Chapter: Biotechnology Applying the Genetic Revolution: Transgenic Animals
Alternative Approaches to Making Transgenic Animals
ALTERNATIVE
APPROACHES TO MAKING TRANSGENIC ANIMALS
Although nuclear injection was the first approach used to make transgenic animals, and is still the most widely used, there are several alternative procedures. As discussed, engineered retroviruses have been used in gene therapy to introduce DNA into the chromosomes of animal cells. Retroviruses can infect the cells of early embryos, including embryonic stem cells (see later discussion). Hence, retrovirus vectors may introduce transgenes.
The advantages of using a
retrovirus are that only a single copy of the retrovirus plus transgene is
integrated into the genome. In addition, use of retroviruses does not require
skill in microinjection. The retrovirus carrying the transgene construct is
added to the fertilized egg and allowed to infect as normal. The egg is then
transplanted into a pseudopregnant female mouse. The remainder of the procedure
is as before.
Disadvantages are that virus
DNA is introduced along with the transgene and that retroviruses can carry only
limited amounts of DNA. Furthermore, founder animals made using retroviruses
are always chimeras, because insertion of the virus does not occur exactly when
the nuclei fuse. Consequently, retroviruses are rarely used in attempts to
create fully transgenic animals. On the other hand, partially transgenic
animals that have some sectors or tissues altered are useful because the
transgenic tissues may be compared with normal tissue within the same animal.
This can eliminate any doubts on whether the defects or changes caused by the
transgene are merely due to differences among animals or truly due to transgene
expression.
Embryonic stem cells may also be used to generate transgenic animals. Stem cells are the precursor cells to particular tissues of the body. Embryonic stem cells are derived from the blastocyst, a very early stage of the embryo, and retain the ability to develop into any body tissue, including the germline. Embryonic stem cells can be cultured and DNA can be introduced as for any cultured cell line. For successful creation of transgenic animals, embryonic stem cells must be maintained under conditions that avoid differentiation.
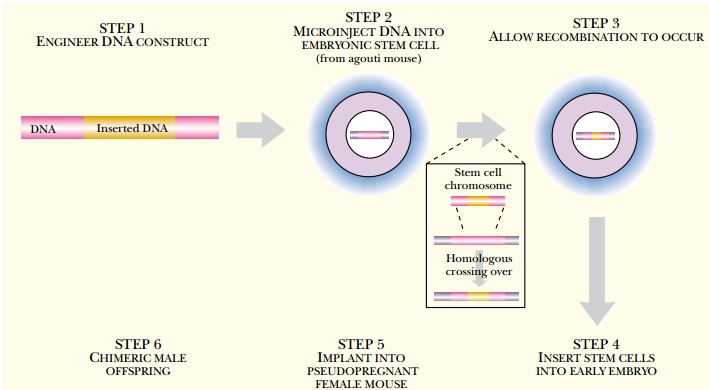
 Engineered embryonic stem
cells are then inserted into the central cavity of an early embryo at the
blastocyst stage (Fig. 15.5). This creates a mixed embryo and results in an
animal that is a genetic chimera consisting of some transgenic tissues and
others that are normal.
Engineered embryonic stem
cells are then inserted into the central cavity of an early embryo at the
blastocyst stage (Fig. 15.5). This creates a mixed embryo and results in an
animal that is a genetic chimera consisting of some transgenic tissues and
others that are normal.
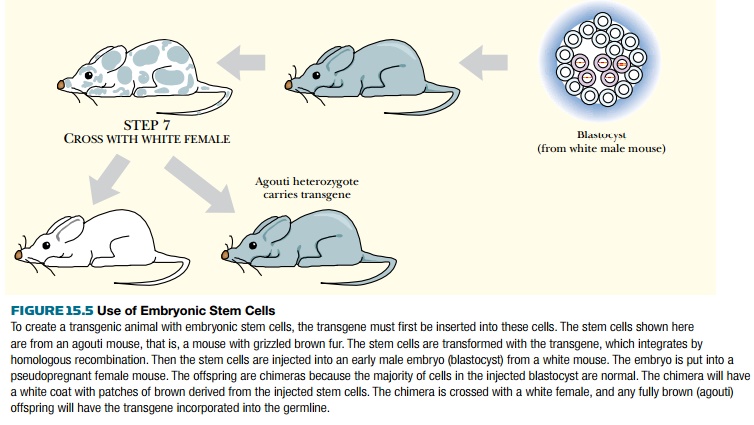
If the host embryo and the
embryonic stem cells are from different genetic lines with different fur
colors, the result is an animal with a patchwork coat. This allows the
transgenic sectors of the animal to be identified easily. This chimeric founder
animal must then be mated with a wild-type animal. If the embryonic stem cells
have contributed to the germline, then the coat color characteristic of this
cell line will be transmitted to the offspring. In mice black (recessive) and agouti (dominant) coat colors are often
used. The males because the resulting male chimeras can father many children
when crossed with wild-type females.
Related Topics