Chemistry - Crystal lattice and unit cell | 12th Chemistry : UNIT 6 : Solid State
Chapter: 12th Chemistry : UNIT 6 : Solid State
Crystal lattice and unit cell
Crystal
lattice and unit cell:
Crystalline solid is
characterised by a definite orientation of atoms, ions or molecules, relative
to one another in a three dimensional pattern. The regular arrangement of these
species throughout the crystal is called a crystal lattice. A basic repeating
structural unit of a crystalline solid is called a unit cell. The following
figure illustrates the lattice point and the unit cell.
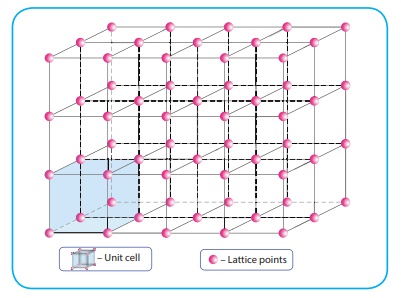
A crystal may be
considered to consist of large number of unit cells, each one in direct contact
with its nearer neighbour and all similarly oriented in space. The number of
nearest neighbours that surrounding a particle in a crystal is called the
coordination number of that particle.
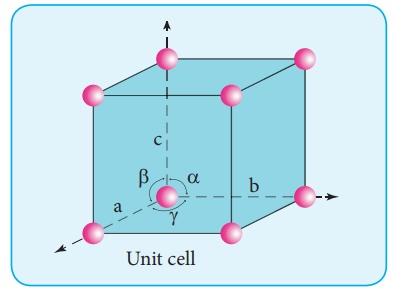
A unit cell is
characterised by the three edge lengths or lattice constants a ,b and c and the
angle between the edges α, and γ
Related Topics