Chapter: Biotechnology Applying the Genetic Revolution: Transgenic Animals
Transgenic Insects
TRANSGENIC
INSECTS
Several insects can now be
genetically modified. The fruit fly Drosophila
has been investigated at the molecular level for a long time and, not
surprisingly, methods exist for introducing novel genetic material into these
flies.
P elements are transposons found in Drosophila and other insects, where they cause hybrid dysgenesis. In flies carrying a P element, the frequency of
transposition is very low because of
synthesis of a repressor protein encoded by the resident P element. When
P-carrying males are crossed with P-negative females, the transposition
frequency in the fertilized egg is very high for a brief period, due to lack of
repressor. Random insertion of P elements then causes a high mutation rate and
lowers the proportion of viable offspring, that is, hybrid dysgenesis.
P elements are flanked by
perfect 31 base-pair inverted repeats. Any DNA sequence that is included
between these inverted repeats will be transposed. Therefore engineered P
elements can be used to introduce any sequence of DNA into a strain of fruit
flies or other susceptible insects.
DNA may be microinjected into embryos of P-negative strains of Drosophila. In fruit flies the diploid nucleus resulting from fusion of the sperm and egg nuclei divides multiple times without cell division, resulting in a giant cell with many nuclei, known as a syncytium. Microinjection is normally done at this stage, and incoming DNA usually integrates into at least some of the nuclei that will give rise to the future germline cells (Fig. 15.16). These nuclei are clustered at one end of the fertilized egg—the posterior end. The nuclei then migrate to the outer membrane where a cleavage furrow forms around each nucleus. These furrows expand to form individual cells for each nucleus. The center part remains undivided and acts as a yolk, providing nutrients to the developing larva.
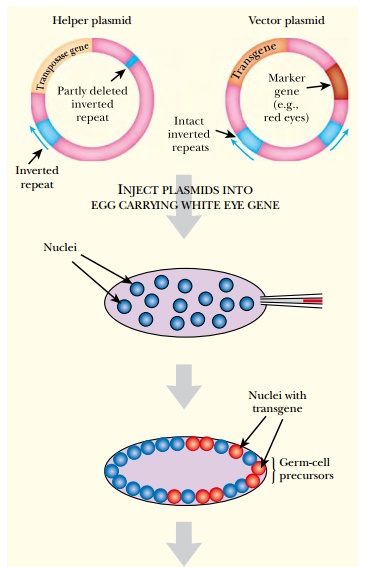

The incoming P element is normally carried on a bacterial plasmid that was constructed in a bacterial host. The P element transposes into the Drosophila chromosomes, and the plasmid sequences are left behind. In practice, two P elements are often used. One, the helper, provides the transposase but cannot itself move because of defective 31 base-pair inverted repeats (see Fig. 15.16). The other P element, the vector, carries the desired transgene and has intact 31 base-pair inverted repeats, but lacks the transposase gene. Transposition of the vector depends on transposase made by the helper. Once the P element vector has inserted into a particular location on the insect chromosome, it cannot move in future cell generations, because it has no transposase of its own. Ideally, it will be inherited stably.
The presence of the P element
is monitored by appropriate marker genes. Selectable markers used in flies
include neo (neomycin resistance) and
adh (alcohol dehydrogenase).
Alternatively, eye color genes may be used to reveal the presence of a P element
vector. Eye color cannot be positively selected; instead, flies are screened
for changes in eye color. For example, flies defective in the rosy
gene may be used as host. These
flies have brown eyes because of lack of xanthine dehydrogenase, which is
involved in synthesis of red eye pigment. If a wild-type copy of the rosy genes is included in the P element
vector, it will restore the red eye color. If the offspring of a rosy−/− transgenic fly has red eyes, this implies that
the transgene was inserted into the germline, and all the cells in the
offspring will have the transgene.
Related Topics