Chapter: Medical Surgical Nursing: Fluid and Electrolytes: Balance and Distribution
Homeostatic Mechanisms
HOMEOSTATIC
MECHANISMS
The body is equipped with remarkable homeostatic mechanisms to keep the
composition and volume of body fluid within narrow limits of normal. Organs
involved in homeostasis include the kid-neys, lungs, heart, adrenal glands,
parathyroid glands, and pitu-itary gland.
Kidney Functions
Vital to the regulation of fluid and electrolyte balance, the kid-neys
normally filter 170 L of plasma every day in the adult, while excreting only
1.5 L of urine. They act both autonomously and in response to blood-borne
messengers, such as aldosterone and antidiuretic hormone (ADH). Major functions
of the kidneys in maintaining normal fluid balance include the following:
•
Regulation of ECF volume and osmolality by
selective re-tention and excretion of body fluids
•
Regulation of electrolyte levels in the ECF by
selective re-tention of needed substances and excretion of unneeded substances
•
Regulation of pH of the ECF by retention of
hydrogen ions
•
Excretion of metabolic wastes and toxic substances
Given these functions,
it is readily apparent that renal failure will result in multiple fluid and
electrolyte problems. Renal func-tion declines with advanced age, as do muscle
mass and daily ex-ogenous creatinine production. Thus, high-normal and
minimally elevated serum creatinine values may indicate substantially re-duced
renal function in the elderly.
Heart and Blood Vessel Functions
The pumping action of
the heart circulates blood through the kidneys under sufficient pressure to
allow for urine formation. Failure of this pumping action interferes with renal
perfusion and thus with water and electrolyte regulation.
Lung Functions
The lungs are also vital in maintaining homeostasis. Through
ex-halation, the lungs remove approximately 300 mL of water daily in the normal
adult. Abnormal conditions, such as hyperpnea (abnormally deep respiration) or
continuous coughing, increase this loss; mechanical ventilation with excessive
moisture decreases it. The lungs also have a major role in maintaining
acid–base bal-ance. Changes from normal aging result in decreased respiratory
function, causing increased difficulty in pH regulation in older adults with major
illness or trauma.
Pituitary Functions
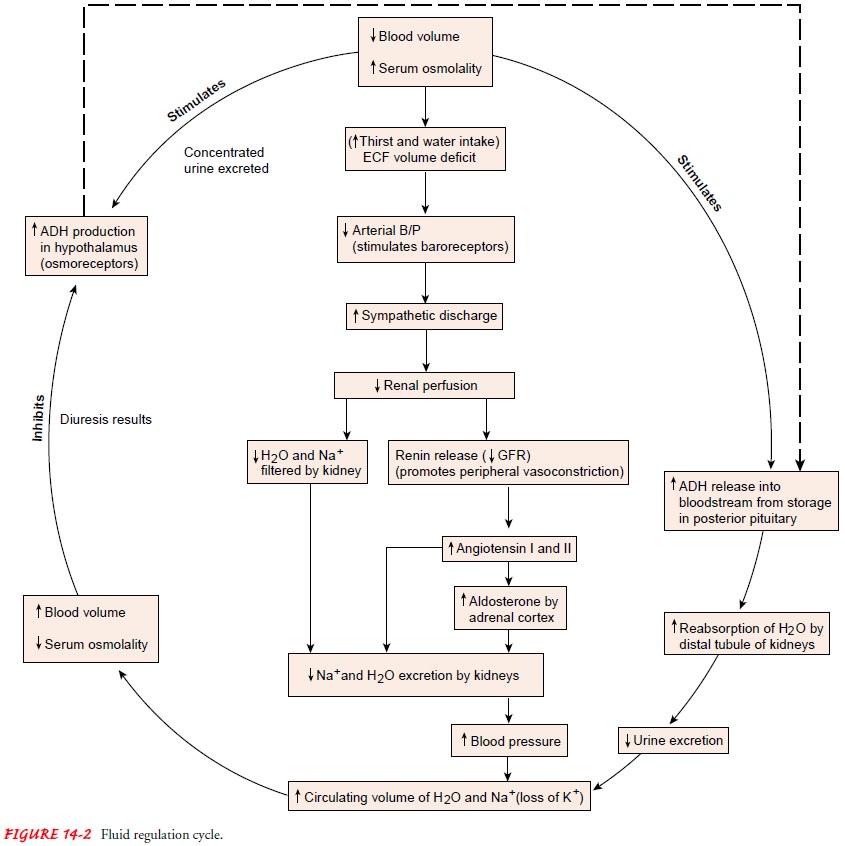
The hypothalamus manufactures ADH, which is stored in the pos-terior
pituitary gland and released as needed. ADH is sometimes called the
water-conserving hormone because it causes the body to retain water. Functions
of ADH include maintaining the osmotic pressure of the cells by controlling the
retention or excretion of water by the kidneys and by regulating blood volume
(Fig. 14-2).
