Chapter: Medical Surgical Nursing: Oncology: Nursing Management in Cancer Care
Chemotherapy - Management of Cancer
CHEMOTHERAPY
In chemotherapy, antineoplastic agents are used in an attempt to
destroy tumor cells by interfering with cellular functions and reproduction.
Chemotherapy is used primarily to treat systemic disease rather than lesions
that are localized and amenable to surgery or radiation. Chemotherapy may be
combined with sur-gery or radiation therapy, or both, to reduce tumor size
pre-operatively, to destroy any remaining tumor cells postoperatively, or to
treat some forms of leukemia. The goals of chemotherapy (cure, control,
palliation) must be realistic because they will de-fine the medications to be
used and the aggressiveness of the treatment plan.
Cell Kill and the Cell Cycle
Each time a tumor is exposed to a chemotherapeutic agent, a per-centage
of tumor cells (20% to 99%, depending on dosage) is de-stroyed. Repeated doses
of chemotherapy are necessary over a prolonged period to achieve regression of
the tumor. Eradication of 100% of the tumor is nearly impossible, but a goal of
treat-ment is to eradicate enough of the tumor so that the remaining tumor
cells can be destroyed by the body’s immune system.
Actively proliferating
cells within a tumor (growth fraction) are the most sensitive to
chemotherapeutic agents. Nondividing cells capable of future proliferation are
the least sensitive to antineo-plastic medications and consequently are
potentially dangerous. The nondividing cells must be destroyed, however, to
eradicate a cancer completely. Repeated cycles of chemotherapy are used to kill
more tumor cells by destroying these nondividing cells as they begin active
cell division.
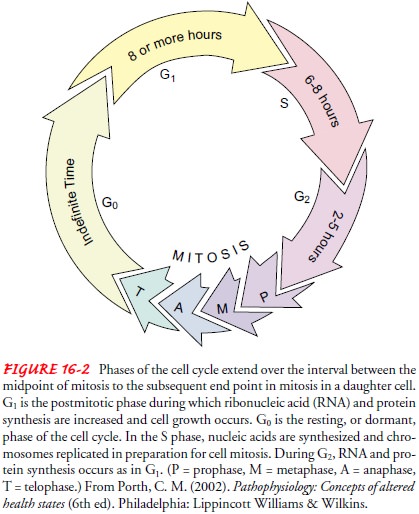
Reproduction of both healthy and malignant cells
follows the cell cycle pattern (Fig. 16-2). The cell cycle time is the time
re-quired for one tissue cell to divide and reproduce two identical daughter
cells. The cell cycle of any cell has four distinct phases, each with a vital
underlying function:
•
G1
phase—RNA and protein synthesis occur.
•
S
phase—DNA synthesis occurs.
·
G2
phase—premitotic phase; DNA synthesis is complete, mitotic spindle forms.
·
Mitosis—cell
division occurs.
The G0 phase, the resting or dormant
phase of cells, can occur after mitosis and during the G1 phase. In
the G0 phase are those dangerous cells that are not actively dividing
but have the poten-tial for replicating. The administration of certain
chemothera-peutic agents (as well as administration of some other forms of
therapy) is coordinated with the cell cycle.
Classification of Chemotherapeutic Agents
Certain chemotherapeutic agents (cell cycle–specific drugs) de-stroy
cells actively reproducing by means of the cell cycle. Many of these agents are
specific to certain phases of the cell cycle. Most affect cells in the S phase
by interfering with DNA and RNA syn-thesis. Others, such as the vinca or plant
alkaloids, are specific to the M phase, where they halt mitotic spindle
formation.
Chemotherapeutic agents
that act independently of the cell cycle phases are termed cell
cycle–nonspecific agents. These agents usually have a prolonged effect on
cells, leading to cel-lular damage or death. Many treatment plans combine cell
cycle–specific and cell cycle–nonspecific agents to increase the number of
vulnerable tumor cells killed during a treatment period.
Chemotherapeutic agents are also classified according to var-ious
chemical groups, each with a different mechanism of action. These include the
alkylating agents, nitrosureas, antimetabolites, antitumor antibiotics, plant
alkaloids, hormonal agents, and mis-cellaneous agents. The classification,
mechanism of action, com-mon drugs, cell cycle specificity, and common side
effects of antineoplastic agents are listed in Table 16-6.

