Chapter: Medical Surgical Nursing: Fluid and Electrolytes: Balance and Distribution
Mixed AcidŌĆōBase Disorders and Compensation
MIXED ACIDŌĆōBASE DISORDERS
At times patients can simultaneously experience two or more in-dependent
acidŌĆōbase disorders. A normal pH in the presence of changes in the PaCO2 and plasma HCO3ŌłÆ concentration immediately suggests a mixed
disorder. The only mixed disorder that cannot occur is a mixed respiratory
acidosis and alkalosis, because it is impossible to have alveolar
hypoventilation and hyperventila-tion at the same time. An example of a mixed
disorder is the simul-taneous occurrence of metabolic acidosis and respiratory
acidosis during respiratory and cardiac arrest.
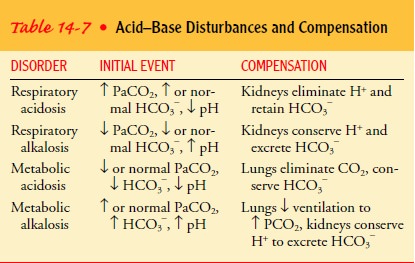
COMPENSATION
Generally, the pulmonary and renal systems compensate for each other to
return the pH to normal. In a single acidŌĆōbase disorder, the system not causing
the problem will try to compensate by re-turning the ratio of bicarbonate to
carbonic acid to the normal 20 1. The lungs compensate for metabolic
disturbances by changing CO2 excretion. The kidneys compensate for respiratory disturbances by
altering bicarbonate retention and H+ secretion.
In respiratory acidosis,
excess hydrogen is excreted in the urine in exchange for bicarbonate ions. In
respiratory alkalosis, the renal excretion of bicarbonate increases, and
hydrogen ions are retained. In metabolic acidosis, the compensatory mecha-nisms
increase the ventilation rate and the renal retention of bicarbonate.
In metabolic alkalosis, the respiratory system compensates by decreasing
ventilation to conserve CO2 and raise the PaCO2. Be-cause the lungs respond to acidŌĆōbase disorders within minutes,
compensation for metabolic imbalances occurs faster than com-pensation for
respiratory imbalances. Table 14-7 summarizes compensation effects.
Related Topics