Chapter: Essential Anesthesia From Science to Practice : Applied physiology and pharmacology : A brief pharmacology related to anesthesia
The local anesthetics
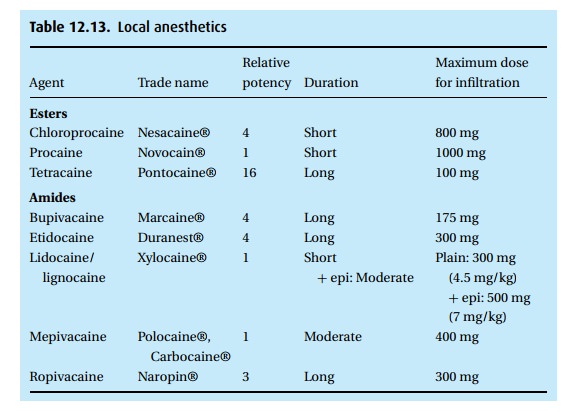
The local anesthetics (Table 12.13)
Instead
of flooding the whole system, from head to toe, with an inhalation or
intravenous anesthetic, we can inject an anesthetic locally; directly on a
nerve; place it into the epidural or subarachnoid space, catching several
nerves at once; or paint or spray it on mucous membrane as a topical
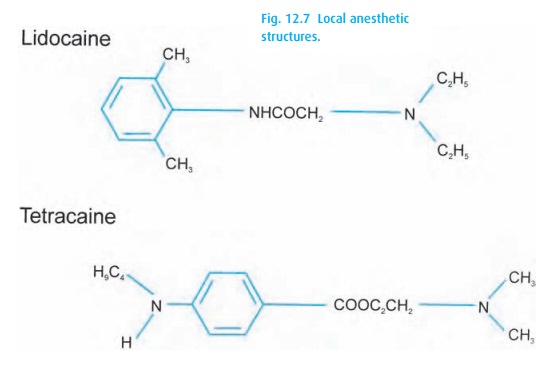
anesthetic. Local anesthetics come in two chemical classes: esters and amides,
with tetracaine (Pontocaine®)
A trick for remembering the class of local
anesthetics: if there is an ‘I’ before the “caine” it is an amIde. The trick to
the trick, though, is this only works for the generic name of the drug, e.g., bupivacaine is an amide, even when
found in a bottle labeled Marcaine®.
Local
anesthetics interfere with nerve conduction by blocking ion fluxes through
sodium channels. This blockade occurs from the inside of the cell. Local agents
are weak bases with pKb (pH at which half of the base is ionized) values
between 8 and 9; at a lower pH, more of the drug will be ionized and vice
versa. Only the lipid-soluble, non-ionized form can penetrate cell membranes.
Once inside, the cationic form of the drug is favored because the interior of
the cell tends to be more acidic than the outside. This is fortuitous, since
the cationic form will go to work on the ion channel. In general, an acidic
medium – for exam-ple, inflamed tissue – will favor ionization and thus delay
penetration of the drug, while an alkaline medium (such as adding bicarbonate
to a highly acidic com-mercial preparation of lidocaine) can hasten the
movement of the drug through membranes.
Different
nerves exhibit different sensitivities to local anesthetics. We see the
clinical evidence of this during spinal anesthesia where the block for cold
sensa-tion and sympathetic activity extends to higher dermatome levels than for
other sensations and motor activity. This is commonly attributed to resistance
to block-ade provided by the thick, heavy myelin sheath coating the motor (Aα)
fibers, which is lacking on the skinny non-myelinated preganglionic sympathetic
(B) fibers and postganglionic sympathetic and dorsal root (C) fibers. However,
the picture is quite complex. The sensitivity will also be influenced by the
position of the nerve in a nerve bundle exposed to the local anesthetic, the
speed of nerve conduction, and by how much of the nerve must be exposed to the
anesthetic to block it.
Once
injected or applied to a membrane, the drug will be carried away by the blood.
To delay this, we often add epinephrine to the local anesthetic, which
con-stricts blood vessels, thus decreasing tissue perfusion and prolonging the
local anesthetic effect. It does not take much epinephrine. Solutions of as
little as 1 to 800 000 have been found to do the trick. However, frequently we
add epinephrine (adrenaline) in a concentration of 1 to 200 000 so that if we
inject into the blood stream (rather than around the nerve), the patient will
get a little tachycardia, alert-ing us to stop the injection immediately.