Chemotherapeutic agents from each category may be used to enhance the
tumor cell kill during therapy by creating multiple cellular lesions. Combined
medication therapy relies on medica-tions of differing toxicities and with
synergistic actions. Using combination drug therapy also prevents development
of drug-resistant mechanisms.
Combining older medications
with other agents, such as lev-amisole, leucovorin, hormones, or interferons
(IFN), has shown some benefit in combating resistance of cells to
chemotherapeu-tic agents. Newer investigational agents are being studied for
effectiveness in resistant tumor lines. For more information about
investigative drugs, see Chart 16-4.

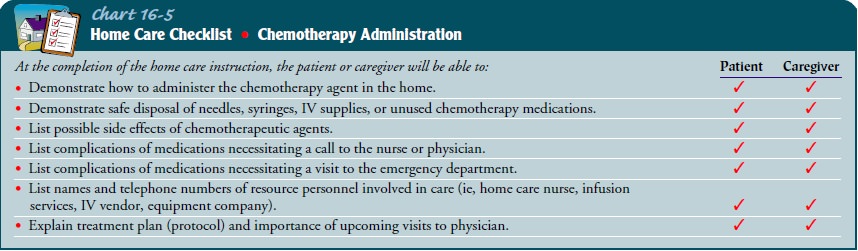
Administration of Chemotherapeutic Agents
Chemotherapeutic agents may be administered in the hospital, clinic, or
home setting by topical, oral, intravenous, intramuscular, subcutaneous,
arterial, intracavitary, and intrathecal routes. The administration route
usually depends on the type of agent, the re-quired dose, and the type,
location, and extent of tumor being treated. Guidelines for the administration
of chemotherapy have been developed by the Oncology Nursing Society. Patient
edu-cation is essential to maximize safety if chemotherapy is admin-istered in
the patient’s home (Chart 16-5).
DOSAGE
Dosage of antineoplastic agents is based primarily on the patient’s total body surface area, previous response to chemotherapy or ra-diation therapy, and major organ function.
SPECIAL PROBLEMS: EXTRAVASATION
Special care must be taken whenever intravenous vesicant agents are administered. Vesicants are those agents that, if deposited into the subcutaneous tissue (extravasation), cause tissue necrosis and damage to underlying tendons, nerves, and blood vessels. Al-though the complete mechanism of tissue destruction is unclear,it is known that the pH of many antineoplastic drugs is responsi-ble for the severe inflammatory reaction as well as the ability of these drugs to bind to tissue DNA. Sloughing and ulceration of the tissue may be so severe that skin grafting may be necessary. The full extent of tissue damage may take several weeks to become ap-parent. Medications classified as vesicants include dactinomycin, daunorubicin, doxorubicin (Adriamycin), nitrogen mustard, mito-mycin, vinblastine, vincristine, and vindesine.
Only specially trained
physicians and nurses should administer vesicants. Careful selection of
peripheral veins, skilled venipunc-ture, and careful administration of
medications are essential. In-dications of extravasation during administration
of vesicant agents include the following:
• Absence of blood return from the intravenous catheter
• Resistance to flow of intravenous fluid
• Swelling, pain, or redness at the site
If extravasation is
suspected, the medication administration is stopped immediately, and ice is
applied to the site (unless the ex-travasated vesicant is a vinca alkaloid).
The physician may aspi-rate any infiltrated medication from the tissues and
inject a neutralizing solution into the area to reduce tissue damage.
Se-lection of the neutralizing solution depends on the extravasated agent.
Examples of neutralizing solutions include sodium thio-sulfate, hyaluronidase,
and sodium bicarbonate. Recommenda-tions and guidelines for managing vesicant
extravasation have been issued by individual medication manufacturers,
pharmacies, and the Oncology Nursing Society, and they differ from one
medication to the next.
