as a flat sheet and then submerged in formalin.
Some specimens may be so distorted that they cannot be easily opened and pinned
flat without the risk of cutting across structures (e.g., fistulae,
diverti-cula) and disrupting important relationships. These distorted specimens
may be best handled by infusing formalin into the lumen of the bowel and then
clamping both ends of the specimen. Whether the bowel is submerged or infused,
fecal material should first be rinsed from the muco-sal surface (a more
formidable challenge in the unopened specimen) using a gentle stream of an
isotonic solution.
Specimen Photography
Photographs of the specimen should be liberally taken to document further the gross findings, es-pecially the distribution and nature of the muco-sal alterations. Photograph the specimen after it has been opened and fixed. Photographs of the unopened bowel are generally useless. Fixation tends to both accentuate the mucosal alterations and reduce the amount of reflected light. Always position the specimen anatomically on the photography table. Total colectomies, for ex-ample, should be positioned so that the ascending colon is to the anatomic right, the descending colon is to the left, and the transverse colon is to the top and center. Finally, include close-up photographs to illustrate the details of the mucosal pathology.
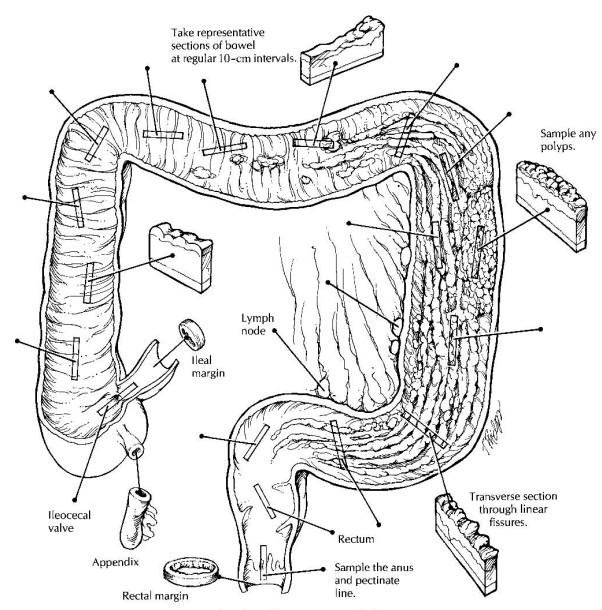
Tissue Sampling
To
evaluate the distribution of inflammatory changes in the specimen, all areas of
the bowel should be sampled for histologic evaluation. One method that
consistently ensures adequate sam-pling is to submit representative sections at
10-cm intervals, beginning at the distal end of the specimen and proceeding
proximally in a step-wise fashion. This not only will ensure that the mucosa is
well sampled but will also provide information on the distribution of the
disease process. Of course, sections of any focal lesions such as ulcers or
polyps should be submitted in addition to these interval sections. Sections
should also be taken of the appendix and the ileocecal valve when these
structures are present. When no tumor is grossly apparent, the resection
margins may be taken as shave sections. In addi-tion to sampling the mesentery
for lymph nodes, submit sections of mesenteric blood vessels and of any focal
lesions such as fistula tracts or areas of fat necrosis. Indicate the site from
which all sections were taken on the Polaroid or digital photographs. Take
longitudinal sections (i.e., par-allel to the teniae coli). Exceptions are
permitted when, for example, a linear ulcer is best demon-strated by a
transverse section through the bowel.
Once
these principles regarding gross descrip-tion, fixation, dissection, and
sampling are mas-tered, the inflammatory bowel specimen can be handled with
relative ease. The initial step is to identify the structures that are present
in the re-sected specimen. The large intestine is readily distinguished from
the small intestine by its larger diameter and the presence of longitudinal
muscle bands (the teniae coli), sacculations (the haustra), and the appendices
epiploicae. In addi-tion, the small intestine shows mucosal folds that stretch
across the entire circumference of the bowel, whereas the mucosal folds of the
large in-testine are discontinuous. Several features may be helpful in
appreciating the various regions of the large intestine. The cecum is usually
quite apparent, and it can be used to identify the origin of the ascending
colon. The transverse colon can be recognized by its large mesenteric pedicle attachment,
while the sigmoid colon has a rela-tively short mesenteric pedicle. When a
portion of rectum is included in the specimen, it can be distinguished from the
sigmoid colon by the absence of a peritoneal surface lining.
The
initial description should be limited to a list of the structures present and
the dimensions of each. Further handling of the bowel is facili-tated by
removing the mesenteric fat. As de-scribed previously, these soft tissues
should be removed according to their anatomic location, and each portion should
be clearly labeled so it can be easily identified and retrieved. Most
pro-sectors have an easier time finding mesenteric lymph nodes while these
tissues are still in the fresh state. Remember, most of the lymph nodes are
found at the junction of the bowel and mesen-teric fat. Open the bowel along
its entire length, cutting the small bowel along its mesenteric at-tachment and
the large bowel along the anterior teniae. Gently rinse any fecal material from
the mucosal surface using a stream of isotonic solu-tion. Pin the opened bowel
flat on a solid surface, and submerge it in formalin.
Once the
specimen is fixed, complete the exam-ination of the mucosa, noting in the gross
descrip-tion the distribution and characteristics of the mucosal alterations.
Place the bowel in its correct anatomic position, and photograph it. Take a
Polaroid or digital photograph of the entire specimen, so that the sites from
which histologic sections were taken can be indicated on the image, and take
close-up views of focal lesions.
Remember
to section all regions of the bowel by using a method of stepwise sectioning at
regu-lar (i.e., 10-cm) intervals. Specific bowel sections should include the
proximal and distal resection margins, the ileocecal valve, the appendix, and
any focal lesions. If a neoplasm is not found, sam-pling of representative
lymph nodes from each level will suffice. Sampling of the mesentery should also
include a section of the mesenteric blood vessels and sections of any focal
lesions.
Important Issues to Address in Your Surgical Pathology Report on Non-Neoplastic Intestinal Disease
· What
procedure was performed, and what structures/organs are present?
· What
disease processes are present, and what is their location?
·
For inflammatory processes, are any
diver-ticula, strictures, fistulae, or perforations present?
·
Does the mucosa show any preneoplastic or
neoplastic changes?
·
Do the lymph nodes show any evidence of an
inflammatory process or metastatic disease? Record the number of lymph nodes examined
and the presence or absence of lymph node metastases.
·
What is the status of the mesenteric vessels?
Related Topics