Adrenal Functions
Aldosterone, a
mineralocorticoid secreted by the zona glomerulosa (outer zone) of the adrenal
cortex, has a profound effect on fluid balance. Increased secretion of
aldosterone causes sodium reten-tion (and thus water retention) and potassium
loss. Conversely, decreased secretion of aldosterone causes sodium and water loss
and potassium retention.
Cortisol, another
adrenocortical hormone, has only a fraction of the mineralocorticoid potency of
aldosterone. When secreted in large quantities, however, it can also produce
sodium and fluid retention and potassium deficit.
Parathyroid Functions
The parathyroid glands, embedded in the thyroid gland, regulate calcium
and phosphate balance by means of parathyroid hormone (PTH). PTH influences
bone resorption, calcium absorption from the intestines, and calcium
reabsorption from the renal tubules.
Other Mechanisms
Changes in the volume of
the interstitial compartment within the ECF can occur without affecting body
function. The vascular compartment, however, cannot tolerate change as readily
and must be carefully maintained to ensure that tissues receive ade-quate
nutrients.
BARORECEPTORS
The baroreceptors are small nerve receptors that detect changes in
pressure within blood vessels and transmit this information to the central
nervous system. They are responsible for monitoring the circulating volume, and
they regulate sympathetic and para-sympathetic neural activity as well as
endocrine activities. They are categorized as low-pressure and high-pressure
baroreceptor systems. Low-pressure baroreceptors are located in the cardiac atria,
particularly the left atrium. The high-pressure barorecep-tors are nerve
endings in the aortic arch and in the cardiac sinus. Another high-pressure
baroreceptor is located in the afferent arteriole of the juxtaglomerular
apparatus of the nephron.
As arterial pressure decreases, baroreceptors transmit fewer im-pulses
from the carotid sinuses and the aortic arch to the vasomotor center. A
decrease in impulses stimulates the sympathetic nervous system and inhibits the
parasympathetic nervous system. The out-come is an increase in cardiac rate,
conduction, and contractility and in circulating blood volume. Sympathetic
stimulation constricts renal arterioles; this increases the release of
aldosterone, decreases glomerular filtration, and increases sodium and water
reabsorption.
RENIN–ANGIOTENSIN–ALDOSTERONE SYSTEM
Renin is an enzyme that converts angiotensinogen, an inactive substance
formed by the liver, into angiotensin I. Renin is released by the
juxtaglomerular cells of the kidneys in response to decreased renal perfusion.
Angiotensin-converting enzyme (ACE) converts angiotensin I to angiotensin II.
Angiotensin II, with its vasocon-strictor properties, increases arterial
perfusion pressure and stim-ulates thirst. As the sympathetic nervous system is
stimulated, aldosterone is released in response to an increased release of
renin. Aldosterone is a volume regulator and is also released as serum
potassium increases, serum sodium decreases, or adrenocortico-tropic hormone
increases.
ADH AND THIRST
ADH and the thirst mechanism have important roles in main-taining sodium
concentration and oral intake of fluids. Oral intake is controlled by the
thirst center located in the hypothalamus. As serum concentration or osmolality
increases or blood volume de-creases, neurons in the hypothalamus are
stimulated by intra-cellular dehydration; thirst then occurs, and the person
increases oral intake of fluids. Water excretion is controlled by ADH,
aldosterone, and baroreceptors, as mentioned previously. The presence or absence
of ADH is the most significant factor in deter-mining whether the urine that is
excreted is concentrated or dilute.