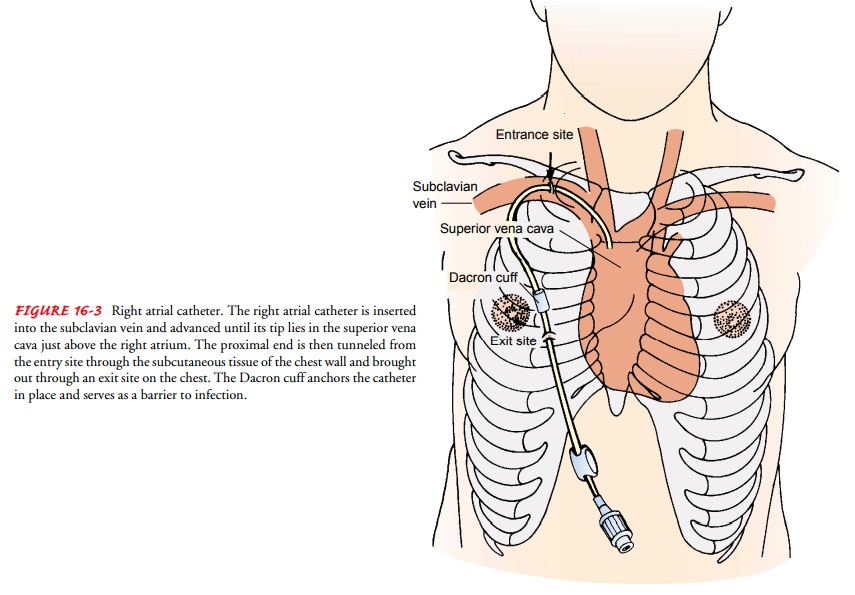
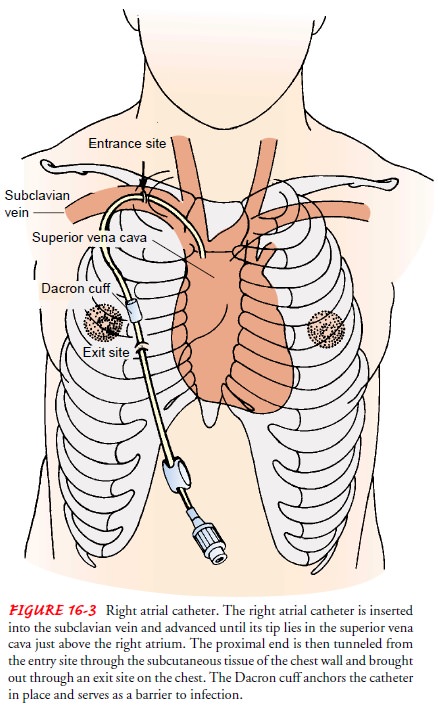
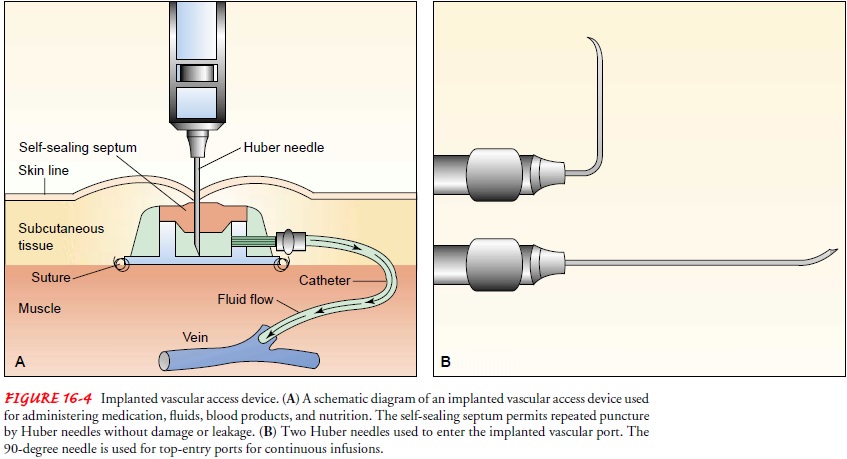
When frequent, prolonged
administration of antineoplastic vesicants is anticipated, right atrial
Silastic catheters or venous ac-cess devices may be inserted to promote safety
during medication administration and reduce problems with access to the
circula-tory system (Figs. 16-3 and 16-4). Complications associated with their
use include infection and thrombosis.
TOXICITY
Toxicity associated with
chemotherapy can be acute or chronic. Cells with rapid growth rates (eg,
epithelium, bone marrow, hair follicles, sperm) are very susceptible to damage,
and various body systems may be affected as well.
Gastrointestinal System.