Greater epinephrine concentrations will not further prolong the local
anesthetic effect, but will cause more tachycardia (experienced by patients as
“butterflies in the stomach,” headache, and appre-hension). The drugs are
metabolized according to their structure: the esters fall prey to plasma
cholinesterase and undergo hydrolysis. Microsomal enzymes in the liver go to
work on the amides. Occasionally, the products of biotransforma-tion of local
anesthetics cause mischief, for example some patients are allergic to para-aminobenzoic acid, which forms
during ester hydrolysis. Methemoglobin-emia (and reduced oxygen carrying
capacity) has been observed after the use of prilocaine (Citanest®) and
benzocaine, the latter a topical anesthetic (with a sad history of causing
contact dermatitis) found in some sprays.
Lidocaine
has seen widespread use as an antiarrhythmic drug. Its mechanism of action as a
local anesthetic also works on the heart muscle where it can block sodium
channels. This can explain its effect on phase IV depolarization, and thus
decreased excitability and automaticity. The therapeutic effect of small
intra-venous doses of lidocaine (1 mg/kg as a bolus or 40 mcg/kg/min as an
infusion – up or down titrated to effect) alert us to the fact that local
anesthetics do have cardiac effects, not all of which are welcomed. Dangerous
cardiac toxicity (hypotension, A–V block, ventricular fibrillation) has been
triggered by bupivacaine mistakenly injected intravenously. All local
anesthetics can have such cardiac toxicity; how-ever, it is a particular
problem with bupivacaine as its duration of binding with sodium receptors is
much longer than that of other agents. Importantly, victims of
bupivacaine-induced cardiac toxicity have survived after prolonged resuscitation.
Local
anesthetics will also affect the central nervous system when injected
intravenously or when a large peripheral dose is rapidly absorbed. Thus, both
procaine and lidocaine have been used as intravenous anesthetics. However,
their margin of safety is too narrow to recommend their routine use. With
overdose, convulsions are common. As many as 4 out of 1000 patients might
exhibit some CNS excitation during a regional local anesthetic. Typically, the
patients complain of numbness around mouth and tongue, dizziness, tingling, and
tinnitus, and they often become restless before seizing. We treat seizures with
manual ventilation with oxygen and a small intravenous dose of, for example,
thiopental (20 to 50 mg bolus for the average adult) or midazolam (1 mg bolus).
We have a large selection of local
anesthetics available. The drugs differ primar-ily in their duration of action.
Depending on dose and concentration, we have at our disposal everything from
the long-acting tetracaine (Pontocaine®), bupiva-caine (Marcaine®) and
etidocaine (Duranest®), to the short-acting chloropro-caine (Citanest®) and
procaine (Novocain®). Lidocaine and mepivacaine fit into the intermediate
category.
Additives
Bicarbonate
As
mentioned above, we add bicarbonate to those drugs prepared at a particularly
acidic pH (lidocaine, chloroprocaine) to speed onset of anesthesia (it also
reduces burning when making a skin wheal).
Epinephrine
We might
add epinephrine to the local anesthetic solution to (i) prolong the dur-ation
of anesthesia, particularly for vasodilating local anesthetics such as
lido-caine; (ii) reduce peak plasma concentration of the local anesthetic, also
more important for vasodilating agents; (iii) increase the density of regional
anesthetic
blocks
(by an unknown mechanism); and (iv) as a marker for intravascular injec-tion.
Because of epinephrine instability in an alkaline environment, commercial local
anesthetic preparations containing epinephrine are highly acidic. We can add
bicarbonate, and/or use plain local anesthetics to which we add epinephrine
ourselves. Remember that 1:200 000 epinephrine is only 5 mcg/mL – use a
tuber-culin syringe and measure carefully! Important note: because we fear
necrosis of the tip we do not add epinephrine to blocks placed at an “end
organ,” e.g., digits, penis, nose, ears.
Clonidine (Catapres®)
Through
unclear mechanisms, small doses of clonidine enhance and prolong regional
anesthesia. One mcg/kg added to the local anesthetic for a Bier block appears
to delay the onset of tourniquet pain. In epidural and spinal anesthesia, 50 to
75 mcg clonidine has been found to augment the effect of both local anesthetics
and opioids.
Opioids
We add
opioids to neuraxial anesthetics to prolong the analgesic effect. Man-ageable
side effects include itching, nausea, and vomiting. Respiratory depres-sion,
though less common, concerns us greatly, and we usually employ pulse oximetry
on the post-surgical ward. Neuraxial morphine carries a risk of delayed
respiratory depression, so we continue to monitor about 24 hours after the last
dose.
Related Topics