OSMORECEPTORS
Located on the surface of the hypothalamus, osmoreceptors sense changes
in sodium concentration. As osmotic pressure increases, the neurons become
dehydrated and quickly release impulses to the pos-terior pituitary, which
increases the release of ADH. ADH travels in the blood to the kidneys, where it
alters permeability to water, causing increased reabsorption of water and
decreased urine output. The retained water dilutes the ECF and returns its
concentration to normal. Restoration of normal osmotic pressure provides
feedback to the osmoreceptors to inhibit further ADH release (see Fig. 14-2).
RELEASE OF ATRIAL NATRIURETIC PEPTIDE
Atrial natriuretic peptide (ANP) is released by cardiac cells in the atria of the heart in response to increased atrial pressure.
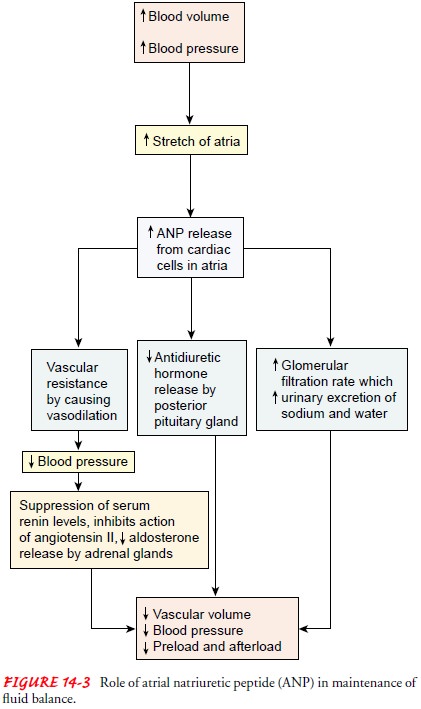
Any disorder that results in volume expansion or increased cardiac filling pressures
(eg, high sodium intake, heart failure, chronic renal fail-ure, atrial
tachycardia, or use of vasoconstrictor agents) will increase the release of
ANP. The action of ANP is the direct opposite of the
renin–angiotensin–aldosterone system and decreases blood pressure and volume
(Fig. 14-3). The ANP measured in plasma is normally 20 to 77 pg/mL (20—77
ng/L). This level increases in acute heart failure, paroxysmal atrial
tachycardia, hyperthy-roidism, subarachnoid hemorrhage, and small cell lung
cancer.The
level decreases in chronic heart failure and with the use of medications such
as urea (Ureaphil) and prazosin (Minipress).
Gerontologic Considerations
Normal physiologic changes of aging, including reduced renal and respiratory function and reserve and alterations in the ratio of body fluids to muscle mass, may alter the responses of an elderly person to fluid and electrolyte changes and acid–base disturbances.
In addition, the frequent
use of medications in older adults can affect renal and cardiac function and
fluid balance, thereby increasing the likelihood of fluid and electrolyte
disturbances. Routine pro-cedures, such as the vigorous administration of
laxatives before colon x-ray studies, may produce a serious fluid volume
deficit, necessitating the use of intravenous (IV) fluids to prevent
hy-potension and other effects of hypovolemia.
Alterations in fluid and electrolyte balance that may produce minor
changes in young and middle-aged adults have the poten-tial to produce profound
changes in older adults, accompanied by a rapid onset of signs and symptoms. In
other elderly patients, the clinical manifestations of fluid and electrolyte
disturbances may be subtle or atypical. For example, fluid deficit or reduced
sodium levels (hyponatremia) may cause confusion in the elderly person, whereas
in young and middle-aged people the first sign commonly is increased thirst.
Rapid infusion of an excessive vol-ume of IV fluids may produce fluid overload
and cardiac failure in the elderly patient. These reactions are likely to occur
more quickly and with the administration of smaller volumes of fluid than in
healthy young and middle-aged adults because of the de-creased cardiac reserve
and reduced renal function that accom-pany aging.
Increased sensitivity to fluid and electrolyte changes in the elderly
patient requires careful assessment, with attention to in-take and output of
fluids from all sources and to changes in daily weight; careful monitoring of
side effects and interactions of med-ications; and prompt reporting and
management of disturbances.
Related Topics