Nausea
and vomiting are the mostcommon side effects of chemotherapy and may persist
for up to 24 hours after its administration. The vomiting centers in the brain
are stimulated by (1) activation of the receptors found in the chemoreceptor
trigger zone (CTZ) of the medulla; (2) stim-ulation of peripheral autonomic
pathways (gastrointestinal tract and pharynx); (3) stimulation of the
vestibular pathways (inner ear imbalances, labyrinth input); (4) cognitive
stimulation (cen-tral nervous system disease, anticipatory nausea and
vomiting); and (5) a combination of these factors.
Medications that can decrease nausea and vomiting include sero-tonin blockers, such as ondansetron, granisetron, and dolasetron, which block serotonin receptors of the gastrointestinal tract and CTZ, and dopaminergic blockers, such as metoclopramide (Reglan), which block dopamine receptors of the CTZ. Pheno-thiazines, sedatives, corticosteroids, and histamines are used in combination with serotonin blockers with the more emetogenic chemotherapeutic regimens (Bremerkamp, 2000).
Delayed nausea and
vomiting that occur later than 48 to 72 hours after chemotherapy are
troublesome for some patients. To minimize discomfort, some antiemetic medications
are nec-essary for the first week at home after chemotherapy. Relaxation
techniques and imagery can also help to decrease stimuli con-tributing to
symptoms. Altering the patient’s diet to include small frequent meals, bland
foods, and comfort foods may reduce the frequency or severity of these
symptoms.
Although the epithelium that lines the oral cavity quickly renews
itself, its rapid rate of proliferation makes it susceptible to the effects of
chemotherapy. As a result, stomatitis and anorexia are common. The entire
gastrointestinal tract is susceptible to mucositis (inflam-mation of the
mucosal lining), and diarrhea is a common result. Antimetabolites and antitumor
antibiotics are the major culprits in mucositis and other gastrointestinal
symptoms. Irinotecan is re-sponsible for causing diarrhea, which can be severe
in some patients.
Hematopoietic System.
Most
chemotherapeutic agents causemyelosuppression
(depression of bone marrow function), re-sulting in decreased production of
blood cells. Myelosuppression decreases the number of WBCs (leukopenia), red
blood cells (anemia), and platelets (thrombocytopenia) and increases the risk
for infection and bleeding. Depression of these cells is the usual reason for
limiting the dose of the chemotherapeutic agents. Monitoring blood cell counts
frequently is essential, as is pro-tecting the patient from infection and
injury, particularly while the blood cell counts are depressed.
Other agents, called colony-stimulating factors (granulocyte
colony-stimulating factor [G-CSF], granulocyte-macrophage colony-stimulating
factor [GM-CSF], and erythropoietin [EPO]), can be administered after
chemotherapy. G-CSF and GM-CSF stimulate the bone marrow to produce WBCs,
especially neu-trophils, at an accelerated rate, thus decreasing the duration
of neutropenia. The colony-stimulating factors decrease the episodes of
infection and the need for antibiotics and allow for more timely cycling of
chemotherapy with less need to reduce the dosage. EPO stimulates red blood cell
production, thus decreas-ing the symptoms of chronic administered anemia.
Renal System.
Chemotherapeutic
agents can damage the kid-neys because of their direct effects during excretion
and the ac-cumulation of end products after cell lysis. Cisplatin, methotrexate,
and mitomycin are particularly toxic to the kidneys. Rapid tumor cell lysis
after chemotherapy results in increased urinary excretion of uric acid, which
can cause renal damage. In addition, intra-cellular contents are released into
the circulation, resulting in ex-cessive levels of potassium and phosphates
(hyperkalemia and hyperphosphatemia) and diminished levels of calcium
(hypo-calcemia). (See later discussion of tumor lysis syndrome.)
Monitoring blood urea
nitrogen, serum creatinine, creatinine clearance, and serum electrolyte levels
is essential. Adequate hy-dration, alkalinization of the urine to prevent
formation of uric acid crystals, and the use of allopurinol are frequently
indicated to prevent these side effects.
Cardiopulmonary System.
Antitumor
antibiotics (daunorubicinand doxorubicin) are known to cause irreversible
cumulative car-diac toxicities, especially when total dosage reaches 550 mg/m2. Cardiac ejection fraction (volume of blood
ejected from the heart with each beat) and signs of congestive heart failure
must be mon-itored closely. Bleomycin, carmustine (BCNU), and busulfan are
known for their cumulative toxic effects on lung function. Pul-monary fibrosis
can be a long-term effect of prolonged dosage withthese agents. Therefore, the
patient is monitored closely for changes in pulmonary function, including
pulmonary function test results. Total cumulative doses of bleomycin are not to
exceed 400 units.
Reproductive System.
Testicular and ovarian function
can be af-fected by chemotherapeutic agents, resulting in possible sterility.
Normal ovulation, early menopause, or permanent sterility may result. In men,
temporary or permanent azoospermia (absence of spermatozoa) may develop.
Reproductive cells may be damaged during treatment, resulting in chromosomal
abnormalities in off-spring. Banking of sperm is recommended for men before
treat-ments are initiated to protect against sterility or any mutagenic damage
to sperm.
Patients and their partners need to be informed about potential changes
in reproductive function resulting from chemotherapy. They are advised to use
reliable methods of birth control while re-ceiving chemotherapy and not to
assume that sterility has resulted.
Neurologic System.
The
taxanes and plant alkaloids, especiallyvincristine, can cause neurologic damage
with repeated doses. Pe-ripheral neuropathies, loss of deep tendon reflexes,
and paralytic ileus may occur. These side effects are usually reversible and
dis-appear after completion of chemotherapy. Cisplatin is also re-sponsible for
peripheral neuropathies; hearing loss due to damage to the acoustic nerve can
also occur.
Miscellaneous.
Fatigue
is a distressing side effect for most pa-tients that greatly affects quality of
life. Fatigue can be debilitat-ing and last for months after treatment.
Nursing Management in Chemotherapy
The nurse has an important role in assessing and managing many of the
problems experienced by the patient undergoing chemother-apy. Because of the
systemic effects on normal as well as malig-nant cells, these problems are
often widespread, affecting many body systems.
ASSESSING FLUID AND ELECTROLYTE STATUS
Anorexia, nausea,
vomiting, altered taste, and diarrhea put the patient at risk for nutritional
and fluid and electrolyte distur-bances. Changes in the mucosa of the
gastrointestinal tract may lead to irritation of the oral cavity and intestinal
tract, further threatening the patient’s nutritional status. Therefore, it is
im-portant for the nurse to assess the patient’s nutritional and fluid and electrolyte
status frequently and to use creative ways to en-courage an adequate fluid and
dietary intake.
MODIFYING RISKS FOR INFECTION AND BLEEDING
Suppression of the bone marrow and immune system is an ex-pected consequence of chemotherapy and frequently serves as a guide in determining appropriate chemotherapy dosage. How-ever, this effect also increases the risk for anemia, infection, and bleeding disorders. Therefore, nursing assessment and care focus on identifying and modifying factors that further increase the pa-tient’s risk. Aseptic technique and gentle handling are indicated to prevent infection and trauma. Laboratory test results, particu-larly blood cell counts, are monitored closely. Untoward changes in blood test results and signs of infection and bleeding must be reported promptly. The patient and family members are in-structed about measures to prevent these problems at home (see Plan of Nursing Care for more information).
ADMINISTERING CHEMOTHERAPY
The local effects of the chemotherapeutic agent are also of con-cern.
The patient is observed closely during its administration be-cause of the risk
and consequences of extravasation (particularly of vesicant agents, which may
produce necrosis if deposited in the subcutaneous tissues). Local difficulties
or problems with admin-istration of chemotherapeutic agents are brought to the
attention of the physician promptly so that corrective measures can be taken
immediately to minimize local tissue damage.
IMPLEMENTING SAFEGUARDS
Nurses involved in handling chemotherapeutic agents may be ex-posed to
low doses of the drugs by direct contact, inhalation, and ingestion. Urinalyses
of personnel repeatedly exposed to cytotoxic agents demonstrate mutagenic
activity. Although not all muta-gens are carcinogenic, they can produce
permanent inheritable changes in the genetic material of cells.
Although long-term
studies of nurses handling chemothera-peutic agents have not been conducted, it
is known that chemother-apeutic agents are associated with secondary formation
of cancers and chromosome abnormalities. Additionally, nausea, vomiting,
dizziness, alopecia, and nasal mucosal ulcerations have been re-ported in
health care personnel who have handled chemother-apeutic agents.
Because of known and potential hazards associated with han-dling
chemotherapeutic agents, the Occupational Safety and Health Administration,
Oncology Nursing Society, hospitals, and other health care agencies have
developed specific precautions for those involved in the preparation and
administration of chemother-apy (Chart 16-6).

Related Topics